Abstract
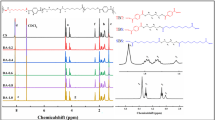
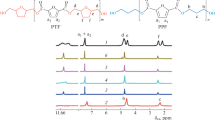
The aim of the study was to study the effect of cheap bio-based monomer 2,5-thiophenedicarboxylic acid on poly(butylene furandicarboxylate). Bio-based copolyesters were successfully synthesized by two-step melt polycondensation using 2,5-thiophenedicarboxylic acid (TDCA), 2,5-furandicarboxylic acid (FDCA) and 1,4-butanediol (BDO). Compared with FDCA, the price of TDCA is lower. The main purpose of this study is to add different amounts of TDCA to reduce the cost of polyester and to investigate the effect of TDCA on the properties of poly(butylene furandicarboxylate) (PBF). It can be determined that the expected copolymers were indeed synthesized by FTIR and 1H NMR spectra. DSC and WAXD results suggested that PBFTF27, PBFTF51 and PBFTF77 show isodimorphism behaviors. Because the angle between two carboxyl groups in TDCA is larger than that of FDCA, and the electronegativity of sulfur atom is weaker than that of oxygen atom, the glass transition temperature decreases due to the increase in the flexibility of the chain with the increase in TDCA. Compared with PBF, the toughness of poly(butylene 2,5-thiophenedicarboxylate) (PBTF) and copolyesters increased significantly. Poly(butylene furandicarboxylate-co-butylene 2,5-thiophenedicarboxylate) can be used in the packaging applications.













Similar content being viewed by others
References
Gandini A, Lacerda TM, Carvalho AJF, Trovatti E (2016) Progress of polymers from renewable resources: furans, vegetable oils, and polysaccharides. Chem Rev 116(3):1637–1669
Wang GQ, Jiang M, Zhang Q, Wang R, Liang QD, Wang HH, Zhou GY (2019) Partially bio-based and tough polyesters, poly(ethylene 2,5-thiophenedicarboxylate-co-1,4-cyclohexanedimethylene 2,5-thiophenedicarboxylate)s. Express Polym Lett 13:938–947
Wang G, Song J (2021) Synthesis and characterization of bio-based polyesters derived from 1,10-decanediol. J Appl Polym Sci 138(39):51163
Chatterjee C, Pong F, Sen A (2015) Chemical conversion pathways for carbohydrates. Green Chem 17(1):40–71
Jain A, Jonnalagadda SC, Ramanujachary KV, Mugweru A (2015) Selective oxidation of 5-hydroxymethyl-2-furfural to furan-2,5-dicarboxylic acid over spinel mixed metal oxide catalyst. Catal Commun 58:179–182
van Putten R-J, van der Waal JC, de Jong E, Rasrendra CB, Heeres HJ, de Vries JG (2013) Hydroxymethylfurfural, A Versatile Platform Chemical Made from Renewable Resources. Chem Rev 113(3):1499–1597
Desroches M, Benyahya S, Besse V, Auvergne R, Boutevin B, Caillol S (2014) Synthesis of bio-based building blocks from vegetable oils: A platform chemicals approach. Lipid Technol 26(2):35–38
S Natesan, JS Samuel, AK Srinivasan (2021) Design and development of Schiff’s base (SB)-modified polylactic acid (PLA) antimicrobial film for packaging applications. Polym Bull
N More, M Avhad, S Utekar, A More (2022) Polylactic acid (PLA) membrane—significance, synthesis, and applications: a review. Polym Bull
Garcia-Campo MJ, Quiles-Carrillo L, Sanchez-Nacher L, Balart R, Montanes N (2019) High toughness poly(lactic acid) (PLA) formulations obtained by ternary blends with poly(3-hydroxybutyrate) (PHB) and flexible polyesters from succinic acid. Polym Bull 76(4):1839–1859
Adorna JA, Ventura RLG, Dang VD, Doong R-A, Ventura J-RS (2022) Biodegradable polyhydroxybutyrate/cellulose/calcium carbonate bioplastic composites prepared by heat-assisted solution casting method. J Appl Polym Sci 139(7):51645
Mokhena TC, Sadiku ER, Ray SS, Mochane MJ, Motaung TE (2021) The effect of expanded graphite/clay nanoparticles on thermal, rheological, and fire-retardant properties of poly(butylene succinate). Polym Compos 42(12):6370–6382
Hallstein J, Gomoll A, Lieske A, Büsse T, Balko J, Brüll R, Malz F, Metzsch-Zilligen E, Pfaendner R, Zehm D (2021) Unraveling the cause for the unusual processing behavior of commercial partially bio-based poly(butylene succinates) and their stabilization. J Appl Polym Sci 138(28):50669
Salusjärvi L, Havukainen S, Koivistoinen O, Toivari M (2019) Biotechnological production of glycolic acid and ethylene glycol: current state and perspectives. Appl Microbiol Biotechnol 103(6):2525–2535
Mincheva R, Delangre A, Raquez J-M, Narayan R, Dubois P (2013) Biobased Polyesters with Composition-Dependent Thermomechanical Properties: Synthesis and Characterization of Poly(butylene succinate-co-butylene azelate). Biomacromol 14(3):890–899
Muñoz-Guerra S, Lavilla C, Japu C, Martínez de Ilarduya A (2014) Renewable terephthalate polyesters from carbohydrate-based bicyclic monomers. Green Chem 16(4):1716–1739
Gomes M, Gandini A, Silvestre AJD, Reis B (2011) Synthesis and characterization of poly(2,5-furan dicarboxylate)s based on a variety of diols. J Polym Sci, Part A Polym Chem 49(17):3759–3768
Veryasov G, Matsumoto K, Hagiwara R (2015) The Discrete AlF52–Fluoroaluminate Anion in the Structure of [Tetraethylammonium] 2[AlF5](H2O)2. Eur J Inorg Chem 2015(32):5306–5310
Miller SA (2013) Sustainable polymers: opportunities for the next decade. ACS Macro Lett 2(6):550–554
Sousa AF, Vilela C, Fonseca AC, Matos M, Freire CSR, Gruter G-JM, Coelho JFJ, Silvestre AJD (2015) Correction: biobased polyesters and other polymers from 2,5-furandicarboxylic acid: a tribute to furan excellency. Polym Chem 6(33):6096–6096
Knoop RJI, Vogelzang W, van Haveren J, van Es DS (2013) High molecular weight poly(ethylene-2,5-furanoate); critical aspects in synthesis and mechanical property determination. J Polym Sci, Part A Polym Chem 51(19):4191–4199
Burgess SK, Kriegel RM, Koros WJ (2015) Carbon dioxide sorption and transport in amorphous poly(ethylene furanoate). Macromolecules 48(7):2184–2193
van Berkel JG, Guigo N, Kolstad JJ, Sipos L, Wang B, Dam MA, Sbirrazzuoli N (2015) Isothermal crystallization kinetics of poly (ethylene 2,5-furandicarboxylate). Macromol Mater Eng 300(4):466–474
Araujo CF, Nolasco MM, Ribeiro-Claro PJA, Rudić S, Silvestre AJD, Vaz PD, Sousa AF (2018) Inside PEF: chain conformation and dynamics in crystalline and amorphous domains. Macromolecules 51(9):3515–3526
Soccio M, Martínez-Tong DE, Alegría A, Munari A, Lotti N (2017) Molecular dynamics of fully biobased poly(butylene 2,5-furanoate) as revealed by broadband dielectric spectroscopy. Polymer 128:24–30
Sousa AF, Guigo N, Pozycka M, Delgado M, Soares J, Mendonca PV, Coelho JFJ, Sbirrazzuoli N, Silvestre AJD (2018) Tailored design of renewable copolymers based on poly(1,4-butylene 2,5-furandicarboxylate) and poly(ethylene glycol) with refined thermal properties. Polym Chem-Uk 9(6):722–731
Jiang M, Liu Q, Zhang Q, Ye C, Zhou G (2012) A series of furan-aromatic polyesters synthesized via direct esterification method based on renewable resources. J Polym Sci, Part A: Polym Chem 50(5):1026–1036
Zhu J, Cai J, Xie W, Chen P-H, Gazzano M, Scandola M, Gross RA (2013) Poly(butylene 2,5-furan dicarboxylate), a biobased alternative to PBT: synthesis Physical Properties, and Crystal Structure. Macromolecules 46(3):796–804
Vannini M, Marchese P, Celli A, Lorenzetti C (2015) Fully biobased poly(propylene 2,5-furandicarboxylate) for packaging applications: excellent barrier properties as a function of crystallinity. Green Chem 17(8):4162–4166
Dai Z, Guo F, Zhang S, Zhang W, Yang Q, Dong W, Jiang M, Ma J, Xin F (2020) Bio-based succinic acid: an overview of strain development, substrate utilization, and downstream purification. Biofuels Bioprod Biorefin 14(5):965–985
Papageorgiou GZ, Tsanaktsis V, Papageorgiou DG, Exarhopoulos S, Papageorgiou M, Bikiaris DN (2014) Evaluation of polyesters from renewable resources as alternatives to the current fossil-based polymers Phase transitions of poly(butylene 2,5-furan-dicarboxylate). Polymer 55(16):3846–3858
Wu L, Mincheva R, Xu Y, Raquez J-M, Dubois P (2012) High molecular weight poly(butylene succinate-co-butylene furandicarboxylate) Copolyesters: from catalyzed polycondensation reaction to thermomechanical properties. Biomacromol 13(9):2973–2981
Wu B, Xu Y, Bu Z, Wu L, Li B-G, Dubois P (2014) Biobased poly(butylene 2,5-furandicarboxylate) and poly(butylene adipate-co-butylene 2,5-furandicarboxylate)s: From synthesis using highly purified 2,5-furandicarboxylic acid to thermo-mechanical properties. Polymer 55(16):3648–3655
Zheng M, Zang X, Wang G, Wang P, Lu B, Ji J (2017) Poly(butylene 2,5-furandicarboxylate-ε-caprolactone): a new bio-based elastomer with high strength and biodegradability. Express Polym Lett 11:611–621
Soccio M, Costa M, Lotti N, Gazzano M, Siracusa V, Salatelli E, Manaresi P, Munari A (2016) Novel fully biobased poly(butylene 2,5-furanoate/diglycolate) copolymers containing ether linkages: Structure-property relationships. Eur Polym J 81:397–412
Hu H, Zhang R, Wang J, Ying WB, Shi L, Yao C, Kong Z, Wang K, Zhu J (2019) A mild method to prepare high molecular weight poly(butylene furandicarboxylate-co-glycolate) copolyesters: effects of the glycolate content on thermal, mechanical, and barrier properties and biodegradability. Green Chem 21(11):3013–3022
Diao L, Su K, Li Z, Ding C (2016) Furan-based co-polyesters with enhanced thermal properties: poly(1,4-butylene-co-1,4-cyclohexanedimethylene-2,5-furandicarboxylic acid). Rsc Adv 6(33):27632–27639
Ouyang Q, Liu J, Li C, Zheng L, Xiao Y, Wu S, Zhang B (2019) A facile method to synthesize bio-based and biodegradable copolymers from furandicarboxylic acid and isosorbide with high molecular weights and excellent thermal and mechanical properties. Polym Chem Uk 10(41):5594–5601
Wang G, Liang Y, Jiang M, Zhang Q, Wang R, Wang H, Zhou G (2020) High Tg and tough poly(butylene 2,5-thiophenedicarboxylate-co-1,4-cyclohexanedimethylene 2,5-thiophenedicarboxylate)s: Synthesis and characterization. J Appl Polym Sci 137(18):48634
Ma J, Pang Y, Wang M, Xu J, Ma H, Nie X (2012) The copolymerization reactivity of diols with 2,5-furandicarboxylic acid for furan-based copolyester materials. J Mater Chem 22(8):3457–3461
Ma J, Yu X, Xu J, Pang Y (2012) Synthesis and crystallinity of poly(butylene 2,5-furandicarboxylate). Polymer 53(19):4145–4151
Guidotti G, Gigli M, Soccio M, Lotti N, Gazzano M, Siracusa V, Munari A (2018) Poly(butylene 2,5-thiophenedicarboxylate): An Added Value to the Class of High Gas Barrier Biopolyesters. Polymers 10:167
Guidotti G, Gigli M, Soccio M, Lotti N, Gazzano M, Siracusa V, Munari A (2018) Ordered structures of poly(butylene 2,5-thiophenedicarboxylate) and their impact on material functional properties. Eur Polym J 106:284–290
Acknowledgements
The authors greatly acknowledge the financial supports from “14th five-year” Science and Technology Research Program of the Education Department of Jilin Province (JJKH20210254KJ) and Development Foundation of Science and Technology in Jilin Province of China (20200401032GX).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, G., Dong, Y., Hao, X. et al. Bio-based poly(butylene furandicarboxylate-co-butylene 2,5-thiophenedicarboxylate): synthesis, thermal properties, crystallization properties and mechanical properties. Polym. Bull. 80, 5373–5395 (2023). https://doi.org/10.1007/s00289-022-04330-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-022-04330-y