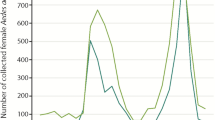
Abstract
Throughout the vector-borne disease modeling literature, there exist two general frameworks for incorporating vector management strategies (e.g. area-wide adulticide spraying and larval source reduction campaigns) into vector population models, namely, the “implicit” and “explicit” control frameworks. The more simplistic “implicit” framework facilitates derivation of mathematically rigorous results on disease suppression and optimal control, but the biological connection of these results to real-world “explicit” control actions that could guide specific management actions is vague at best. Here, we formally define a biological and mathematical relationship between implicit and explicit control, and we provide mathematical expressions relating the strength of implicit control to management-relevant properties of explicit control for four common intervention strategies. These expressions allow the optimal control and basic reproduction number analyses typically utilized in implicit control modeling to be interpreted directly in terms of real-world actions and real-world monetary costs. Our methods reveal that only certain sub-classes of explicit control protocols are able to be represented as implicit controls, and that implicit control is a meaningful approximation of explicit control only when resonance-like synergistic effects between multiple explicit controls have negligible effects on population reduction. When non-negligible synergy exists, implicit control results, despite their mathematical tidiness, fail to provide accurate predictions regarding vector control and disease spread. Collectively, these elements build an effective bridge between analytically interesting and mathematically tractable implicit control and the challenging, action-oriented explicit control.








Similar content being viewed by others
References
Agusto F, Lenhart S (2013) Optimal control of the spread of malaria superinfectivity. J Biol Syst 21:1340002. https://doi.org/10.1142/S0218339013400020
Agusto FB (2017) Optimal control and cost-effectiveness analysis of a three age-structured transmission dynamics of chikungunya virus. Dis Continuous Dyn Syst Ser B 22:293–320. https://doi.org/10.3934/dcdsb.2017034
Agusto FB, Elmojtaba IM (2017) Optimal control and cost-effective analysis of malaria/visceral leishmaniasis co-infection. PLoS ONE 12:e0171102. https://doi.org/10.1371/journal.pone.0171102
Agusto FB, Marcus N, Oksun KO (2012) Application of optimal control to the epidemology of malaria. Electr J Differ Equ 2012:1–22
Agusto FB, Bewick S, Fagan WF (2017) Mathematical model of Zika virus with veritcal transmission. Infect Dis Model 2:244–267. https://doi.org/10.1016/j.idm.2017.05.003
Bacaer N (2007) Approximation of the basic reproduction number \(R_0\) for vector-borne diseases with a periodic vector population. Bull Math Biol 69:1067–1091. https://doi.org/10.1007/s11538-006-9166-9
Bacaer N, Guernaoui S (2006) The epdemic threshold of vectir-borne diseases with seasonality: The case of cutaneous leishmaniasis in Chichaoua, Morocco. J Math Biol 56:421–436. https://doi.org/10.1007/s00285-006-0015-0
Bainov D, Simeonov P (1993) Impulsive differntial equations: Periodic solutions and applications, Pitman monographs and surveys in pure and applied mathematics, vol 66. Longman Scientific and Technical, Harlow Essex, England
Barmark DH, Dorso CO, Otero M, Solari HG (2014) Modeling interventions during an dengue outbreak. Epidemio Infect 142:545–561. https://doi.org/10.1017/S0950268813001301
Bonds JAS (2012) Ultra-low-volume space sprays in mosuito control: a critical review. Med Vet Entoml 26:121–130. https://doi.org/10.1111/j.1365-2915.2011.00992.x
Bowman C, Gumel AB, van den Driessche P, Wu J, Zhu H (2005) A mathematical model for assessing control strategies against West Nile virus. Bull Math Biol 67:1107–1133. https://doi.org/10.1016/j.bulm.2005.01.002
Boyce R, Lenhart A, Kroeger A, Velayudhan R, Roberts B, Horstick O (2013) Bacillus thuringiensis israelensis (Bti) for the control of dengue vectors: systematic literature review. Trop Med Int Health 18:564–577. https://doi.org/10.1111/tmi.12087
Burattini MN, Chen M, Chow A, Coutinho FAB, Goh KT, Lopez LF, Ma S, Massad E (2008) Modelling the control strategies against dengue in Singapore. Epidemio Infect 136:309–319. https://doi.org/10.1017/S0950268807008667
Bustamam A, Aldila D, Yuwanda A (2018) Understanding dengue control for short- and long-term intervention with a mathematical model approach. J Appl Math. https://doi.org/10.1155/2018/9674138
Chavex JP, Gotz T, Siegmund S, Wijaya KP (2017) An SIR-Dengue transmission model with seasonal effects and impulsive control. Math Biosci 289:29–39. https://doi.org/10.1016/j.mbs.2017.04.005
Church K, Smith R (2018) Continuous approximation of linear impulsive systems and a new form of robust stability. J Math Anal Appl 457:616–644. https://doi.org/10.1016/j.jmaa.2017.08.026
Cilek JE (2008) Application of insecticides to vegetation as barriers against host-seeking mosquitoes. J Am Mosq Control Assoc 24:172–176. https://doi.org/10.2987/8756-971X(2008)24[172:AOITVA]2.0.CO;2
Demers J, Bewick S, Caillouët KA, Fagan WF, Robertson SL (2020) Managing disease outbreaks: the importance of vector mobility and spatially heterogeneous control. PLoS Comput Biol 16:e1008136. https://doi.org/10.1371/journal.pcbi.1008136
Diekmann O, Heesterbeek JAP, Metz JAJ (1990) On the definition and the computation of the basic reproduction ratio \(R_0\) in heterogeneous populations. J Math Biol 28:365–382. https://doi.org/10.1007/BF00178324
Doyle MA, Kline DL, amd P E Kaufman SAA, (2009) Efficacy of residual bifenthrin applied to landscape vegetation against Aedes albopictus. J Am Mosq Control Assoc 25:179–183. https://doi.org/10.2987/08-5804.1
van den Driessche P (2017) Reproduction numbers of infectious disease models. Infect Dis Model 2:288–303. https://doi.org/10.1016/j.idm.2017.06.002
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48. https://doi.org/10.1016/S0025-5564(02)00108-6
Esteva L, Vargas C (1998) Analysis of a dengue disease transmission model. Dis Continuous Dyn Syst Ser B 150:131–151. https://doi.org/10.1016/S0025-5564(98)10003-2
Faraji A, Unlu I (2016) The eye of the tiger, the thrill of the fight: Effective larval and adult control measures against the Asian tiger mosquito, Aedes albopictus (Diptera: Culicidae), in North America. J Med Entoml 53. https://doi.org/10.1093/jme/tjw096
Fatmawati Tasman H (2015) An optimal control strategy to reduce the spread of malaria resistance. Math Biosci 262:73–79. https://doi.org/10.1016/j.mbs.2014.12.005
Gourley SA, Liu R, Wu J (2007) Some vector borne diseases with structured host populations: Extinction and spatial spread. SIAM J Appl Math 2:408–433. https://doi.org/10.1137/050648717
Griffin JT (2015) The interaction between seasonality and pulsed interventions against malaria in their effects on the reproduction number. PLoS Comput Biol 11:e1004057. https://doi.org/10.1371/journal.pcbi.1004057
Gwalani H, Hawamdeh F, Mikler AR, Xiong K (2018) Modeling the 2013 Zika outbreak in French Polynesia: intervention strategies. Appl Syst Innov 1:31. https://doi.org/10.3390/asi1030031
Haddad W, Chellaboina V, Nersesov S (2006) Impulsive and Hybrid Dynamical Systems: Stability, Dissipativity, and Control. Princeton Series in Applied Mathematics, Princeton University Press, Princeton, NJ
Knerer G, Currie CSM, Brailsford SC (2015) Impact of combined vector-control and vaccination strategies on transmission dynamics of dengue fever: a model-based analysis. Health Care Manag Sci 18:205–217. https://doi.org/10.1007/s10729-013-9263-x
Lenhart S, Workman JT (2007) Optimal control applied to biological methods. Mathematical and computational biology series, Chapman and Hall/CRC, Boca Raton, Fl
Lou Y, Zhao X (2011) A reaction diffusion malaria model with incubation period in the vector population. J Math Biol 62:543–568. https://doi.org/10.1007/s00285-010-0346-8
Oki M, Sunahara T, Hashizume M, Yamamoto T (2011) Optimal timing of insecticide fogging to minimize dengue cases: Modeling dengue transmission among various seasonalities and transmission intensities. PLoS One 5:e1367. https://doi.org/10.1371/journal.pntd.0001367
Okosun KO, Ouifki R, Marcus N (2011) Optimal control analysis of a malaria disease transmission model that includes treatment and vaccination with waning immunity. BioSystems pp 136–145, https://doi.org/10.1016/j.biosystems.2011.07.006
Pratt HD, Moore CG (1993) Mosquitoes of public health importance and their control. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, revised edn
Rodrigues HS, Monteiro MTT, Torres DFM (2013) Bioeconomic perspectives to an optimal control dengue model. Int J Comput Math 90:1–22
Schaefer E, Caillouet K, Robertson S (2018) Methods for prophylactic management of West Nile virus using a stage-structured avian host-vector model with vaccination, larvicide, and adulticide. Nat Resour Model 31:e12165. https://doi.org/10.1111/nrm.12165
Sepulveda LS, Vasileva O (2016) Optimal control approach to dengue reduction and prevention in Cali, Colombia. Math Meth Appl Sci 39:5475–5496. https://doi.org/10.1002/mma.3932
Wang W, Zhao X (2008) Threshold dynamics for compartmental epidemic models in periodic environments. J Dyn Diff Equat 20:699–717. https://doi.org/10.1007/s10884-008-9111-8
Yakob L, Clements ACA (2013) A mathematical model of chikungunya dynamics and control: the major epidemic on Reunion Island. PLos ONE 8:e57448. https://doi.org/10.1371/journal.pone.0057448
Zhu D, Ren J, Zhu H (2018) Spatial-temporal basic reproduction number and dynamics for a dengue disease diffusion model. Math Method Appl Sci 41:5388–5403. https://doi.org/10.1002/mma.5085
Acknowledgements
The authors would like to acknowledge F. Agusto and K. Caillouet for useful discussions. This work was supported by DOD SERDP contract W912HQ-16-C-0054 to S. Bewick and W.F. Fagan and a grant from the Simons Foundation (426126, SR).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Demers, J., Robertson, S.L., Bewick, S. et al. Implicit versus explicit vector management strategies in models for vector-borne disease epidemiology. J. Math. Biol. 84, 48 (2022). https://doi.org/10.1007/s00285-022-01742-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00285-022-01742-2