Abstract
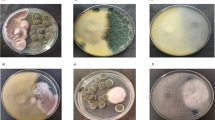
Agarwood oil, often called attar, is a valuable perfume retainer derived from Aquilaria malaccensis. It is obtained through a process of fermentation followed by distillation. The microbes involved in fermentation during agarwood processing have neither been documented nor their role in the process deciphered yet. The present study characterizes the fungi and bacteria engaged in the agarwood fermentation and assesses their colony forming units (CFU) in three categories of agarwood fermentation broth (AFB) collected from three small-scale industries of Hojai, Assam at varying time points. It was interesting to note that grade-A AFB contained the highest CFU count for both bacteria and fungi; the metabolite profile also revealed that the oil obtained from grade-A agarwood comprised of the highest number of compounds. The CFU count increased from 0 to 21 days during fermentation. A total of 105 bacteria and 9 fungi were isolated from 3 different grades of AFB. Shannon index (H' = 0.81) was observed maximum in grade-C AFB and fisher index (α = 2.6) observed maximum for grade-A AFB. The genus Bacillus with a Pi value of 0.61 exhibited dominance among isolated bacteria, while the genus Galactomyces was dominant among fungi with a Pi value of 0.43. The metabolite profiles of three grades of oil obtained after fermentation and one solvent extracted (S.E.) grade agarwood oil analyzed using GC–MS, which showed distinct differences among the oil. The outcomes of this study are expected to create new opportunities for improving oil production methods by modulating biochemical processes involved in fermentation.





Similar content being viewed by others
Availability of Data and Material
The genomic DNA sequences of the AFB bacterial and fungal isolates are openly available from the NCBI GenBank database under the accession number MG430354-MG430405 for bacteria and MG396996- MG397001 for fungi.
References
Pojanagaroon S, Kaewrak C (2005) Mechanical methods to stimulate aloes wood formation in Aquilaria crassna Pierre ex H Lec (Kritsana) trees. Acta Hort 676:161–66. https://doi.org/10.17660/actahortic.2005.676.20
Mohamed R, Jong PL, Kamziah AK (2014) Fungal inoculation inducers agarwood in young Aquilaria malaccensis trees in the nursery. J Forestry Res 25(1):201–204. https://doi.org/10.1007/s11676-013-0395-0
Thanh LV, Do TV, Son NH, Sato T, Kozan O (2015) Impacts of biological, chemical and mechanical treatments on sesquiterpene content in stems of planted Aquilaria crassna trees. Agroforest Syst 89:973–981. https://doi.org/10.1007/s10457-015-9829-3
Akter S, Islam MT, Zulkefeli M, Khan SI (2013) Agarwood production- A multidisciplinary field to be explored Bangladesh. Int J Pharm Life Sci 2(1):22–32. https://doi.org/10.3329/ijpls.v2i1.15132
Naziz PS, Das R, Sen S (2019) The Scent of Stress: Evidence from the unique fragrance of agarwood. Front Plant Sci 10:840. https://doi.org/10.3389/fpls.2019.00840
Bhattacharyya B, Datta A, Baruah HK (1952) On the formation and development of agaru in Aquilaria agallocha. Scientific Culture 18(5):240–241
Schippmann U (2001) Medicinal plants of significant trade. CITES Project S - 109, Plants Committee Document PC9 9.1.3 (rev.). pp. 51–58. BFN Scripten – 39, BFN- German Federal Agency for Nature conservation.
Yaacob S (1999) Agarwood: Trade and CITES Implementation in Malaysia. Unpublished report prepared for TRAFFIC Southeast Asia, Malaysia
CITES (2004) Convention on international trade in endangered species of wild fauna and flora. consideration of proposals for amendment of appendices I and II- Aquilaria spp. and Gyrinops spp. Thirteenth meeting of the conference of the parties, Bangkok
Jok VA, Radzi NC, Hamid KHK (2015) Agarwood oil yield as a result of changes in cell morphology due to soaking process. Procedia Soc Behav Sci 195:2443–2450
Persoon GA, Heuveling van Beek H (2008) Growing ‘The wood of the Gods’ In: Snelder D.J. Lasco R.D. Leiden (ed) Agarwood production in Southeast Asia. Smallholder tree growing for rural development and environmental services. lessons from Asia, Springer, Netherlands, pp 245–262
Mohamed R, Jong PL, Zali MS (2010) Fungal diversity in wounded stems of Aquilaria malaccensis. Fungal Divers 43:67–74. https://doi.org/10.1007/s13225-010-0039-z
Premalatha K, Kalra A (2013) Molecular phylogenetic identification of endophytic fungi isolated from resinous and healthy wood of Aquilaria malaccensis, a red listed and highly exploited medicinal tree. Fungal Ecol 6(3):205–211. https://doi.org/10.1016/j.funeco.2013.01.005
Chhipa H, Choudhary K, Kaushik N (2017) Artificial production of agarwood oil in Aquilaria sp. by fungi: a review. Phytochem Rev 16(5):835–860. https://doi.org/10.1007/s11101-017-9492-6
Jayachandran K, Sekar I, Parthiban KT, Amirtham D, Suresh KK (2014) Analysis of different grades of Agarwood (Aquilaria malaccensis Lamk.) oil through GC-MS. IJNPR 5(1):44–47
Nimnoi P, Pongsilp N, Lumyong S (2010) Endophytic Actinomycetes isolated from Aquilaria crassna Pierre ex Lec and screening of plant growth promoters production. World J Microbiol Biotechnol 26(2):193–203. https://doi.org/10.1007/s11274-009-0159-3
Bhore SJ, Preveena J, Kandaswamy KI (2013) Isolation and identification of bacterial endophytes from pharmaceutical agarwood producing Aquilaria species. Pharmacognosy Res 5(2):134–137. https://doi.org/10.4103/0974-8490.110545
Sen S, Talukdar NC, Khan M (2015) A simple metabolite profiling approach reveals critical biomolecular linkages in fragrant agarwood oil production from Aquilaria malaccensis – a traditional agro-based industry in North East India. Curr Sci 108(1):63–71. https://doi.org/10.18520/cs%2Fv108%2Fi1%2F63-71
Chen P, Yan L, Zhang S, Wu Z, Li S, Yan X, Wang N, Liang N, Li H (2017) Optimizing bioconversion of ferulic acid to vanillin by Bacillus subtilis in the stirred packed reactor using Box-Behnken design and desirability function. Food Sci Biotechnol 26(1):143–152. https://doi.org/10.1007/s10068-017-0019-0
Pitcher DG, Saunders NA, Owen RJ (1989) Rapid extraction of bacterial genomic DNA with guanidium thiocyanate. Lett Appl Microbiol 8(4):151–156. https://doi.org/10.1111/j.1472-765X.1989.tb00262.x
Jasalavich CA, Ostrofsky A, Jellison J (2000) Detection and identification of decay fungi in spruce wood by restriction fragment length polymorphism analysis of amplified genes encoding rRNA. Appl Env Microbiol 66(11):4725–4734. https://doi.org/10.1128/aem.66.11.4725-4734.2000
Whittaker RH (1972) Evolution and measurement of species diversity. Taxonomy 21:213–251
Camargo JA (1992) Temporal and spatial variations in dominance, diversity and biotic indices along a limestone stream receiving a trout farm effluent. Water Air Soil Pollut 63:343–359. https://doi.org/10.1007/BF00475501
Chhipa H, Kaushik N (2017) Fungal and bacterial diversity isolated from Aquilaria malaccensis tree and soil, induces agarospirol formation within 3 months after artificial infection. Front Microbiol 8:1286. https://doi.org/10.3389/fmicb.2017.01286
Nguyen TTT, Nguyen VD (2014) Biodiversity of major bacterial groups in association with agarwood (Aquilaria crassna) in Khanh Hoa province, Vietnam. J Viet Env 6(2):132–137. https://doi.org/10.13141/jve.vol6.no2.pp132-137
Cui JL, Guo SX, Xiao PG (2011) Antitumor and antimicrobial activities of endophytic fungi from medicinal parts of Aquilaria sinensis. J Zhejiang Univ Sci B 12(5):385–392. https://doi.org/10.1631/jzus.B1000330
Sen S, Dehingia M, Talukdar NC, Khan M (2017) Chemometric analysis reveals links in the formation of fragrant bio-molecules during agarwood (Aquilaria malaccensis) and fungal interactions. Sci Rep 7:44406. https://doi.org/10.1038/srep44406
Bailey MJ, Pessa E (1990) Strain and process for production of polygalacturonase. Enzyme Microbiol Technol 12:266–2716. https://doi.org/10.1016/0141-0229(90)90098-B
Vyas P, Gulati A (2009) Organic acid production in vitro and plant growth promotion in maize under controlled environment by phosphate-solubilizing fluorescent Pseudomonas. BMC Microbiol 9:174. https://doi.org/10.1186/1471-2180-9-174
Romero-Garcia S, Hernandez-Bustos C, Merino E, Gosset G, Martinez A (2009) Homolactic fermentation from glucose and cellobiose using Bacillus subtilis. Microb cell fact. https://doi.org/10.1186/1475-2859-8-23
Neesa L, Jahan N, Khann MAAN, Rahman MS (2017) Cellulolytic Bacillus may or may not produce β-glucosidase due to their environmental origin – a case study. J Bioremediat Biodegrad 8(6):412. https://doi.org/10.4172/2155-6199.1000412
Pervaiz I, Ahmad S, Mukhtar F, Arshad A, Imran M, Mahmood W (2015) microbial biotransformation of dexamethasone by Bacillus subtilis (ATCC 6051). Pharm Chem J 49(6):405–408. https://doi.org/10.1007/s11094-015-1294-9
Chang TS, Chiang CM, Siao YY, Wu JY (2018) Sequential biotransformation of antcin k by Bacillus subtilis ATCC 6633. Catalysts 8(9):349. https://doi.org/10.3390/catal8090349
Promon SK, Kamal W, Rahman SS, Hossain MM, Choudhury N (2018) Bioethanol production using vegetable peels medium and the effective role of cellulolytic bacterial (Bacillus subtilis) pre-treatment. F1000Res 7:271. https://doi.org/10.12688/f1000research.13952.2
Perumal M, Prabakaran JJ, kamaraj M, (2013) Isolation and characterization of potential cyanide degrading Bacillus nealsonii from different industrial effluents. Int J ChemTech Res 5(5):2357–2364
Yun H, Liang B, Kong D, Li Z, Qi G, Wang A (2017) Enhanced biotransformation of triclocarban by Ochrobactrum sp. TCC-1 under anoxic nitrate respiration conditions. Curr Microbiol. https://doi.org/10.1007/s00284-017-1214-1
Xu-Fen ZHU, Xiao-Yun WU, Yun DAI (2003) Fermentation conditions and properties of a chitosanase from Acinetobacter sp. C-17. Biosci Biotechnol Biochem 67(2):284–290. https://doi.org/10.1271/bbb.67.284
Chen YL, Yang CH, Yang FC, Ismail W, Wang PH, Shih CJ, Wu YC, Chiang YR (2016) Identification of Comamonas testosteroni as an androgen degrader in sewage. Sci Rep 6:35386. https://doi.org/10.1038/srep35386
An CY, Sun ZZ, Shen L, Ji HF (2017) Biotransformation of food spice curcumin by gut bacterium Bacillus megaterium DCMB-002 and its pharmacological implications. Food nutri res. https://doi.org/10.1080/16546628.2017.1412814
Kumar D, Dubey KK (2017) An efficient process for the transformation of betulin to betulinic acid by a strain of Bacillus megaterium. 3 Biotech 7(3):157. https://doi.org/10.1007/s13205-017-0759-9
Zhang C, Xu SH, Ma BL, Wang WW, Yu BY, Zhang J (2017) New derivatives of ursolic acid through the biotransformation by Bacillus megaterium CGMCC 1.1741 as inhibitors on nitric oxide production. Bio org Med Chem Lett 27:2575–2578. https://doi.org/10.1016/j.bmcl.2017.03.076
Sogani M, Mathur N, Bhtanagar P, Sharma P (2012) Biotransformation of amide using Bacillus sp. isolation strategy, strain characteristics and enzyme immobilization. Int J Environ Sci Technol 9:119–127. https://doi.org/10.1007/s13762-011-0005-7
Kim KM, Park JS, Coi H, Kim MS, Seo JH, Pandey RP (2018) Biosynthesis of novel daidzein derivatives using Bacillus amyloliquefaciens whole cells. Biocatal Biotransform 36(6):469–475. https://doi.org/10.1080/10242422.2018.1461212
Slivinski CT, Mallmann E, Araújo JM, Mitchell DA, Krieger N (2012) Production of surfactin by Bacillus pumilus UFPEDA 448 in solid-state fermentation using a medium based on okara with sugarcane bagasse as a bulking agent. Process Biochem 47(12):1848–1855. https://doi.org/10.1016/j.procbio.2012.06.014
Rao KV, Weisner NT (1981) Microbial Transformation of Quercetin by Bacillus cereus. Appl Environ Microbiol 42(3):450–452. https://doi.org/10.1128/aem.42.3.450-452.1981
Cichewicz RH, Kouzi SA (1998) Biotransformation of Resveratrol to Piceid by Bacillus cereus. J Nat Prod 61(10):1313–1314. https://doi.org/10.1021/np980139b
Priyalaxmi R, Murugan a, Raja P, Raj KD, (2014) Bioremediation of cadmium by Bacillus safensis (JX126862), a marine bacterium isolated from mangrove sediments. Int J Curr Microbiol App Sci 3(12):326–335
Kowal M, Rybarczyk W, Kowalski SJ (1992) On the mechanical behaviour of water soaked wood at various states of stress. Wood Sci Technol 26:295–306. https://doi.org/10.1007/BF00200165
Turner S, Pryer KM, Miao VPW, Palmer JD (1999) Investigating deep phylogenetic relationships among cyanobacteria and plastids by small subunit rRNA sequence analysis. J Eukaryot Microbiol 46:327–338
Phongpaichit S, Rungjindamal N, Rukachaisirikul V, Sakayaroj J (2006) Antimicrobial activity in cultures of endophytic fungi isolated from Garcinia species. FEMS Immunol Med Microbiol 48(3):367–372. https://doi.org/10.1111/j.1574-695x.2006.00155.x
Acknowledgements
The authors would like to thank Dr. Mohammad Ghaznavi Idris, Assistant Professor, Department of Bioengineering and Technology, GUIST, Gauhati University, Guwahati, as well as the two anonymous reviewers for their constructive advice in improving the manuscript's quality. MRI also acknowledges the Ministry of Minority Affairs, Govt. of India, for the Maulana Azad National Fellowship.
Funding
This research was supported by the Department of Biotechnology, Government of India, Research (Grant no BT/PR6346/GBD/27/405/2012).
Author information
Authors and Affiliations
Contributions
SB: Conceptualization, funding acquisition, resources, Editing and Supervision of manuscript, RI: Conducted fieldwork, methodology, statistical analysis, manuscript writing, RI and CC: Experimentation and analysis.
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no conflict of interest to declare relevant to the contents of this article.
Ethics Approval
Not applicable.
Consent for Publication
All authors agreed with the content and gave explicit consent to submit this article to the journal Current Microbiology.
Code Availability
Not applicable.
Declaration of Deposition in Repositories
All the genomic DNA sequences of the AFB bacterial and fungal isolates are deposited to the NCBI GenBank database (MG430354-MG430405, MG396996- MG397001).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Islam, M.R., Chakraborty, C. & Banu, S. Isolation and Characterization of Bacteria and Fungi Associated with Agarwood Fermentation. Curr Microbiol 79, 313 (2022). https://doi.org/10.1007/s00284-022-02999-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-022-02999-y