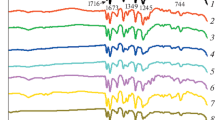
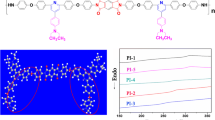
Abstract
A new series of six-membered ring sulfonated polyimides with different combinations of two comonomers in the nonsulfonated diamine was prepared by one-step high-temperature polycondensation in m-cresol to improve the solubility of the resulting sulfonated polyimides. They are based on 1,4,5,8-naphthalenetetracarboxylic dianhydride, 4,4′-diaminostilbene-2,2-disulfonic acid sulfonated diamine, and equimolar mixture of 3,5,3′,5′-tetramethylbenzidine (TMB) and 4,4′-oxydianiline, bis(4-aminophenyl)methane, or bis(4-(aminophenoxy)-4-phenyl)isopropylidene nonsulfonated diamines. The introduction of TMB comonomer in the nonsulfonated diamine resulted in a remarkable improvement in the solubility of the resulting polyimides in comparison with the corresponding single-monomer nonsulfonated diamine polyimides. Flexible, transparent, and tough membranes were prepared by solution casting method from the different polyimides. The membranes were characterized with FTIR and 1H-NMR spectroscopy, differential scanning calorimetry, thermogravimetric analysis, water uptake, and ion-exchange capacity measurements. They exhibit high thermal stability and good correlation between the ion-exchange capacity and water uptake values.








Similar content being viewed by others
References
Rikukawa M, Sanui K (2000) Proton-conducting polymer electrolyte membranes based on hydrocarbon polymers. Prog Polym Sci 25:1463–1502
Carrete L, Friedrich KA, Stimming U (2001) Fuel cells: fundamentals and applications. Fuel Cell 1:5–39
Peighambardoust SJ, Rowshanzamir S, Amjadi M (2010) Review of the proton exchange membranes for fuel cell applications. Int J Hydrogen Energy 35:9349–9384
Mauritz KA, Moore RB (2004) State of understanding of Nafion. Chem Rev 104:4535–4586
Steele BCH, Heinzel A (2001) Materials for fuel-cell technologies. Nature 414:345–352
Kreuer KD (2001) On the development of proton conducting polymer membranes for hydrogen and methanol fuel cells. J Membr Sci 185:29–39
Vogel C, Komber H, Quetschke A, Butwilowski W, Pötschke A, Schlenstedt K, Meier-Haack J (2011) Side-chain sulfonated random and multiblock poly(ether sulfone)s for PEM applications. React Funct Polym 71:828–842
Wang C, Li N, Shin DW, Lee SY, Kang NR, Lee YM, Guiver MD (2011) Fluorene-based poly(arylene ether sulfone)s containing clustered flexible pendant sulfonic acids as proton exchange membranes. Macromolecules 44:7296–7306
Chen Y, Lee CH, Rowlett JR, McGrath JE (2012) Synthesis and characterization of multiblock semi-crystalline hydrophobic poly(ether ether ketone)–hydrophilic disulfonated poly(arylene ether sulfone) copolymers for proton exchange membranes. Polymer 53:3143–3153
Shin DW, Lee SY, Lee CH, Lee KS, Park CH, McGrath JE, Zhang M, Moore RB, Lingwood MD, Madsen LA, Kim YT, Hwang I, Lee YM (2013) Sulfonated poly(arylene sulfide sulfone nitrile) multiblock copolymers with ordered morphology for proton exchange membranes. Macromolecules 46:7797–7804
Pandey RP, Thakur AK, Shahi VK (2014) Sulfonated polyimide/acid-functionalized graphene oxide composite polymer electrolyte membranes with improved proton conductivity and water-retention properties. ACS Appl Mater Interfaces 6(19):16993–17002
Ma L, Xu J, Han S, Yang M, Wang Z, Ni H, Gui Y (2014) Synthesis and characterization of sulfonated polymers containing triazoles as low-humidity proton exchange membranes. J Polym Res 21:551
Yao H, Feng P, Liu P, Liu B, Zhang Y, Guan S, Jiang Z (2015) Highly sulfonated co-polyimides containing crosslinkable hydrophobic tetrafluorostyrol side-groups for proton exchange membranes. Polym. Chem. 6:2626–2635
Al-Madani G, Kailani MH, Al-Hussein M (2015) Test system for through-plane conductivity measurements of hydrogen proton exchange membranes. Int J Electrochem Sci 10:6465–6475
Kobayashi T, Rikukawa M, Sanui K, Ogata N (1998) Proton-conducting polymers derived from poly(ether-etherketone) and poly(4-phenoxybenzoyl-1,4-phenylene). Solid State Ionics 106:219–225
Alberti G, Casciola M, Massinelli L, Bauer B (2001) Polymeric proton conducting membranes for medium temperature fuel cells (110–160 C). J Membr Sci 185:73–81
Jones DJ, Rozière J (2001) Recent advances in the functionalisation of polybenzimidazole and polyetherketone for fuel cell applications. J Membr Sci 185:41–58
Lufrano F, Gatto I, Staiti P, Antonucci V, Passalacqua E (2001) Sulfonated polysulfone ionomer membranes for fuel cells. Solid State Ionics 145:47–51
Genova-Dimitrova P, Baradie B, Foscallo F, Poinsignon C, Sanchez JY (2001) Ionomeric membranes for proton exchange membrane fuel cell (PEMFC): sulfonated polysulfone associated with phosphatoantimonic acid. J Membr Sci 185:59–71
Staiti P, Lufrano F, Arico AS, Passalacqua E, Antonucci V (2001) Sulfonated polybenzimidazole membranes—preparation and physico-chemical characterization. J Membr Sci 188:71–78
Wycisk R, Pintauro PN (1996) Sulfonated polyphosphazene ion-exchange membranes. J Membr Sci 119:155–160
Faure S, Cornet N, Gebel G, Mercier R, Pineri M, Sillion B (1997) New materials for fuel cell and modern battery systems. In: Savadogo O, Roberge PR (eds) Proceedings of the second international symposium on new materials for fuel cell and modern battery systems. Montreal, Juillet, pp 818
Genies C, Mercier R, Sillion B, Cornet N, Gebel G, Pineri M (2001) Soluble sulfonated naphthalenic polyimides as materials for proton exchange membranes. Polymer 42:359–373
Okamoto KI (2003) Sulfonated polyimides for polymer electrolyte membrane fuel cell. J Photopolym Sci Technol 16:247–254
Miyatake K, Asano N, Watanabe M (2003) Synthesis and properties of novel sulfonated polyimides containing 1,5-Naphthylene Moieties. J Polym Chem 41:3901–3907
Einsla B, Hong YT, Kim YS, Wang F, Gunduz N, McGrath J (2004) Sulfonated naphthalene dianhydride based polyimide copolymers for proton-exchange-membrane fuel cells. I. Monomer and copolymer synthesis. J Polym Sci Part A Polym Chem 42:862–874
Guo X, Fang J, Tanaka K, Kita H, Okamoto KI (2004) Synthesis and properties of novel sulfonated polyimides from 2,2′-bis(4-aminophenoxy)biphenyl-5,5′-disulfonic acid. J Polym Sci Part A Polym Chem 42:1432–1440
Yamada O, Yin Y, Tanaka K, Kit H, Okamoto KI (2005) Polymer electrolyte fuel cells based on main-chain-type sulfonated polyimides. Electrochim Acta 50:2655–2659
Fang J, Guoa X, Xua H, Okamoto KI (2006) Sulfonated polyimides: synthesis, proton conductivity and water stability. J Power Sources 159:4–11
Shang Y, Xie X, Jin H, Guo J, Wang Y, Feng S et al (2006) Synthesis and characterization of novel sulfonated naphthalenic polyimides as proton conductive membrane for DMFC applications. Eur Polym J 42:2987–2993
Ye X, Bai H, Ho WSW (2006) Synthesis and characterization of new sulfonated polyimides as proton-exchange membranes for fuel cells. J Membr Sci 279:570–577
Yin Y, Yamada O, Tanaka K, Okamoto KI (2006) On the development of naphthalene-based sulfonated polyimide membranes for fuel cell applications. Polym J 38:197–219
Li N, Cui Z, Zhang S, Xing W (2007) Synthesis and characterization of rigid-rod sulfonated polyimides bearing sulfobenzoyl side groups as proton exchange membranes. J Membr Sci 295:148–158
Zhai F, Guo X, Fang J, Xu H (2007) Synthesis and properties of novel sulfonated polyimide membranes for direct methanol fuel cell application. J Membr Sci 296:102–109
Li Y, Jin R, Wang Z, Cui Z, Xing W, Gao L (2007) Synthesis and properties of novel sulfonated polyimides containing binaphthyl groups as proton-exchange membranes for fuel cells. J Polym Sci, Part A: Polym Chem 45:222–231
Rabiee A, Mehdipour-Ataei S, Banihashemi A, Yeganeh H (2008) Preparation of new membranes based on sulfonated aromatic copolyimides. Polym Adv Technol 19:361–370
Mehdipour-Ataei S, Banihashemi A, Yeganeh H, Rabiee A (2008) Preparation and characterization of novel sulfonated copolyimide membranes. e-Polym 11
Li Y, Jin R, Cui Z, Wang Z, Xing W, Qiu X et al (2007) Synthesis and characterization of novel sulfonated polyimides from 1,4-bis (4-aminophenoxy)-naphthyl-2,7-disulfonic acid. Polymer 48:2280–2287
Marestin C, Gebel G, Diat O, Mercier R (2008) Sulfonated polyimides. Adv Polym Sci 216:185–258
Genies C, Mercier R, Sillion B, Petiaud R, Cornet N, Gebel G, Pinéri M (2001) Stability study of sulfonated phthalic and naphthalenic polyimide structures in aqueous medium. Polymer 42:5097–5105
Genies C, Mercier R, Sillion S, Cornet N, Gebel G, Pineri M (2001) Polymer 42:359–373
Zhang Y, Litt M, Savinell RF, Wainright JS (1999) Molecular design considerations in the synthesis of high-conductivity PEMs for fuel cells. Abstr Pap Am Chem Soc 218:U560–U560
Zhang Y, Litt M, Savinell RF, Wainright JS, Vendramini J (2000) Molecular design of polyimides toward high proton conducting materials. Abstr Pap Am Chem Soc 220:U307–U307
Bessonov MI, Koton MM, Kudryavtsev VV, Laius LA (1988) Polyimides–thermally stable polymers. J Polym Sci Part C Polym Lett 26:159–163
Fang J, Guo X, Harada S, Watari T, Tanaka K, Kita H, Okamoto K (2002) Novel sulfonated polyimides as polyelectrolytes for fuel cell application. 1. Synthesis, proton conductivity, and water stability of polyimides from 4,4′-diaminodiphenyl ether-2,2′-disulfonic Acid. Macromolecules 35:9022–9028
Kopitzke RW, Linkous CA, Anderson HR, Nelson GL (2000) Conductivity and water uptake of aromatic-based proton exchange membrane electrolytes. J Electrochem Soc 147:1677–1681
Ye X, Bai H, Ho WSW (2006) Synthesis and characterization of new sulfonated polyimides as proton-exchange membranes for fuel cells. J Membr Sci 279:570–577
Acknowledgments
We thank the University of Jordan for providing chemicals and facilities that made this work possible. We also thank Miss Ruba Zalloum and Miss Fatima Mustafa for their assistance in performing the FTIR, TGA, and 1H-NMR measurements.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Abu-Orabi, F.M., Kailani, M.H., Sweileh, B.A. et al. Sulfonated polyimide copolymers based on 4,4′-diaminostilbene-2,2′-disulfonic acid and 3,5,3′,5′-tetramethylbenzidine with enhanced solubility. Polym. Bull. 74, 895–909 (2017). https://doi.org/10.1007/s00289-016-1752-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-016-1752-x