Abstract
Purpose
S-1 chemotherapy is effective against advanced biliary tract cancer. The purpose was to evaluate the efficacy and safety of S-1-based concurrent chemoradiotherapy in patients with advanced biliary tract cancer.
Methods
Patients with pathologically-proven advanced biliary tract cancer were eligible. S-1 was orally administered at a dose of 40 mg/m2, twice daily from day 1 to 14 and from day 22 to 35; concurrent radiotherapy of 180–200 cGy per fraction was delivered in 25–28 fractions. After treatment completion, tumor response was evaluated by computed tomography. In the first stage of the optimal two-stage phase II design, 18 patients were required.
Results
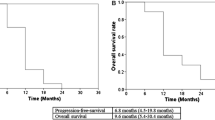
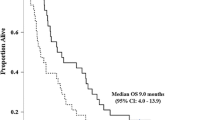
Twenty patients were enrolled between August 2006 and February 2009. The median age was 62.5 years (range 45–77 years). The median follow-up time was 11.6 months (range 1.9–49.1 months). Fifteen patients (75 %) had extrahepatic cholangiocarcinoma, two patients (10 %) had intrahepatic cholangiocarcinoma, and three patients (15 %) had gallbladder cancer. After treatment, a partial response was achieved in three patients (15 %), and stable disease was achieved in 14 patients (70 %). The overall response rate was 15 %, and the disease stabilization rate was 85 %. There was no grade 4 toxicity or treatment-related death. The common grade 3 toxicities were thrombocytopenia (15 %), neutropenia (10 %), and nausea (10 %). The median progression-free survival and median overall survival were 5.9 months (range 2.2–9.5 months) and 10.8 months (range 1.1–20.4 months), respectively.
Conclusions
This study shows that S-1-based concurrent chemoradiotherapy is feasible and tolerable in patients with advanced biliary tract cancer. It will be further confirmed in a following large-scale phase II study.


Similar content being viewed by others
References
Valle J, Wasan H, Palmer DH, Cunningham D, Anthoney A, Maraveyas A, Madhusudan S, Iveson T, Hughes S, Pereira SP, Roughton M, Bridgewater J, Investigators ABCT (2010) Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med 362:1273–1281. doi:10.1056/NEJMoa0908721
Yi SW, Kang DR, Kim KS, Park MS, Seong J, Park JY, Bang SM, Song SY, Chung JB, Park SW (2014) Efficacy of concurrent chemoradiotherapy with 5-fluorouracil or gemcitabine in locally advanced biliary tract cancer. Cancer Chemother Pharmacol 73:191–198. doi:10.1007/s00280-013-2340-5
Ghafoori AP, Nelson JW, Willett CG, Chino J, Tyler DS, Hurwitz HI, Uronis HE, Morse MA, Clough RW, Czito BG (2011) Radiotherapy in the treatment of patients with unresectable extrahepatic cholangiocarcinoma. Int J Radiat Oncol Biol Phys 81:654–659. doi:10.1016/j.ijrobp.2010.06.018
Ueno H, Okusaka T, Ikeda M, Takezako Y, Morizane C (2004) Phase II study of S-1 in patients with advanced biliary tract cancer. Br J Cancer 91:1769–1774. doi:10.1038/sj.bjc.6602208
Sasaki T, Isayama H, Yashima Y, Yagioka H, Kogure H, Arizumi T, Togawa O, Matsubara S, Ito Y, Nakai Y, Sasahira N, Hirano K, Tsujino T, Tada M, Kawabe T, Omata M (2009) S-1 monotherapy in patients with advanced biliary tract cancer. Oncology 77:71–74. doi:10.1159/000226214
Furuse J, Okusaka T, Boku N, Ohkawa S, Sawaki A, Masumoto T, Funakoshi A (2008) S-1 monotherapy as first-line treatment in patients with advanced biliary tract cancer: a multicenter phase II study. Cancer Chemother Pharmacol 62:849–855. doi:10.1007/s00280-007-0673-7
Shirasaka T, Shimamato Y, Ohshimo H, Yamaguchi M, Kato T, Yonekura K, Fukushima M (1996) Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs 7:548–557
Saif MW, Syrigos KN, Katirtzoglou NA (2009) S-1: a promising new oral fluoropyrimidine derivative. Expert Opin Investig Drugs 18:335–348. doi:10.1517/13543780902729412
Shirasaka T (2009) Development history and concept of an oral anticancer agent S-1 (TS-1): its clinical usefulness and future vistas. Jpn J Clin Oncol 39:2–15. doi:10.1093/jjco/hyn127
Ikeda M, Ioka T, Ito Y, Yonemoto N, Nagase M, Yamao K, Miyakawa H, Ishii H, Furuse J, Sato K, Sato T, Okusaka T (2013) A multicenter phase II trial of S-1 with concurrent radiation therapy for locally advanced pancreatic cancer. Int J Radiat Oncol Biol Phys 85:163–169. doi:10.1016/j.ijrobp.2012.03.059
Kim HM, Bang S, Park JY, Seong J, Song SY, Chung JB, Park SW (2009) Phase II trial of S-1 and concurrent radiotherapy in patients with locally advanced pancreatic cancer. Cancer Chemother Pharmacol 63:535–541. doi:10.1007/s00280-008-0836-1
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Simon R (1989) Optimal two-stage designs for phase II clinical trials. Control Clin Trials 10:1–10
Leong E, Chen WW, Ng E, Van Hazel G, Mitchell A, Spry N (2012) Outcomes from combined chemoradiotherapy in unresectable and locally advanced resected cholangiocarcinoma. J Gastrointest Cancer 43:50–55. doi:10.1007/s12029-010-9213-5
Deodato F, Clemente G, Mattiucci GC, Macchia G, Costamagna G, Giuliante F, Smaniotto D, Luzi S, Valentini V, Mutignani M, Nuzzo G, Cellini N, Morganti AG (2006) Chemoradiation and brachytherapy in biliary tract carcinoma: long-term results. Int J Radiat Oncol Biol Phys 64:483–488. doi:10.1016/j.ijrobp.2005.07.977
Czito BG, Anscher MS, Willett CG (2006) Radiation therapy in the treatment of cholangiocarcinoma. Oncology (Williston Park) 20:873–884 (discussion 886-878, 893-875)
Das P, Wolff RA, Abbruzzese JL, Varadhachary GR, Evans DB, Vauthey JN, Baschnagel A, Delclos ME, Krishnan S, Janjan NA, Crane CH (2006) Concurrent capecitabine and upper abdominal radiation therapy is well tolerated. Radiat Oncol 1:41. doi:10.1186/1748-717X-1-41
Macdonald OK, Crane CH (2002) Palliative and postoperative radiotherapy in biliary tract cancer. Surg Oncol Clin N Am 11:941–954
Pretz JL, Wo JY, Mamon HJ, Kachnic LA, Hong TS (2013) Chemoradiation therapy: localized esophageal, gastric, and pancreatic cancer. Surg Oncol Clin N Am 22:511–524. doi:10.1016/j.soc.2013.02.005
Conflict of interest
The authors have no financial conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
H. M. Kim and K. J. Lee equally contributed to this work.
Rights and permissions
About this article
Cite this article
Kim, H.M., Lee, K.J., Cha, J. et al. A pilot study of S-1-based concurrent chemoradiotherapy in patients with biliary tract cancer. Cancer Chemother Pharmacol 74, 861–865 (2014). https://doi.org/10.1007/s00280-014-2565-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-014-2565-y