Abstract
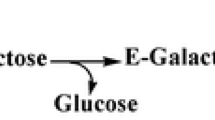
β-Galactosidase is a glycoside hydrolase enzyme that possesses both hydrolytic and transgalactosylation properties and has several benefits and advantages in the food and dairy industries. The catalytic process of β-galactosidase involves the transfer of a sugar residue from a glycosyl donor to an acceptor via a double-displacement mechanism. Hydrolysis prevails when water acts as an acceptor, resulting in the production of lactose-free products. Transgalactosylation prevails when lactose acts as an acceptor, resulting in the production of prebiotic oligosaccharides. β-Galactosidase is also obtained from many sources including bacteria, yeast, fungi, plants, and animals. However, depending on the origin of the β-galactosidase, the monomer composition and their bonds may differ, thereby influencing their properties and prebiotic efficacy. Thus, the increasing demand for prebiotics in the food industry and the search for new oligosaccharides have compelled researchers to search for novel sources of β-galactosidase with diverse properties. In this review, we discuss the properties, catalytic mechanisms, various sources and lactose hydrolysis properties of β-galactosidase.



Similar content being viewed by others
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
References
Panesar PS, Panesar R, Singh RS et al (2006) Microbial production, immobilization and applications of β-D-galactosidase. J Chem Technol Biotechnol 81:530–543. https://doi.org/10.1002/jctb.1453
Chanalia P, Gandhi D, Attri P, Dhanda S (2018) Purification and characterization of β-galactosidase from probiotic Pediococcus acidilactici and its use in milk lactose hydrolysis and galactooligosaccharide synthesis. Bioorg Chem 77:176–189. https://doi.org/10.1016/j.bioorg.2018.01.006
Aburto C, Castillo C, Cornejo F et al (2019) β-Galactosidase from Exiguobacterium acetylicum: cloning, expression, purification and characterization. Bioresour Technol 277:211–215. https://doi.org/10.1016/j.biortech.2019.01.005
Kareb O, Aïder M (2019) Whey and its derivatives for probiotics, prebiotics, synbiotics, and functional foods: a critical review. Probiotics Antimicrob Proteins 11:348–369. https://doi.org/10.1007/s12602-018-9427-6
Ugidos-Rodríguez S, Matallana-González MC, Sánchez-Mata MC (2018) Lactose malabsorption and intolerance: a review. Food Funct 9(8):4056–4068
Husain Q (2010) β Galactosidases and their potential applications: a review. Crit Rev Biotechnol 30:41–62. https://doi.org/10.3109/07388550903330497
Kalathinathan P, Kodiveri Muthukaliannan G (2020) A statistical approach for enhanced production of β-galactosidase from Paracoccus sp. and synthesis of galacto-oligosaccharides. Folia Microbiol 65:811–822. https://doi.org/10.1007/s12223-020-00791-8
Saqib S, Akram A, Halim SA, Tassaduq R (2017) Sources of β-galactosidase and its applications in food industry. 3 Biotech 7:1–7. https://doi.org/10.1007/s13205-017-0645-5
Kalathinathan P, Pulicherla K, Sain A et al (2021) New alkali tolerant β-galactosidase from Paracoccus marcusii KGP—a promising biocatalyst for the synthesis of oligosaccharides derived from lactulose (OsLu), the new generation prebiotics. Bioorg Chem 115:105207. https://doi.org/10.1016/j.bioorg.2021.105207
Xavier JR, Ramana KV, Sharma RK (2018) β-galactosidase: biotechnological applications in food processing. J Food Biochem 42:e12564. https://doi.org/10.1111/jfbc.12564
Lu L, Guo L, Wang K et al (2020) β-Galactosidases: a great tool for synthesizing galactose-containing carbohydrates. Biotechnol Adv 39:107465. https://doi.org/10.1016/j.biotechadv.2019.107465
Vera C, Guerrero C, Aburto C et al (2020) Conventional and non-conventional applications of β-galactosidases. BBA-Proteins Proteomics 1868:140271. https://doi.org/10.1016/j.bbapap.2019.140271
Panesar PS, Kaur R, Singh RS, Kennedy JF (2018) Biocatalytic strategies in the production of galacto-oligosaccharides and its global status. Int J Biol Macromol 111:667–679. https://doi.org/10.1016/j.ijbiomac.2018.01.062
Panesar P, Marwaha S, Kumar H (2010) Enzymes in food processing: fundamentals and potential applications. In: Enzymes in food processing: fundamentals and potential applications
Mlichová Z, Rosenberg M (2006) Current trends of β-galactosidase application in food technology. J Food Nutr Res 45:47–54
Xu X, Fan X, Fan C et al (2019) Production optimization of an active β-galactosidase of Bifidobacterium animalis in heterologous expression systems. Biomed Res Int 2019:1–10
Kittibunchakul S, Pham M, Tran A, Nguyen T (2019) β -Galactosidase from Lactobacillus helveticus DSM 20075: biochemical characterization and recombinant expression for applications in dairy industry. Int J Mol Sci 20:947. https://doi.org/10.3390/ijms20040947
Watzlawick H, Altenbuchner J (2019) Multiple integration of the gene ganA into the Bacillus subtilis chromosome for enhanced β-galactosidase production using the CRISPR/Cas9 system. AMB Express 9(1):1–11. https://doi.org/10.1186/s13568-019-0884-4
Li D, Li S, Wu Y et al (2020) Cloning and characterization of a new β-galactosidase from Alteromonas sp. QD01 and its potential in synthesis of galacto-oligosaccharides. Mar Drugs 18(6):312. https://doi.org/10.3390/md18060312
Alikunju AP, Joy S, Salam JA et al (2018) Functional characterization of a new cold-adapted β-galactosidase from an arctic fjord sediment bacteria Enterobacter ludwigii MCC 3423. Catal Lett 148:3223–3235. https://doi.org/10.1007/s10562-018-2504-3
Núñez-Montero K, Salazar R, Santos A et al (2021) Antarctic rahnella inusitata: a producer of cold-stable β-galactosidase enzymes. Int J Mol Sci 22(8):4144. https://doi.org/10.3390/ijms22084144
Bekler F, Yalaz S, Acer O, Guven K (2017) Purification of thermostable b-galactosidase from Anoxybacillus sp. KP1 and estimation of combined effect of some chemicals on enzyme activity using semiparametric errors in variables model. Fresenius Environ Bull 26:2253–2261
Murphy J, Ryan MP, Walsh G (2020) Purification and characterization of a novel β-galactosidase from the thermoacidophile Alicyclobacillus vulcanalis. Appl Biochem Biotechnol 191(3):1190–1206. https://doi.org/10.1007/s12010-020-03233-w
Zhou Z, He N, Han Q et al (2021) Characterization and application of a new β-galactosidase Gal42 from marine bacterium Bacillus sp. BY02. Front Microbiol 12:3015. https://doi.org/10.3389/fmicb.2021.742300
Wang X, Chen S, Fan L et al (2022) Characterization of a halotolerant GH2 family β-galactosidase GalM from Microvirga sp. strain MC18. Protein Expr Purif 194:106074. https://doi.org/10.1016/j.pep.2022.106074
Xu Y, Wu Q, Bai L et al (2021) Cloning, expression, and bioinformatics analysis and characterization of a β-galactosidase from Bacillus coagulans T242. J Dairy Sci 104(3):2735–2747. https://doi.org/10.3168/jds.2020-18942
Zolnere K, Ciprovica I (2017) The comparison of commercially available β-galactosidases for dairy industry: review. In: Research for Rural Development. pp 215–222
Kamran A, Bibi Z, Aman A, Qader SAU (2017) Hyper production of Β-galactosidase from newly isolated strain of Aspergillus nidulans. J Food Process Eng 40(3):e12452. https://doi.org/10.1111/jfpe.12452
Martarello RD, Cunha L, Cardoso SL et al (2019) Optimization and partial purification of beta-galactosidase production by Aspergillus niger isolated from Brazilian soils using soybean residue. AMB Express 9:1–13. https://doi.org/10.1186/s13568-019-0805-6
Vidya CH, Gnanesh Kumar BS, Chinmayee CV, Singh SA (2020) Purification, characterization and specificity of a new GH family 35 galactosidase from Aspergillus awamori. Int J Biol Macromol 156:885–895. https://doi.org/10.1016/j.ijbiomac.2020.04.013
Yamada M, Chiba S, Endo Y, Isobe K (2017) New alkalophilic β-galactosidase with high activity in alkaline pH region from Teratosphaeria acidotherma AIU BGA-1. J Biosci Bioeng 123:15–19. https://doi.org/10.1016/j.jbiosc.2016.07.003
Zerva A, Limnaios A, Kritikou AS et al (2021) A novel thermophile β-galactosidase from Thermothielavioides terrestris producing galactooligosaccharides from acid whey. New Biotechnol 63:45–53. https://doi.org/10.1016/j.nbt.2021.03.002
Nag D, Kumar V, Kumar V, Kumar S (2021) Short communications a new extracellular b-galactosidase producing Kluyveromyces sp. PCH397 from yak milk and its applications for lactose hydrolysis and prebiotics synthesis. Indian J Microbiol 61:391–395. https://doi.org/10.1007/s12088-021-00955-1
Kokkiligadda A, Beniwal A, Saini P, Vij S (2016) Utilization of cheese whey using synergistic immobilization of β-galactosidase and Saccharomyces cerevisiae cells in dual matrices. Appl Biochem Biotechnol 179:1469–1484. https://doi.org/10.1007/s12010-016-2078-8
Bosso A, Iglecias Setti AC, Tomal AB et al (2019) Substrate consumption and beta-galactosidase production by Saccharomyces fragilis IZ 275 grown in cheese whey as a function of cell growth rate. Biocatal Agric Biotechnol 21:101335. https://doi.org/10.1016/j.bcab.2019.101335
Murphy J, Walsh G (2019) Purification and characterization of a novel thermophilic β-galactosidase from Picrophilus torridus of potential industrial application. Extremophiles 23:783–792. https://doi.org/10.1007/s00792-019-01133-4
Letsididi R, Hassanin HAM, Koko MYF et al (2018) Lactulose production by a thermostable glycoside hydrolase from the hyperthermophilic archaeon Caldivirga maquilingensis IC-167. J Sci Food Agric 98:928–937. https://doi.org/10.1002/jsfa.8539
Bentahar J, Doyen A, Beaulieu L, Deschênes JS (2019) Acid whey permeate: An alternative growth medium for microalgae Tetradesmus obliquus and production of β-galactosidase. Algal Res 41:101559. https://doi.org/10.1016/j.algal.2019.101559
Yadav A, Kayastha AM (2020) Lens culinaris β-galactosidase (Lsbgal): insights into its purification, biochemical characterization and trisaccharides synthesis. Bioorg Chem 95:103543. https://doi.org/10.1016/j.bioorg.2019.103543
Ghannad Sabzevary A, Hosseini R (2016) Two main domains with different roles discovered an a new tomato beta-galactosidase. Russ J Bioorg Chem 42:522–531. https://doi.org/10.1134/S106816201605006X
Erich S, Kuschel B, Schwarz T et al (2015) Novel high-performance metagenome β-galactosidases for lactose hydrolysis in the dairy industry. J Biotechnol 210:27–37. https://doi.org/10.1016/j.jbiotec.2015.06.411
Liu P, Wang W, Zhao J, Wei D (2019) Screening novel β-galactosidases from a sequence-based metagenome and characterization of an alkaline β-galactosidase for the enzymatic synthesis of galactooligosaccharides. Protein Expr Purif 155:104–111. https://doi.org/10.1016/j.pep.2018.12.001
Liu P, Wu J, Liu J, Ouyang J (2021) Engineering of a β-galactosidase from Bacillus coagulans to relieve product inhibition and improve hydrolysis performance. J Dairy Sci 104(10):10566–10575. https://doi.org/10.3168/jds.2021-20388
Nguyen TT, Van VuH, Nguyen NTH et al (2016) Effect of mutations to amino acid A301 and F361 in thermostability and catalytic activity of the β-galactosidase from Bacillus subtilis VTCC-DVN-12–01. BMC Biochem 17(1):1–11. https://doi.org/10.1186/s12858-016-0070-0
Katrolia P, Liu X, Li G, Kopparapu NK (2019) Enhanced properties and lactose hydrolysis efficiencies of food-grade β-galactosidases immobilized on various supports: a comparative approach. Appl Biochem Biotechnol 188:410–423. https://doi.org/10.1007/s12010-018-2927-8
Serey M, Vera C, Guerrero C, Illanes A (2021) Immobilization of Aspergillus oryzae β-galactosidase in cation functionalized agarose matrix and its application in the synthesis of lactulose. Int J Biol Macromol 167:1564–1574. https://doi.org/10.1016/j.ijbiomac.2020.11.110
Nguyen VD, Styevkó G, Ta LP et al (2018) Immobilization and some properties of commercial enzyme preparation for production of lactulose-based oligosaccharides. Food Bioprod Process 107:97–103. https://doi.org/10.1016/j.fbp.2017.11.004
Klein MP, Hackenhaar CR, Lorenzoni ASG et al (2016) Chitosan crosslinked with genipin as support matrix for application in food process: support characterization and β-D-galactosidase immobilization. Carbohydr Polym 137:184–190. https://doi.org/10.1016/j.carbpol.2015.10.069
Selvarajan E, Mohanasrinivasan V, Subathra Devi C, George Priya Doss C (2015) Immobilization of β-galactosidase from lactobacillus plantarum HF571129 on ZnO nanoparticles: characterization and lactose hydrolysis. Bioprocess Biosyst Eng 38:1655–1669. https://doi.org/10.1007/s00449-015-1407-6
Lima PC, Gazoni I, de Carvalho AMG et al (2021) β-galactosidase from Kluyveromyces lactis in genipin-activated chitosan: an investigation on immobilization, stability, and application in diluted UHT milk. Food Chem 349:129050. https://doi.org/10.1016/j.foodchem.2021.129050
Tizchang S, Khiabani MS, Mokarram RR et al (2021) Immobilization of β-galactosidase by halloysite-adsorption and entrapment in a cellulose nanocrystals matrix. Biochim Biophys Acta 1865(6):129896. https://doi.org/10.1016/j.bbagen.2021.129896
Kuribayashi LM, do RioRibeiro VP, de Santana RC et al (2021) Immobilization of β-galactosidase from Bacillus licheniformis for application in the dairy industry. Appl Microbiol Biotechnol 105:3601–3610. https://doi.org/10.1007/s00253-021-11325-8
Sun J, Yao C, Wang W et al (2018) Cloning, expression and characterization of a novel cold-adapted β-galactosidase from the deep-sea bacterium Alteromonas sp. ML52. Mar Drugs 16(12):469. https://doi.org/10.3390/md16120469
Yao C, Sun J, Wang W et al (2019) A novel cold-adapted β-galactosidase from Alteromonas sp. ML117 cleaves milk lactose effectively at low temperature. Process Biochem 82:94–101. https://doi.org/10.1016/j.procbio.2019.04.016
Li N, Liu Y, Wang C et al (2021) Overexpression and characterization of a novel GH4 galactosidase with β-galactosidase activity from Bacillus velezensis SW5. J Dairy Sci 104(9):9465–9477. https://doi.org/10.3168/jds.2021-20258
Luan S, Duan X (2022) A novel thermal-activated β-galactosidase from Bacillus aryabhattai GEL-09 for lactose hydrolysis in milk. Foods 11(3):372. https://doi.org/10.3390/foods11030372
Liu P, Xie J, Liu J, Ouyang J (2019) A novel thermostable β-galactosidase from Bacillus coagulans with excellent hydrolysis ability for lactose in whey. J Dairy Sci 102:9740–9748. https://doi.org/10.3168/jds.2019-16654
Kalathinathan P, Kodiveri Muthukaliannan G (2021) Characterisation of a potential probiotic strain Paracoccus marcusii KGP and its application in whey bioremediation. Folia Microbiol. https://doi.org/10.1007/s12223-021-00886-w
Vasileva N, Ivanov Y, Damyanova S, Kostova I (2016) Hydrolysis of whey lactose by immobilized β-galactosidase in a bioreactor with a spirally wound membrane. Int J Biol Macromol 82:339–346. https://doi.org/10.1016/j.ijbiomac.2015.11.025
Panesar PS, Kumari S, Panesar R (2010) Potential applications of immobilized β-galactosidase in food processing industries. Enzym Res. https://doi.org/10.4061/2010/473137
Elsayed EA, Danial EN, Wadaan MA, El-Enshasy HA (2019) Production of β-galactosidase in shake-flask and stirred tank bioreactor cultivations by a newly isolated Bacillus licheniformis strain. Biocatal Agric Biotechnol 20:101231. https://doi.org/10.1016/j.bcab.2019.101231
Wang L, Mou Y, Guan B et al (2020) Genome sequence of the psychrophilic Cryobacterium sp. LW097 and characterization of its four novel cold-adapted β-galactosidases. Int J Biol Macromol 163:2068–2083. https://doi.org/10.1016/j.ijbiomac.2020.09.100
Alikkunju AP, Sainjan N, Silvester R et al (2016) Screening and characterization of cold-active β-galactosidase producing psychrotrophic Enterobacter ludwigii from the sediments of arctic fjord. Appl Biochem Biotechnol 180:477–490. https://doi.org/10.1007/s12010-016-2111-y
Xia Y, He L, Mao J et al (2018) Purification, characterization, and gene cloning of a new cold-adapted β-galactosidase from Erwinia sp. E602 isolated in northeast China. J Dairy Sci 101:6946–6954. https://doi.org/10.3168/jds.2018-14605
Huang J, Zhu S, Zhao L et al (2020) A novel β-galactosidase from Klebsiella oxytoca ZJUH1705 for efficient production of galacto-oligosaccharides from lactose. Appl Microbiol Biotechnol 104:6161–6172. https://doi.org/10.1007/s00253-020-10679-9
Ding H, Zeng Q, Zhou L et al (2017) Biochemical and structural insights into a novel thermostable β-1,3-galactosidase from Marinomonas sp. BSi20414. Mar Drugs 15(1):13. https://doi.org/10.3390/md15010013
Mahdian SMA, Karimi E, Tanipour MH et al (2016) Expression of a functional cold active β-galactosidase from Planococcus sp.-L4 in Pichia pastoris. Protein Expr Purif 125:19–25. https://doi.org/10.1016/j.pep.2015.09.008
Cardoso BB, Silvério SC, Abrunhosa L et al (2017) β-Galactosidase from Aspergillus lacticoffeatus: a promising biocatalyst for the synthesis of novel prebiotics. Int J Food Microbiol 257:67–74. https://doi.org/10.1016/j.ijfoodmicro.2017.06.013
Vidya B, Palaniswamy M, Angayarkanni J et al (2020) Purification and characterization of β-galactosidase from newly isolated Aspergillus terreus (KUBCF1306) and evaluating its efficacy on breast cancer cell line (MCF-7). Bioorg Chem 94:103442. https://doi.org/10.1016/j.bioorg.2019.103442
Paulo AJ, Wanderley MCDA, de Oliveira RJV et al (2021) Production and partial purification by PEG/citrate ATPS of a β-galactosidase from the new promising isolate Cladosporium tenuissimum URM 7803. Prep Biochem Biotechnol 51(3):289–299. https://doi.org/10.1080/10826068.2020.1815054
Bentahar J, Doyen A, Beaulieu L, Deschênes JS (2019) Investigation of β-galactosidase production by microalga Tetradesmus obliquus in determined growth conditions. J Appl Phycol 31:301–308. https://doi.org/10.1007/s10811-018-1550-y
Haghju S, Bari MR, Khaled-Abad MA (2018) Affecting parameters on fabrication of β-D-galactosidase immobilized chitosan/poly (vinyl alcohol) electrospun nanofibers. Carbohydr Polym 200:137–143. https://doi.org/10.1016/j.carbpol.2018.07.096
Nguyen VD, Styevkó G, Madaras E et al (2019) Immobilization of β-galactosidase on chitosan-coated magnetic nanoparticles and its application for synthesis of lactulose-based galactooligosaccharides. Process Biochem 84:30–38. https://doi.org/10.1016/j.procbio.2019.05.021
Guerrero C, Aburto C, Suárez S et al (2018) Effect of the type of immobilization of β-galactosidase on the yield and selectivity of synthesis of transgalactosylated oligosaccharides. Biocatal Agric Biotechnol 16:353–363. https://doi.org/10.1016/j.bcab.2018.08.021
Julio-Gonzalez LC, Hernández-Hernández O, Javier Moreno F et al (2019) High-yield purification of commercial lactulose syrup. Sep Purif Technol 224:475–480. https://doi.org/10.1016/j.seppur.2019.05.053
Botvynko A, Bednářová A, Henke S et al (2019) Production of galactooligosaccharides using various combinations of the commercial β-galactosidases. Biochem Biophys Res Commun 517:762–766. https://doi.org/10.1016/j.bbrc.2019.08.001
Xin Y, Guo T, Zhang Y et al (2019) A new β-galactosidase extracted from the infant feces with high hydrolytic and transgalactosylation activity. Appl Microbiol Biotechnol 103:8439–8448. https://doi.org/10.1007/s00253-019-10092-x
Ding H, Zhou L, Zeng Q et al (2018) Heterologous expression of a thermostable β-1,3-galactosidase and its potential in synthesis of galactooligosaccharides. Mar Drugs 16(11):415. https://doi.org/10.3390/md16110415
Thøgersen MS, Christensen SJ, Jepsen M et al (2020) Transglycosylating β-D-galactosidase and α-L-fucosidase from Paenibacillus sp. 3179 from a hot spring in East Greenland. Microbiologyopen 9(3):e980. https://doi.org/10.1002/mbo3.980
Acknowledgements
The authors would like to acknowledge the Vellore Institute of Technology for providing space and infrastructure for carrying out the project titled ‘Production, purification and characterisation of β-galactosidase from Paracoccus marcusii for the synthesis of prebiotic carbohydrates’.
Funding
This study did not receive any specific grants from funding agencies in the public, commercial, or not-for-profit sector.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the data analysis and manuscript preparation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical Approval
No ethical permission was required for this study which did not involve experiments involving animal or human samples.
Consent for Publication
Not Applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kalathinathan, P., Sain, A., Pulicherla, K. et al. A Review on the Various Sources of β-Galactosidase and Its Lactose Hydrolysis Property. Curr Microbiol 80, 122 (2023). https://doi.org/10.1007/s00284-023-03220-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-023-03220-4