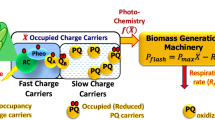
Abstract
Dynamic light regimes strongly impact microalgal photosynthesis efficiency. Finding the optimal way to supply light is then a tricky problem, especially when the growth rate is inhibited by overexposition to light and, at the same time, there is a lack of light in the deepest part of the culture. In this paper, we use the Han model to study the theoretical microalgal growth rate by applying periodically two different light intensities. Two approaches are considered depending on the period of the light pattern. For a large light period, we demonstrate that the average photosynthetic rate can be improved under some conditions. Moreover, we can also enhance the growth rate at steady state as given by the PI-curve. Although, these conditions change through the depth of a bioreactor. This theoretical improvement in the range of 10–15% is due to a recovery of photoinhibited cells during the high irradiance phase. We give a minimal value of the duty cycle for which the optimal irradiance is perceived by the algae culture under flashing light regime.






Similar content being viewed by others
References
Abu-Ghosh S, Fixler D, Dubinsky Z et al (2015) Continuous background light significantly increases flashing-light enhancement of photosynthesis and growth of microalgae. Bioresour Technol 187:144–148
Abu-Ghosh S, Fixler D, Dubinsky Z et al (2015) Flashing light enhancement of photosynthesis and growth occurs when photochemistry and photoprotection are balanced in dunaliella salina. Eur J Phycol 50(4):469–480. https://doi.org/10.1080/09670262.2015.1069404
Abu-Ghosh S, Fixler D, Dubinsky Z et al (2016) Flashing light in microalgae biotechnology. Bioresour Technol 203:357–363. https://doi.org/10.1016/j.biortech.2015.12.057
Baklouti M, Faure V, Pawlowski L et al (2006) Investigation and sensitivity analysis of a mechanistic phytoplankton model implemented in a new modular numerical tool (eco3m) dedicated to biogeochemical modelling. Progress Oceanogr 71(1):34–58. https://doi.org/10.1016/j.pocean.2006.05.003
Balamurugan S, Sathishkumar R, Li HY (2021) Biotechnological perspectives to augment the synthesis of valuable biomolecules from microalgae by employing wastewater. J Water Process Eng 39(101):713
Béchet Q, Shilton A, Guieysse B (2013) Modeling the effects of light and temperature on algae growth: state of the art and critical assessment for productivity prediction during outdoor cultivation. Biotechnol Adv 31(8):1648–1663
Bernard O, Lu LD (2021) Optimal optical conditions for Microalgal production in photobioreactors. https://hal.archives-ouvertes.fr/hal-03323094, submitted paper
Bernard O, Mairet F, Chachuat B (2015) Modelling of microalgae culture systems with applications to control and optimization. Microalgae Biotechnol 59–87
Combe C, Hartmann P, Rabouille S et al (2015) Long-term adaptive response to high-frequency light signals in the unicellular photosynthetic eukaryote Dunaliella salina. Biotechnol Bioeng 112(6):1111–1121
Demory D, Combe C, Hartmann P et al (2018) How do microalgae perceive light in a high-rate pond? Towards more realistic Lagrangian experiments. R Soc Open Sci 5(5):180523
Fernández-Sevilla JM, Brindley C, Jiménez-Ruíz N et al (2018) A simple equation to quantify the effect of frequency of light/dark cycles on the photosynthetic response of microalgae under intermittent light. Algal Res 35:479–487. https://doi.org/10.1016/j.algal.2018.09.026
Grenier J, Lopes F, Bonnefond H et al (2020) Worldwide perspectives of rotating algal biofilm up-scaling, submitted paper
Han BP (2002) A mechanistic model of algal photoinhibition induced by photodamage to photosystem-II. J Theor Biol 214(4):519–527. https://doi.org/10.1006/jtbi.2001.2468
Hartmann P, Béchet Q, Bernard O (2014) The effect of photosynthesis time scales on microalgae productivity. Bioprocess Biosyst Eng 37(1):17–25
Holmes G (2002) The use of hyperbolic cosines in solving cubic polynomials. Math Gaz 86(507):473–477
Khalil H (2002) Nonlinear systems. Prentice Hall, Hoboken
Lamare PO, Aguillon N, Sainte-Marie J et al (2019) Gradient-based optimization of a rotating algal biofilm process. Automatica 105:80–88. https://doi.org/10.1016/j.automatica.2019.02.043
Lu LD (2021) Lagrangian approach for modelling and optimization of coupled hydrodynamics-photosynthesis. Theses, Sorbonne Université. https://tel.archives-ouvertes.fr/tel-03625562
Milledge JJ (2011) Commercial application of microalgae other than as biofuels: a brief review. Rev Environ Sci Biotechnol 10(1):31–41
Morel A (1988) Optical modeling of the upper ocean in relation to its biogenous matter content (case I waters). J Geophys Res Oceans 93(C9):10749–10768
Nikolaou A, Bernardi A, Meneghesso A et al (2015) A model of chlorophyll fluorescence in microalgae integrating photoproduction, photoinhibition and photoregulation. J Biotechnol 194:91–99. https://doi.org/10.1016/j.jbiotec.2014.12.001
Nikolaou A, Hartmann P, Sciandra A et al (2016) Dynamic coupling of photoacclimation and photoinhibition in a model of microalgae growth. J Theor Biol 390:61–72
Pozzobon V, Perre P (2018) Han’s model parameters for microalgae grown under intermittent illumination: determined using particle swarm optimization. J Theor Biol 437:29–35. https://doi.org/10.1016/j.jtbi.2017.10.010
Prasil O, Adir N, Ohad I (1992) Dynamics of photosystem II: mechanism of photoinhibition and recovery process. Top Photosynth 11:295–348
Raven JA (2011) The cost of photoinhibition. Physiol Plant 142(1):87–104
Sanders JA, Verhulst F, Murdock J (2007) Averaging methods in nonlinear dynamical systems. Applied mathematical sciences, 2nd edn. Springer, New York. https://doi.org/10.1007/978-0-387-48918-6
Schulze PS, Brindley C, Fernández JM et al (2020) Flashing light does not improve photosynthetic performance and growth of green microalgae. Bioresour Technol Rep 9(100):367
Stramski D, Sciandra A, Claustre H (2002) Effects of temperature, nitrogen, and light limitation on the optical properties of the marine diatom Thalassiosira pseudonana. Limnol Oceanogr 47(2):392–403
Wu X, Merchuk JC (2001) A model integrating fluid dynamics in photosynthesis and photoinhibition processes. Chem Eng Sci 56(11):3527–3538. https://doi.org/10.1016/S0009-2509(01)00048-3
Zwillinger D (2018) CRC standard mathematical tables and formulas. Chapman and Hall/CRC, Boca Raton
Acknowledgements
This project has received funding from the Digitalgaesation project within the European Union’s Horizon 2020 research and innovation program under the Marie Skłodowska-Curie grant agreement No. 955520.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix A: Analytical computations
1.1 Case I: Large period T
For the large period case, the evolution system (7) can be approximated by (10) which can be solved explicitly for a constant irradiance I as
Moreover, the system is assumed to be periodic (i.e. \(C(y,0)=C(y,T)\)). For the constant light regime \(I_M(y)\), one has \(C(y,t) = \frac{\alpha (I_M(y))}{\alpha (I_M(y))+k_r}\), \(\forall t\in [0,T]\). Using (18), the T-average growth rate is given by \((1-C)\gamma (I_M(y)) = \frac{K\sigma I_M(y)}{1+\tau \sigma I_M(y)+\frac{k_d}{k_r}\tau (\sigma I_M(y))^2} = \mu _S(I_M(y))\). For the high/low light regime, one has
Using the periodic border condition of C, one has
For a given local optical depth y, the T-average growth rate is given by
where \(\gamma _H(y) := \gamma (I_H(y))\) and \(\gamma _L(y) := \gamma (I_L(y))\).
1.2 Case II: Small period T
For small period case, the dynamics of C is negligible (i.e. C is a constant). Integrating (7) from 0 to T gives
Let us denote by \({\bar{I}}:=\frac{1}{T}\int _0^T I \textrm{d} t\) and by \({\overline{IA}}:=\frac{1}{T}\int _0^T I A \textrm{d} t\), then one finds the constant value for C as
For high/low light regime, one has
The periodicity on A gives
On the other hand, the average of IA can be computed by
To simplify the notations, let us denote by \(a:=-(\sigma I_{\max } + \frac{1}{\tau })\eta T\) and by \(i:=-(\sigma I_{\min } + \frac{1}{\tau })(1-\eta )T\), then \(A(0)=\frac{1-C}{\tau \beta _L\beta _H(y)}\frac{1}{1-e^{a+i}}(\beta _L e^i(1-e^a) + \beta _L(1-e^i))\) and the previous computation can be written as (we omitted the dependence on y)
By using the definition of \(I_H,I_H,\beta _L,\beta _L\), one has \((I_H\beta _L-I_H\beta _L)(\beta _L-\beta _L)=\frac{\sigma }{\tau }(I_{\max }-I_{\min })^2\) and \(I_H\beta _L\eta T + I_H\beta _L(1-\eta )T=(\frac{I_{M}}{\tau }+\sigma I_{\max }I_{\min })T\). Replacing C by (A1) in the previous equation gives
where \(\delta (y,T)=\frac{(1-e^a)(1-e^i)}{1-e^{a+i}}\). In other words
We have that
and we can manipulate the value \({\overline{IA}}\) to get
and rearranging this last equality, we get
where
1.3 Proof of Lemma 1
We split equation (7) into the high/low-flashing light configuration
One can solve this system by using the variation of parameters method and get
where \(\mathop {\textrm{Id}}\limits \) denotes the identity matrix in \({\mathbb {R}}^{2\times 2}\). Imposing then periodic conditions, i.e, \((A(y,0),C(y,0)) = (A(y,T),C(y,T))\), one can evaluate the values of (A(y, 0), C(y, 0)) and \((A(y,\eta T),C(y,\eta T))\):
Replacing finally (A4) in (A3) and vice versa, we obtain
The inverse matrix \((Id-e^{-\eta TM_H}e^{-(1-\eta ) TM_L})\) exists because the matrix \(e^{-\eta TM_H} e^{-(1-\eta ) TM_L}\) has no eigenvalue equal to 1. The solution founded it is then unique.
1.4 Exact growth rate
To calculate the T-average of the growth rate, we calculate the integral \(\int _0^TI(y,t)(A(y, t),C(y,t))dt\):
Using (A5) we can compute
In the same way, using (A6) we can get
Replacing the above calculations on (A7), we have
where
Finally, the average growth rate \(\frac{1}{T}\int _0^TK\sigma IAdt\) is proportional to the first coordinate of (A8) multiplied by \(\frac{K\sigma }{T}\). Denote \(\delta \) the first coordinate of \(\Delta \) and note that
where the first coordinate in (A9) and (A10) multiplied by \(K\sigma \) is exactly \(\mu _S(I_H(y))\) and \(\mu _S(I_L(y))\), respectively. So, the T-average of the growth rate is
1.5 Eigenvalues of matrix M
We will condense the analysis of the eigenvalues of \(M_H\) and \(M_L\) in the matrix
Denoting \(\uplambda _1\) and \(\uplambda _2\) the eigenvalues, then we have
From (A13), \(\uplambda _1\) and \(\uplambda _2\) has the same sign, and since (A12) holds, the two eigenvalues are positive.
Appendix B: Proof of Lemma 3
The second derivative of the function \(\mu _S\) can be computed as
and it is zero in the point \(I_c\) which satisfies
This is a depressed cubic equation, where the determinant correspond to
where if \(\Delta \le 0\) all the roots are real. Note that
In this case, the solutions are given by (Zwillinger 2018):
where
From the three possible solutions, only \(l\cos {\frac{\theta }{3}}\) is positive due to \(\theta /3\in (0,\pi /6)\). In the case that \(\Delta > 0\) then the real solution can be written as (Holmes 2002)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fierro Ulloa, J.I., Lu, LD. & Bernard, O. Theoretical growth rate of microalgae under high/low-flashing light. J. Math. Biol. 86, 48 (2023). https://doi.org/10.1007/s00285-023-01871-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00285-023-01871-2