Abstract
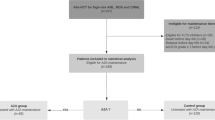
The combination of hypomethylating agents and venetoclax has revolutionized the therapeutic landscape of acute myeloid leukemia (AML), especially for patients previously deemed unfit for curative–intent treatment. Some of these patients undergo allogeneic hematopoietic cell transplant (alloHCT); yet, there are scarce data regarding transplantation outcomes. We conducted a multicenter nationwide retrospective cohort study, including patients with AML who underwent alloHCT in CR1 after frontline treatment with azacitidine plus venetoclax only (aza-ven group). We collected a historical control group of patients who achieved CR1 after first-line intensive chemotherapy only, followed by alloHCT (intensive group). Patients in the aza-ven group (n = 24) were transplanted between 2019 and 2021. Compared to the intensive group, patients in the aza-ven group were older (median age 71.7 vs. 58.4 years), had higher incidence of therapy-related AML and AML with antecedent hematologic disorder and had more often adverse cytogenetics. They had a higher percentage of allografts from matched-unrelated donors, and reduced intensity conditioning was more commonly used. The estimated 12 months non relapse mortality was 19.1% in the aza-ven group and 11.8% in the intensive group. The estimated 12 months relapse-free survival and overall survival were 58% and 63% in the aza-ven group and 54% and 70% in the intensive group, respectively. The cumulative incidence of acute GVHD at 6 months and of chronic GVHD at 12 months were 58% and 40% in the aza-ven group and 62% and 42% in the intensive group, respectively. Analysis of the aza-ven group revealed that HCT-CI score and ELN risk category were predictive of RFS in both univariate analysis as well as multivariate analysis. Our data suggests that alloHCT for AML patients achieving first CR with aza-ven appears feasible, with short-term post-transplant outcomes similar to those expected after traditional intensive chemotherapy.


Similar content being viewed by others
Data availability
Upon request, from corresponding author.
References
Dohner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Buchner T, Dombret H, Ebert BL, Fenaux P, Larson RA, Levine RL, Lo-Coco F, Naoe T, Niederwieser D, Ossenkoppele GJ, Sanz M, Sierra J, Tallman MS, Tien HF, Wei AH, Lowenberg B, Bloomfield CD (2017) Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 129(4):424–447. https://doi.org/10.1182/blood-2016-08-733196
Koreth J, Schlenk R, Kopecky KJ, Honda S, Sierra J, Djulbegovic BJ, Wadleigh M, DeAngelo DJ, Stone RM, Sakamaki H, Appelbaum FR, Dohner H, Antin JH, Soiffer RJ, Cutler C (2009) Allogeneic stem cell transplantation for acute myeloid leukemia in first complete remission: systematic review and meta-analysis of prospective clinical trials. JAMA 301(22):2349–2361. https://doi.org/10.1001/jama.2009.813
Cornelissen JJ, Blaise D (2016) Hematopoietic stem cell transplantation for patients with AML in first complete remission. Blood 127(1):62–70. https://doi.org/10.1182/blood-2015-07-604546
Yanada M (2021) The evolving concept of indications for allogeneic hematopoietic cell transplantation during first complete remission of acute myeloid leukemia. Bone Marrow Transplant. https://doi.org/10.1038/s41409-021-01247-4
DiNardo CD, Jonas BA, Pullarkat V, Thirman MJ, Garcia JS, Wei AH, Konopleva M, Dohner H, Letai A, Fenaux P, Koller E, Havelange V, Leber B, Esteve J, Wang J, Pejsa V, Hajek R, Porkka K, Illes A, Lavie D, Lemoli RM, Yamamoto K, Yoon SS, Jang JH, Yeh SP, Turgut M, Hong WJ, Zhou Y, Potluri J, Pratz KW (2020) Azacitidine and Venetoclax in previously untreated acute myeloid leukemia. N Engl J Med 383(7):617–629. https://doi.org/10.1056/NEJMoa2012971
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, Storer B (2005) Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 106(8):2912–2919. https://doi.org/10.1182/blood-2005-05-2004
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, Apperley J, Slavin S, Pasquini M, Sandmaier BM, Barrett J, Blaise D, Lowski R, Horowitz M (2009) Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant 15(12):1628–1633. https://doi.org/10.1016/j.bbmt.2009.07.004
Services UDoHaH (2017) Common terminology criteria for adverse events v5.0
(CTCAE) [Internet] Princ Pract Clin Trial Med. https://ctep.cancer.gov/protocolDevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_8.5x11.pdf. 2021
Harris AC, Young R, Devine S, Hogan WJ, Ayuk F, Bunworasate U, Chanswangphuwana C, Efebera YA, Holler E, Litzow M, Ordemann R, Qayed M, Renteria AS, Reshef R, Wolfl M, Chen YB, Goldstein S, Jagasia M, Locatelli F, Mielke S, Porter D, Schechter T, Shekhovtsova Z, Ferrara JL, Levine JE (2016) International, multicenter standardization of acute graft-versus-host disease clinical data collection: a report from the Mount Sinai Acute GVHD International Consortium. Biol Blood Marrow Transplant 22(1):4–10. https://doi.org/10.1016/j.bbmt.2015.09.001
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, Palmer J, Weisdorf D, Treister NS, Cheng GS, Kerr H, Stratton P, Duarte RF, McDonald GB, Inamoto Y, Vigorito A, Arai S, Datiles MB, Jacobsohn D, Heller T, Kitko CL, Mitchell SA, Martin PJ, Shulman H, Wu RS, Cutler CS, Vogelsang GB, Lee SJ, Pavletic SZ, Flowers ME (2015) National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant 21 (3):389–401 e381. doi:https://doi.org/10.1016/j.bbmt.2014.12.001
Schuh AC, Dohner H, Pleyer L, Seymour JF, Fenaux P, Dombret H (2017) Azacitidine in adult patients with acute myeloid leukemia. Crit Rev Oncol Hematol 116:159–177. https://doi.org/10.1016/j.critrevonc.2017.05.010
Souers AJ, Leverson JD, Boghaert ER, Ackler SL, Catron ND, Chen J, Dayton BD, Ding H, Enschede SH, Fairbrother WJ, Huang DC, Hymowitz SG, Jin S, Khaw SL, Kovar PJ, Lam LT, Lee J, Maecker HL, Marsh KC, Mason KD, Mitten MJ, Nimmer PM, Oleksijew A, Park CH, Park CM, Phillips DC, Roberts AW, Sampath D, Seymour JF, Smith ML, Sullivan GM, Tahir SK, Tse C, Wendt MD, Xiao Y, Xue JC, Zhang H, Humerickhouse RA, Rosenberg SH, Elmore SW (2013) ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat Med 19(2):202–208. https://doi.org/10.1038/nm.3048
Aldoss I, Pullarkat V, Stein AS (2021) Venetoclax-containing regimens in acute myeloid leukemia. Ther Adv Hematol 12:2040620720986646. https://doi.org/10.1177/2040620720986646
Keith W, Pratz CDD, Arellano ML, Letai AG, Thirman M, Pullarkat VA, Roboz GJ, Becker PS, Hong W-J, Jiang Qi, Hayslip J, Potluri J, Pollyea DA (2019) Outcomes after stem cell transplant in older patients with acute myeloid leukemia treated with venetoclax-based therapies. Blood 134:264. https://doi.org/10.1182/blood-2019-127251
Apel A, Moshe Y, Ofran Y, Gural A, Wolach O, Ganzel C, Canaani J, Zektser M, Duek A, Stemer G, Hellman I, Basood M, Frisch A, Leibovitch C, Koren-Michowitz M (2021) Venetoclax combinations induce high response rates in newly diagnosed acute myeloid leukemia patients ineligible for intensive chemotherapy in routine practice. Am J Hematol. https://doi.org/10.1002/ajh.26190
Sandhu KS, Dadwal S, Yang D, Mei M, Palmer J, Salhotra A, Al Malki M, Aribi A, Ali H, Khaled S, Forman SJ, Snyder D, Nakamura R, Stein AS, Marcucci G, Aldoss I, Pullarkat V (2020) Outcome of allogeneic hematopoietic cell transplantation after venetoclax and hypomethylating agent therapy for acute myelogenous leukemia. Biol Blood Marrow Transplant 26(12):e322–e327. https://doi.org/10.1016/j.bbmt.2020.08.027
Winters AC, Gutman JA, Purev E, Nakic M, Tobin J, Chase S, Kaiser J, Lyle L, Boggs C, Halsema K, Schowinsky JT, Rosser J, Ewalt MD, Siegele B, Rana V, Schuster S, Abbott D, Stevens BM, Jordan CT, Smith C, Pollyea DA (2019) Real-world experience of venetoclax with azacitidine for untreated patients with acute myeloid leukemia. Blood Adv 3(20):2911–2919. https://doi.org/10.1182/bloodadvances.2019000243
Daneshbod Y, Kohan L, Taghadosi V, Weinberg OK, Arber DA (2019) Prognostic significance of complex karyotypes in acute myeloid leukemia. Curr Treat Options Oncol 20(2):15. https://doi.org/10.1007/s11864-019-0612-y
Mrozek K (2008) Cytogenetic, molecular genetic, and clinical characteristics of acute myeloid leukemia with a complex karyotype. Semin Oncol 35(4):365–377. https://doi.org/10.1053/j.seminoncol.2008.04.007
Kuendgen A, Muller-Thomas C, Lauseker M, Haferlach T, Urbaniak P, Schroeder T, Brings C, Wulfert M, Meggendorfer M, Hildebrandt B, Betz B, Royer-Pokora B, Gattermann N, Haas R, Germing U, Gotze KS (2018) Efficacy of azacitidine is independent of molecular and clinical characteristics - an analysis of 128 patients with myelodysplastic syndromes or acute myeloid leukemia and a review of the literature. Oncotarget 9(45):27882–27894. https://doi.org/10.18632/oncotarget.25328
Aldoss I, Yang D, Aribi A, Ali H, Sandhu K, Al Malki MM, Mei M, Salhotra A, Khaled S, Nakamura R, Snyder D, O’Donnell M, Stein AS, Forman SJ, Marcucci G, Pullarkat V (2018) Efficacy of the combination of venetoclax and hypomethylating agents in relapsed/refractory acute myeloid leukemia. Haematologica 103(9):e404–e407. https://doi.org/10.3324/haematol.2018.188094
Daniel A, Pollyea AW, Jordan CT, Smith C, Gutman JA (2020) Allogeneic transplant improves AML outcomes compared to maintenance venetoclax and azacitidine following response to initial venetoclax and azacitidine therapy. Blood 136:24. https://doi.org/10.1182/blood-2020-138821
Candoni A, Rambaldi A, Fanin R, Velardi A, Arcese W, Ciceri F, Lazzarotto D, Lussana F, Olivieri J, Grillo G, Parma M, Bruno B, Sora F, Bernasconi P, Saccardi R, Foa R, Sessa M, Bresciani P, Giglio F, Picardi A, Busca A, Sica S, Perruccio K, Zucchetti E, Diral E, Iori AP, Colombo AA, Tringali S, Santarone S, Irrera G, Mancini S, Zallio F, Malagola M, Albano F, Carella AM, Olivieri A, Tecchio C, Dominietto A, Vacca A, Sorasio R, Orciuolo E, Risitano AM, Leotta S, Cortelezzi A, Mammoliti S, Oldani E, Bonifazi F, Gitmo, (2019) Outcome of allogeneic hematopoietic stem cell transplantation in adult patients with philadelphia chromosome-positive acute lymphoblastic leukemia in the era of tyrosine kinase inhibitors: a registry-based study of the Italian Blood and Marrow Transplantation Society (GITMO). Biol Blood Marrow Transplant 25(12):2388–2397. https://doi.org/10.1016/j.bbmt.2019.07.037
Foa R, Vitale A, Vignetti M, Meloni G, Guarini A, De Propris MS, Elia L, Paoloni F, Fazi P, Cimino G, Nobile F, Ferrara F, Castagnola C, Sica S, Leoni P, Zuffa E, Fozza C, Luppi M, Candoni A, Iacobucci I, Soverini S, Mandelli F, Martinelli G, Baccarani M, Party GALW (2011) Dasatinib as first-line treatment for adult patients with Philadelphia chromosome-positive acute lymphoblastic leukemia. Blood 118(25):6521–6528. https://doi.org/10.1182/blood-2011-05-351403
Author information
Authors and Affiliations
Contributions
OP contributed to research design, acquisition of data, analysis and interpretation of data, drafted the manuscript and approved the submitted and final versions; SS contributed to research design, acquisition of data, analysis and interpretation of data, drafted the manuscript and approved the submitted and final versions; RR contributed to research design, acquisition of data, revised the manuscript critically and approved the submitted and final versions; AS contributed to research design, acquisition of data, revised the manuscript critically and approved the submitted and final versions; LS contributed to the acquisition of data, revised the manuscript critically and approved the submitted and final versions; BA contributed to the acquisition of data, revised the manuscript critically and approved the submitted and final versions; OW contributed to research design, analysis and interpretation of data, revised the manuscript critically and approved the submitted and final versions; TS contributed to research design, analysis and interpretation of data, revised the manuscript critically and approved the submitted and final versions; RY contributed to the acquisition of data, revised the manuscript critically and approved the submitted and final versions; OA contributed to the acquisition of data, revised the manuscript critically and approved the submitted and final versions; PR contributed to research design, analysis and interpretation of data, revised the manuscript critically and approved the submitted and final versions; MY contributed to research design, analysis and interpretation of data, drafted the manuscript and approved the submitted and final versions;
Ethics declarations
Conflicts of interest
None.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pasvolsky, O., Shimony, S., Ram, R. et al. Allogeneic hematopoietic cell transplantation for acute myeloid leukemia in first complete remission after 5-azacitidine and venetoclax: a multicenter retrospective study. Ann Hematol 101, 379–387 (2022). https://doi.org/10.1007/s00277-021-04693-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-021-04693-8