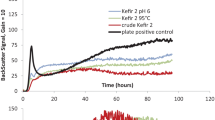
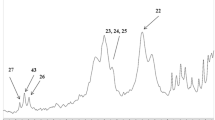
Abstract
The addition of anthocyanin to kefir for the production of more functional and bio-diversified kefir beverages has the potential to increase kefir’s healthful activities. In the present study, anthocyanin extracts, obtained from black carrots, were added into kefir mixture during the fermentation process in different concentrations (1% and 5%, w/v). These kefir samples were then analyzed in terms of their microbiological qualities by metagenomic analysis. The results of the analyses show that the addition of anthocyanin has significant impacts on the community structure of kefir microbiome which in turn directly affects the expected health impacts of the beverage. Kefir with no anthocyanin included predominantly probiotic bacteria such as Lactococcus lactis (34%) and Lactobacillus kefiri (34%). On the other hand, kefir with 1% anthocyanin demonstrated a more balanced distribution of probiotic species like Lb. kefiri (17%), Leuconostoc mesenteroides (9%), and Lc. lactis (5%) at similar abundance rates. 5% anthocyanin kefir demonstrated the highest polarity in the community with a strong dominance of probiotic Lb. kefiri (72%), and distinctly less abundant bacteria such as Streptococcus salivarius subsp. thermophilus (3%). These findings provide that fortification with anthocyanins can be utilized to enhance the quality, composition, and beneficial functions of kefir.





Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Ganatsios V, Nigam P, Plessas S, Terpou A (2021) Kefir as a functional beverage gaining momentum towards its health promoting attributes. Beverages 7(3):48. https://doi.org/10.3390/beverages7030048
Ilıkkan ÖK, Bağdat EŞ (2021) Comparison of bacterial and fungal biodiversity of Turkish kefir grains with high-throughput metagenomic analysi. LWT 152:112375. https://doi.org/10.1016/j.lwt.2021.112375
Sulmiyati S, Said NS, Fahrodi DU, Malaka R, Maruddin F (2019) The physicochemical, microbiology, and sensory characteristics of kefir goat milk with different levels of kefir grain. Trop Anim Sci J 42(2):152–158. https://doi.org/10.5398/tasj.2019.42.2.152
Dertli E, Çon AH (2017) Microbial diversity of traditional kefir grains and their role on kefir aroma. LWT Food Sci Technol 85:151–157. https://doi.org/10.1016/j.lwt.2017.07.017
Blasche S, Kim Y, Mars RAT, Machado D, Maansson M, Kafkia E et al (2021) Metabolic cooperation and spatiotemporal niche partitioning in a kefir microbial community. Nat Microbiol 6(2):196–208. https://doi.org/10.1038/s41564-020-00816-5
HadiNezhad M, Duc C, Han NF, Hosseinian F (2013) Flaxseed soluble dietary fibre enhances lactic acid bacterial survival and growth in kefir and possesses high antioxidant capacity. J Food Res 2(5):152. https://doi.org/10.5539/jfr.v2n5p152
Sabokbar N, Khodaiyan F (2016) Total phenolic content and antioxidant activities of pomegranate juice and whey based novel beverage fermented by kefir grains. J Food Sci Technol 53(1):739–747. https://doi.org/10.1007/s13197-015-2029-3
Azizi NF, Kumar MR, Yeap SK, Abdullah JO, Khalid M, Omar AR et al (2021) Kefir and its biological activities. Foods 10(6):1210. https://doi.org/10.3390/foods10061210
Hamida RS, Shami A, Ali MA, Almohawes ZN, Mohammed AE, Bin-Meferij MM (2021) Kefir: a protective dietary supplementation against viral infection. Biomed Pharmacother 133:110974. https://doi.org/10.1016/j.biopha.2020.110974
Liu JR, Chen MJ, Lin CW (2005) Antimutagenic and antioxidant properties of milk-kefir and soymilk-kefir. J Agric Food Chem 53(7):2467–2474. https://doi.org/10.1021/jf048934k
Fatahi A, Soleimani N, Anticancer AP (2021) Activity of kefir on glioblastoma cancer cell as a new treatment. Int J Food Sci 2021:8180742. https://doi.org/10.1155/2021/8180742
Murray E, Sharma R, Smith KB, Mar KD, Barve R, Lukasik M et al (2019) Probiotic consumption during puberty mitigates LPS-induced immune responses and protects against stress-induced depression- and anxiety-like behaviors in adulthood in a sex-specific manner. Brain Behav Immun 81:198–212. https://doi.org/10.1016/j.bbi.2019.06.016
de Melo EL, Pinto AM, Baima CLB, da Silva HR, da Silva SI, Sanchez-Ortiz BL et al (2020) Evaluation of the in vitro release of isoflavones from soybean germ associated with kefir culture in the gastrointestinal tract and anxiolytic and antidepressant actions in zebrafish (Danio rerio). J Funct Foods 70:103986. https://doi.org/10.1016/j.jff.2020.103986
Lila MA (2004) Anthocyanins and human health: an in vitro investigative approach. J Biomed Biotechnol 2004(5):306–313. https://doi.org/10.1155/S111072430440401X
Kabakci SA, Turkyilmaz M, Ozkan M (2020) Changes in the quality of kefir fortified with anthocyanin-rich juices during storage. Food Chem 326:126977. https://doi.org/10.1016/j.foodchem.2020.126977
Aiello F, Restuccia D, Spizzirri UG, Carullo G, Leporini M, Loizzo MR (2020) Improving kefir bioactive properties by functional enrichment with plant and agro-food waste extracts. Fermentation 6(3):83. https://doi.org/10.3390/fermentation6030083
Du X, Myracle AD (2018) Development and evaluation of kefir products made with aronia or elderberry juice: sensory and phytochemical characteristics. Int Food Res J 25(4):1373–1383
Montibeller MJ, de Lima MP, Tupuna-Yerovi DS, Rios AdO, Manfroi V (2018) Stability assessment of anthocyanins obtained from skin grape applied in kefir and carbonated water as a natural colorant. J Food Process Preserv 42(8):13698. https://doi.org/10.1111/jfpp.13698
Vagiri M, Ekholm A, Andersson SC, Johansson E, Rumpunen K (2012) An optimized method for analysis of phenolic compounds in buds, leaves, and fruits of black currant (Ribes nigrum L.). J Agr Food Chem 60(42):10501–10510
Singleton VL, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Method Enzymol 299:152–178
Apak R, Guclu K, Ozyurek M, Celik SE (2008) Mechanism of antioxidant capacity assays and the CUPRAC (cupric ion reducing antioxidant capacity) assay. Microchim Acta 160(4):413–419
Magoc T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27(21):2957–2963. https://doi.org/10.1093/bioinformatics/btr507
Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7(5):335–336. https://doi.org/10.1038/nmeth.f.303
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods. https://doi.org/10.1038/Nmeth.2604
He Y, Caporaso JG, Jiang XT, Sheng HF, Huse SM, Rideout JR et al (2015) Stability of operational taxonomic units: an important but neglected property for analyzing microbial diversity. Microbiome 3:20–30. https://doi.org/10.1186/s40168-015-0081-x
DeSantis TZ, Hugenholtz P, Keller K, Brodie EL, Larsen N, Piceno YM et al (2006) NAST: a multiple sequence alignment server for comparative analysis of 16S rRNA genes. Nucleic Acids Res 34:W394–W399
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32(5):1792–1797
Sun X-h, Zhou T-t, Wei C-h, Lan W-q, Zhao Y, Pan Y-j et al (2018) Antibacterial effect and mechanism of anthocyanin rich Chinese wild blueberry extract on various foodborne pathogens. Food Control 94:155–161. https://doi.org/10.1016/j.foodcont.2018.07.012
Gonzalez OA, Escamilla C, Danaher RJ, Dai J, Ebersole JL, Mumper RJ et al (2013) Antibacterial effects of blackberry extract target periodontopathogens. J Periodontal Res 48(1):80–86. https://doi.org/10.1111/j.1600-0765.2012.01506.x
Yang H, Hewes D, Salaheen S, Federman C, Biswas D (2014) Effects of blackberry juice on growth inhibition of foodborne pathogens and growth promotion of Lactobacillus. Food Control 37:15–20. https://doi.org/10.1016/j.foodcont.2013.08.042
Zacharof MP, Lovitt RW (2012) Bacteriocins produced by lactic acid bacteria a review article. APCBEE Proc 2:50–56. https://doi.org/10.1016/j.apcbee.2012.06.010
Song AA, In LLA, Lim SHE, Rahim RA (2017) A review on Lactococcus lactis: from food to factory. Microb Cell Fact 16(1):55. https://doi.org/10.1186/s12934-017-0669-x
Carasi P, Trejo FM, Perez PF, De Antoni GL, Serradell Mde L (2012) Surface proteins from Lactobacillus kefir antagonize in vitro cytotoxic effect of clostridium difficile toxins. Anaerobe 18(1):135–142. https://doi.org/10.1016/j.anaerobe.2011.11.002
Carasi P, Jacquot C, Romanin DE, Elie A-M, De Antoni GL, Urdaci MC et al (2014) Safety and potential beneficial properties of Enterococcus strains isolated from kefir. Int Dairy J 39(1):193–200. https://doi.org/10.1016/j.idairyj.2014.06.009
Kim DH, Jeong D, Kang IB, Kim H, Song KY, Seo KH (2017) Dual function of Lactobacillus kefiri DH5 in preventing high-fat-diet-induced obesity: direct reduction of cholesterol and upregulation of PPAR-alpha in adipose tissue. Mol Nutr Food Res 61(11):1700252. https://doi.org/10.1002/mnfr.201700252
Jung MY, Lee C, Seo MJ, Roh SW, Lee SH (2020) Characterization of a potential probiotic bacterium Lactococcus raffinolactis WiKim0068 isolated from fermented vegetable using genomic and in vitro analyses. BMC Microbiol 20(1):136. https://doi.org/10.1186/s12866-020-01820-9
Burton JP, Chanyi RM, Schultz M (2017) Common Organisms and Probiotics: Streptococcus thermophilus (Streptococcus salivarius subsp. thermophilus).In: (Edited by Martin H. Floch ). The Microbiota in Gastrointestinal Pathophysiology, Elsevier, Amsterdam, 165–169
Stanley D, Geier MS, Denman SE, Haring VR, Crowley TM, Hughes RJ et al (2013) Identification of chicken intestinal microbiota correlated with the efficiency of energy extraction from feed. Vet Microbiol 164(1–2):85–92. https://doi.org/10.1016/j.vetmic.2013.01.030
Bae JY, Kim JI, Park S, Yoo K, Kim IH, Joo W et al (2018) Effects of Lactobacillus plantarum and Leuconostoc mesenteroides probiotics on human seasonal and avian influenza viruses. J Microbiol Biotechnol 28(6):893–901. https://doi.org/10.4014/jmb.1804.04001
Lee NK, Lim SM, Cheon MJ, Paik HD (2021) Physicochemical analysis of yogurt produced by Leuconostoc mesenteroides H40 and its effects on oxidative stress in neuronal cells. Food Sci Anim Resour 41(2):261–273. https://doi.org/10.5851/kosfa.2020.e97
Carter GM, Esmaeili A, Shah H, Indyk D, Johnson M, Andreae M et al (2016) Probiotics in human immunodeficiency virus infection: a systematic review and evidence synthesis of benefits and risks. Open Forum Infect Dis 3(4):164. https://doi.org/10.1093/ofid/ofw164
van Hoek AH, Mevius D, Guerra B, Mullany P, Roberts AP, Aarts HJ (2011) Acquired antibiotic resistance genes: an overview. Front Microbiol 2:203. https://doi.org/10.3389/fmicb.2011.00203
Sachi S, Ferdous J, Sikder MH, Azizul Karim Hussani SM (2019) Antibiotic residues in milk: past, present, and future. J Adv Vet Anim Res 6(3):315–332. https://doi.org/10.5455/javar.2019.f350
Guzel-Seydim ZB, Gökırmaklı Ç, Greene AK (2021) A comparison of milk kefir and water kefir: physical, chemical, microbiological and functional properties. Trends Food Sci Technol 113:42–53. https://doi.org/10.1016/j.tifs.2021.04.041
Acknowledgements
The authors thank Scientific Research Projects Coordination Unit of Istanbul University (Project number: FBA-2022-38774) for supporting the current work.
Funding
This study was funded by Scientific Research Projects Coordination Unit of Istanbul University, Turkey. Project number: FBA-2022–38774.
Author information
Authors and Affiliations
Contributions
SA: Conceptualization, Methodology, Resources, Supervision, Project administration, Funding acquisition, Investigation, Writing—Original draft preparation. AAE: Visualization, Writing—Original draft preparation. NT: Investigation, Writing—Original draft preparation. AM: Methodology, MC: Review & Editing. İK: Methodology All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical Approval
Not applicable.
Research Involving Human and animals Participants, Their Data or Biological Material
Not applicable.
Informed Consent
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aydin, S., Erözden, A.A., Tavşanlı, N. et al. Anthocyanin Addition to Kefir: Metagenomic Analysis of Microbial Community Structure. Curr Microbiol 79, 327 (2022). https://doi.org/10.1007/s00284-022-03017-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-022-03017-x