Abstract
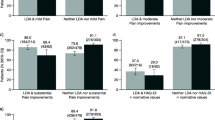
The objective of the study was to investigate relations on group level and agreements on the individual patient level between changes in fatigue, pain and patient global assessment (PaGl) assessed on visual analogue scales (VAS) in patients with rheumatoid arthritis (RA) after initiating or switching biological treatment. Associations with other disease measures were also examined. Traditional disease activity data on 177 patients with RA registered before and after 6-month treatment were extracted from the Danish DANBIO registry. Associations were examined using multiple regression analysis. Agreement between the VAS score changes (∆) was expressed as the bias (mean difference) and the 95 % lower and upper limits of agreement (LoA). All disease measures improved significantly. ∆fatigue, ∆pain and ∆PaGl were independently associated with each other (r partial range 0.38–0.81, p < 0.0001), but not to a significant degree with changes in other measures. Lower and upper LoA [bias] for ∆fatigue versus ∆pain was −44.0 and 51.8 [3.9], for ∆fatigue versus ∆PaGl −38.2 and 52.4 [4.2], and for ∆PaGl versus ∆pain −34.3 and 34.3 [0.0]. ∆fatigue, ∆pain and ∆PaGl were independently but weakly predicted by their own baseline values (r partial range −0.30 to −0.46, p < 0.0001). In conclusion, changes in fatigue, pain and PaGl were independently associated and nearly identical on group level but agreements were poor in individual patients. The changes were poorly explained by other potential predictor variables and by baseline values. The results expose the unpredictable nature of patient-reported VAS scores in individual patients with RA.


Similar content being viewed by others
References
Alamanos Y, Voulgari PV, Drosos AA (2006) Incidence and prevalence of rheumatoid arthritis, based on the 1987 American College of Rheumatology criteria: a systematic review. Semin Arthritis Rheum 36:182–188
Grøn KL, Ornbjerg LM, Hetland ML, Aslam F, Khan NA, Jacobs JW et al (2014) The association of fatigue, comorbidity burden, disease activity, disability and gross domestic product in patients with rheumatoid arthritis. Results from 34 countries participating in the Quest-RA program. Clin Exp Rheumatol 32:869–877
Van Hoogmoed D, Fransen J, Bleijenberg G, van Riel P (2010) Physical and psychosocial correlates of severe fatigue in rheumatoid arthritis. Rheumatology (Oxford) 49:1294–1302
Hewlett S (2011) Fatigue in rheumatoid arthritis: time for a conceptual model. An under-acknowledged patient concern requires a new approach. Rheumatology 50:1004–1006
Pollard LC, Choy EH, Gonzalez J, Khoshaba B, Scott DL (2006) Fatigue in rheumatoid arthritis reflects pain, not disease activity. Rheumatology 45:885–889
Thyberg I, Dahlstrom O, Thyberg M (2009) Factors related to fatigue in women and men with early rheumatoid arthritis: the Swedish TIRA study. J Rehabil Med 41:904–912
Huyser BA, Parker JC, Thoreson R, Smarr KL, Johnson JC, Hoffman R (1998) Predictors of subjective fatigue among individuals with rheumatoid arthritis. Arthritis Rheum 41:2230–2237
Egsmose EL, Madsen OR (2015) Interplay between patient global assessment, pain, and fatigue and influence of other clinical disease activity measures in patients with active rheumatoid arthritis. Clin Rheumatol 34:1187–1194
Belza BL, Henke CJ, Yelin EH, Epstein WV, Gilliss CL (1993) Correlates of fatigue in older adults with rheumatoid arthritis. Nurs Res 42:93–99
Studenic P, Radner H, Smolen JS, Aletaha D (2012) Discrepancies between patients and physicians in their perceptions of rheumatoid arthritis disease activity. Arthritis Rheum 64:2814–2823
Kalyoncu U, Dougados M, Daures JP, Gossec L (2009) Reporting of patient-reported outcomes in recent trials in rheumatoid arthritis: a systematic literature review. Ann Rheum Dis 68:183–190
Kirwan JR, Hewlett SE, Heiberg T, Hughes RA, Carr M, Hehir M et al (2005) Incorporating the patient perspective into outcome assessment in rheumatoid arthritis—progress at OMERACT 7. J Rheumatol 32:2250–2256
Pincus T, Amara I, Segurado OG, Bergman M, Koch GG (2008) Relative efficiencies of physician/assessor global estimates and patient questionnaire measures are similar to or greater than joint counts to distinguish adalimumab from control treatments in rheumatoid arthritis clinical trials. J Rheumatol 35:201–205
Hazes JM, Taylor P, Strand V, Purcaru O, Coteur G, Mease P (2010) Physical function improvements and relief from fatigue and pain are associated with increased productivity at work and at home in rheumatoid arthritis patients treated with certolizumab pegol. Rheumatology (Oxford) 49:1900–1910
Yazici Y, Bergman M, Pincus T (2008) Time to score quantitative rheumatoid arthritis measures: 28-joint count, Disease Activity Score, Health Assessment Questionnaire (HAQ), Multidimensional HAQ (MDHAQ), and Routine Assessment of Patient Index Data (RAPID) scores. J Rheumatol 35:603–609
Fraenkel L, McGraw S (2007) What are the essential elements to enable patient participation in medical decision making? J Gen Intern Med 22:614–619
Sanderson T, Morris M, Calnan M, Richards P, Hewlett S (2010) Patient perspective of measuring treatment efficacy: the rheumatoid arthritis patient priorities for pharmacologic interventions outcomes. Arthritis Care Res (Hoboken) 62:647–656
Kirwan JR, Minnock P, Adebajo A, Bresnihan B, Choy E, de Wit M et al (2007) Patient perspective: fatigue as a recommended patient centered outcome measure in rheumatoid arthritis. J Rheumatol 34:1174–1177
Kirwan JR, Hewlett S (2007) Patient perspective: reasons and methods for measuring fatigue in rheumatoid arthritis. J Rheumatol 34:1171–1173
Felson DT, Anderson JJ, Boers M, Bombardier C, Chernoff M, Fried B et al (1993) The American College of Rheumatology preliminary core set of disease activity measures for rheumatoid arthritis clinical trials. Arthritis Rheum 36:729–740
Aletaha D, Landewe R, Karonitsch T, Bathon J, Boers M, Bombardier C et al (2008) Reporting disease activity in clinical trials of patients with rheumatoid arthritis: EULAR/ACR collaborative recommendations. Arthritis Rheum 59:1371–1377
Khan NA, Spencer HJ, Abda EA, Alten R, Pohl C, Ancuta C et al (2012) Patient’s global assessment of disease activity and patient’s assessment of general health for rheumatoid arthritis activity assessment: Are they equivalent? Ann Rheum Dis 71:1942–1949
Amaya-Amaya J, Botello-Corzo D, Calixto OJ, Calderón-Rojas R, Domínguez AM, Cruz-Tapias P et al (2012) Usefulness of patients-reported outcomes in rheumatoid arthritis focus group. Arthritis 2012:935187
Nikolaus S, Bode C, Taal E, van de Laar MA (2013) Fatigue and factors related to fatigue in rheumatoid arthritis: a systematic review. Arthritis Care Res (Hoboken) 65:1128–1146
Ward MM, Guthrie LC, Alba MI (2015) Measures of arthritis activity associated with patient-reported improvement in rheumatoid arthritis when assessed prospectively versus retrospectively. Arthritis Care Res (Hoboken) 67:776–781
Minnock P, McKee G, Bresnihan B, FitzGerald O, Veale DJ (2014) How much is fatigue explained by standard clinical characteristics of disease activity in patients with inflammatory arthritis? A longitudinal study. Arthritis Care Res (Hoboken) 66:1597–1603
Druce KL, Jones GT, Macfarlane GJ, Basu N (2015) Patients receiving anti-TNF therapies experience clinically important improvements in RA-related fatigue: results from the British Society for Rheumatology Biologics Register for Rheumatoid Arthritis. Rheumatology (Oxford) 54:964–971
Hetland ML, Christensen IJ, Tarp U, Dreyer L, Hansen A, Hansen IT et al (2010) Direct comparison of treatment responses, remission rates, and drug adherence in patients with rheumatoid arthritis treated with adalimumab, etanercept, or infliximab. Arthritis Rheum 62:22–32
Hetland ML (2011) DANBIO—powerful research database and electronic patient record. Rheumatology (Oxford) 50:69–77
Aletaha D, Smolen J (2005) The Simplified Disease Activity Index (SDAI) and the Clinical Disease Activity Index (CDAI): a review of their usefulness and validity in rheumatoid arthritis. Clin Exp Rheumatol 23(Suppl 39):S100–S108
Madsen OR (2013) Agreement between the DAS28-CRP assessed with 3 and 4 variables in patients with rheumatoid arthritis treated with biological agents in the daily clinic. J Rheumatol 40:379–385
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1:307–310
Hallahan M, Rosenthal R (1996) Statistical power: concepts, procedures, and applications. Behav Res Ther 34:489–499
Fransen J, Antoni C, Mease PJ, Uter W, Kavanaugh A, Kalden JR et al (2006) Performance of response criteria for assessing peripheral arthritis in patients with psoriatic arthritis: analysis of data from randomised controlled trials of two tumour necrosis factor inhibitors. Ann Rheum Dis 65:1373–1378
Wolfe F (2004) Fatigue assessments in rheumatoid arthritis: comparative performance of visual analog scales and longer fatigue questionnaires in 7760 patients. J Rheumatol 31:1896–1902
Wells G, Li T, Maxwell L, Maclean R, Tugwell P (2008) Responsiveness of patient reported outcomes including fatigue, sleep quality, activity limitation, and quality of life following treatment with abatacept for rheumatoid arthritis. Ann Rheum Dis 67:260–265
Minnock P, Kirwan J, Bresnihan B (2009) Fatigue is a reliable, sensitive and unique outcome measure in rheumatoid arthritis. Rheumatology 48:1533–1536
Sokka T, Pincus T (2005) Quantitative joint assessment in rheumatoid arthritis. Clin Exp Rheumatol 23(Suppl 39):S58–S62
Campbell RC, Batley M, Hammond A, Ibrahim F, Kingsley G, Scott DL (2012) The impact of disease activity, pain, disability and treatments on fatigue in established rheumatoid arthritis. Clin Rheumatol 31:717–722
Higgins NC, Bailey SJ, LaChapelle DL, Harman K, Hadjistavropoulos T (2015) Coping styles, pain expressiveness, and implicit theories of chronic pain. J Psychol 149:737–750
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interests
OR Madsen has received research grants and/or consultancy/speaker fees from Abbott, BMS, Celgene, MSD, Novartis, Pfizer, Roche and UCB. EM Egsmose declares no conflicts of interest.
Ethical approval
All procedures were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. According to Danish law, ethical approval was not required for the present study.
Informed consent
According to Danish law, informed consent was not required for the present study.
Rights and permissions
About this article
Cite this article
Madsen, O.R., Egsmose, E.M. Fatigue, pain and patient global assessment responses to biological treatment are unpredictable, and poorly inter-connected in individual rheumatoid arthritis patients followed in the daily clinic. Rheumatol Int 36, 1347–1354 (2016). https://doi.org/10.1007/s00296-016-3535-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-016-3535-y