Abstract
Purpose
For unresectable locally advanced (UR-LA) pancreatic cancer, chemoradiotherapy has been recommended by the NCCN guidelines. We designed a chemoradiotherapy protocol using nab-paclitaxel combined with gemcitabine (GnP) for patients with UR-LA pancreatic cancer. The purpose of this phase I study was to determine a recommended dose (RD) for this novel regimen.
Methods
Patients with UR-LA pancreatic cancer were eligible. The frequency of dose-limiting toxicities (DLTs) was evaluated, and the RD was determined. Patients were classified according to the designated dose levels of chemoradiotherapy using the GnP regimen. After additional 6 cycles of the GnP regimen were administered, surgery was considered if the patients had stable disease and tumor marker levels had normalized.
Results
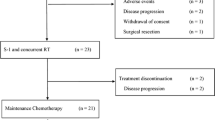
DLT (grade 4 thrombocytopenia) was observed only in 1 of 12 patients, and the RD was set at level 3. Grade 3–4 leukopenia was observed in 9 (75.0%) patients, and neutropenia in 7 (58.3%). The response rate was 41.7%, and the disease control rate was 100%. Conversion surgery was performed in 6 (50%) patients, and curative resection (R0) was performed in all 6 patients (100%). Stratification according to the Evans classification system demonstrated one patient with grade 1b, one with grade 2, two with grade 3, and two with grade 4 disease.
Conclusion
The RD for weekly administration was 800 mg/m2 for gemcitabine and 100 mg/m2 for nab-paclitaxel with a 50.4 Gy radiation. The GnP regimen at this dosage was promising with 6 of 12 patients proceeding to conversion surgery, and should be evaluated further in a phase II trial.

Similar content being viewed by others
References
Egawa S, Toma H, Ohigashi H, Okusaka T, Nakao A, Hatori T et al (2012) Japan Pancreatic Cancer Registry; 30th year anniversary: Japan Pancreas Society. Pancreas 41(7):985–992
Von Hoff DD, Ramanathan RK, Borad MJ, Laheru DA, Smith LS, Wood TE et al (2011) Gemcitabine plus nab-paclitaxel is an active regimen in patients with advanced pancreatic cancer: a phase I/II trial. J Clin Oncol 29(34):4548–4554
Von Hoff DD, Ervin T, Arena FP, Chiorean EG, Infante J, Moore M et al (2013) Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 369(18):1691–1703
Conroy T, Desseigne F, Ychou M, Bouche O, Guimbaud R, Becouarn Y et al (2011) FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 364(19):1817–1825
Network NCC (2017) National Comprehensive Cancer Network NCCN clinical practice guidelines in oncology (NCCN Guidelines)
Moertel CG, Frytak S, Hahn RG, O’Connell MJ, Reitemeier RJ, Rubin J et al (1981) Therapy of locally unresectable pancreatic carcinoma: a randomized comparison of high dose (6000 rads) radiation alone, moderate dose radiation (4000 rads + 5-fluorouracil), and high dose radiation + 5-fluorouracil: the Gastrointestinal Tumor Study Group. Cancer 48(8):1705–1710
Shinchi H, Takao S, Noma H, Matsuo Y, Mataki Y, Mori S et al (2002) Length and quality of survival after external-beam radiotherapy with concurrent continuous 5-fluorouracil infusion for locally unresectable pancreatic cancer. Int J Radiat Oncol Biol Phys 53(1):146–150
Klaassen DJ, MacIntyre JM, Catton GE, Engstrom PF, Moertel CG (1985) Treatment of locally unresectable cancer of the stomach and pancreas: a randomized comparison of 5-fluorouracil alone with radiation plus concurrent and maintenance 5-fluorouracil–an Eastern Cooperative Oncology Group study. J Clin Oncol 3(3):373–378
Gastrointestinal Tumor Study Group (1988) Treatment of locally unresectable carcinoma of the pancreas: comparison of combined-modality therapy (chemotherapy plus radiotherapy) to chemotherapy alone. J Natl Cancer Inst 80(10):751–755
Chauffert B, Mornex F, Bonnetain F, Rougier P, Mariette C, Bouche O et al (2008) Phase III trial comparing intensive induction chemoradiotherapy (60 Gy, infusional 5-FU and intermittent cisplatin) followed by maintenance gemcitabine with gemcitabine alone for locally advanced unresectable pancreatic cancer. Definitive results of the 2000-01 FFCD/SFRO study. Ann Oncol 19(9):1592–1599
Loehrer PJ Sr., Feng Y, Cardenes H, Wagner L, Brell JM, Cella D et al (2011) Gemcitabine alone versus gemcitabine plus radiotherapy in patients with locally advanced pancreatic cancer: an Eastern Cooperative Oncology Group trial. J Clin Oncol 29(31):4105–4112
de Lange SM, van Groeningen CJ, Meijer OW, Cuesta MA, Langendijk JA, van Riel JM et al (2002) Gemcitabine-radiotherapy in patients with locally advanced pancreatic cancer. Eur J Cancer 38(9):1212–1217
Okusaka T, Ito Y, Ueno H, Ikeda M, Takezako Y, Morizane C et al (2004) Phase II study of radiotherapy combined with gemcitabine for locally advanced pancreatic cancer. Br J Cancer 91(4):673–677
Network NCC (2015) National Comprehensive Cancer Network NCCN clinical practice guidelines in oncology (NCCN Guidelines)
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247
Ioka TKT, Takada R et al (2017) The phase I study of concurrent chemoradiotherapy using a combination of gemcitabine and nab-paclitaxel for unresectable locally advanced pancreatic cancer: update. In: Gastrointestinal cancer symposium (abstr 462)
Evans DB, Rich TA, Byrd DR, Cleary KR, Connelly JH, Levin B et al (1992) Preoperative chemoradiation and pancreaticoduodenectomy for adenocarcinoma of the pancreas. Arch Surg 127(11):1335–1339
Moertel CG, Childs DS Jr, Reitemeier RJ, Colby MY Jr, Holbrook MA (1969) Combined 5-fluorouracil and supervoltage radiation therapy of locally unresectable gastrointestinal cancer. Lancet 2(7626):865–867
Ueno H, Ikeda M, Ueno M, Mizuno N, Ioka T, Omuro Y et al (2016) Phase I/II study of nab-paclitaxel plus gemcitabine for chemotherapy-naive Japanese patients with metastatic pancreatic cancer. Cancer Chemother Pharmacol 77(3):595–603
Strobel O, Berens V, Hinz U, Hartwig W, Hackert T, Bergmann F et al (2012) Resection after neoadjuvant therapy for locally advanced, “unresectable” pancreatic cancer. Surgery 152(3 Suppl 1):S33-42
Ferrone CR, Marchegiani G, Hong TS, Ryan DP, Deshpande V, McDonnell EI et al (2015) Radiological and surgical implications of neoadjuvant treatment with FOLFIRINOX for locally advanced and borderline resectable pancreatic cancer. Ann Surg 261(1):12–17
Blazer M, Wu C, Goldberg RM, Phillips G, Schmidt C, Muscarella P et al (2015) Neoadjuvant modified (m) FOLFIRINOX for locally advanced unresectable (LAPC) and borderline resectable (BRPC) adenocarcinoma of the pancreas. Ann Surg Oncol 22(4):1153–1159
Nitsche U, Wenzel P, Siveke JT, Braren R, Holzapfel K, Schlitter AM et al (2015) Resectability after first-line FOLFIRINOX in Initially unresectable locally advanced pancreatic cancer: a single-center experience. Ann Surg Oncol 22(Suppl 3):S1212–S1220
Hackert T, Sachsenmaier M, Hinz U, Schneider L, Michalski CW, Springfeld C et al (2016) Locally advanced pancreatic cancer: neoadjuvant therapy with folfirinox results in resectability in 60% of the patients. Ann Surg 264(3):457–463
Acknowledgements
We are deeply grateful to the members in Nagoya University cancer board of pancreatic cancer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors in this study have no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Yamada, S., Fujii, T., Yokoyama, Y. et al. Phase I study of chemoradiotherapy using gemcitabine plus nab-paclitaxel for unresectable locally advanced pancreatic cancer. Cancer Chemother Pharmacol 81, 815–821 (2018). https://doi.org/10.1007/s00280-018-3554-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-018-3554-3