Abstract
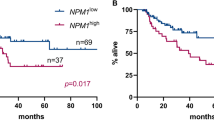
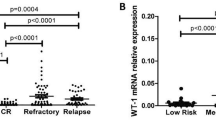
Many studies have confirmed that overexpressed WT1 exists in leukemic cells, especially in AML. However, the immunophenotypic features of this sort of leukemic cells remain to be unclarified. We retrospectively analyzed the immunophenotype of 283 newly diagnosed AML patients with intermediated and poor cytogenetic risk to evaluate the correlation between phenotype and WT1 overexpression. EVI1 transcripts, KMT2A-PTD, FLT3-ITD, and NPM1 mutations were simultaneously assessed. Our results revealed that overexpressed WT1 was significantly associated with the expression of CD117, CD13, and CD123. Besides, leukemic cells with WT1 overexpression also lacked lymphoid and myeloid differentiation-related markers. FAB subtype M2 patients had higher WT1 levels, compared with other FAB subtype. Multivariate analysis was proved that NPM1 mutation, M2 subtype, and the expression of CD123 were independently associated with WT1 overexpression. These indicated that AML with overexpressed WT1 was proliferated and blocked in the early stage of AML development. It presumably provided some clues to detect overexpressed WT1 cells via multiparameter flow cytometry. CD123-targeted drugs might become one of the alternative treatments for patients with WT1 overexpression.
Similar content being viewed by others
References
Yang L, Han Y, Suarez Saiz F, Minden MD (2007) A tumor suppressor and oncogene: the WT1 story. Leukemia 21(5):868–876. https://doi.org/10.1038/sj.leu.2404624
Inoue K, Ogawa H, Sonoda Y, Kimura T, Sakabe H, Oka Y, Miyake S, Tamaki H, Oji Y, Yamagami T, Tatekawa T, Soma T, Kishimoto T, Sugiyama H (1997) Aberrant overexpression of the Wilms tumor gene (WT1) in human leukemia. Blood 89(4):1405–1412
Hidaka D, Onozawa M, Hashiguchi J, Miyashita N, Kasahara K, Fujisawa S, Hayase E, Okada K, Shiratori S, Goto H, Sugita J, Nakagawa M, Hashimoto D, Kahata K, Endo T, Yamamoto S, Tsutsumi Y, Haseyama Y, Nagashima T, Mori A, Ota S, Sakai H, Ishihara T, Imai K, Miyagishima T, Kakinoki Y, Kurosawa M, Kobayashi H, Iwasaki H, Shimizu C, Kondo T, Teshima T (2018) Wilms tumor 1 expression at diagnosis correlates with genetic abnormalities and polymorphism but is not independently prognostic in acute myelogenous leukemia: a Hokkaido leukemia net study. Clin Lymphoma Myeloma Leuk 18(11):e469–e479. https://doi.org/10.1016/j.clml.2018.07.291
Cilloni D, Renneville A, Hermitte F, Hills RK, Daly S, Jovanovic JV, Gottardi E, Fava M, Schnittger S, Weiss T, Izzo B, Nomdedeu J, van der Heijden A, van der Reijden BA, Jansen JH, van der Velden VH, Ommen H, Preudhomme C, Saglio G, Grimwade D (2009) Real-time quantitative polymerase chain reaction detection of minimal residual disease by standardized WT1 assay to enhance risk stratification in acute myeloid leukemia: a European LeukemiaNet study. J Clin Oncol 27(31):5195–5201. https://doi.org/10.1200/JCO.2009.22.4865
Paietta E, Wiernik PH, Andersen J, Bennett J, Yunis J (1993) Acute myeloid leukemia M4 with inv(16) (p13q22) exhibits a specific immunophenotype with CD2 expression. Blood 82(8):2595
Andrieu V, Radford-Weiss I, Troussard X, Chane C, Valensi F, Guesnu M, Haddad E, Viguier F, Dreyfus F, Varet B, Flandrin G, Macintyre E (1996) Molecular detection of t(8;21)/AML1-ETO in AML M1/M2: correlation with cytogenetics, morphology and immunophenotype. Br J Haematol 92(4):855–865
Gorczyca W (2012) Acute promyelocytic leukemia: four distinct patterns by flow cytometry immunophenotyping. Pol J Pathol 63(1):8–17
(2019) National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology. Acute Myeloid Leukemia. Version 3.2018 (URL: https://www.nccn.org)
Zhou Y, Wood BL (2017) Methods of detection of measurable residual disease in AML. Curr Hematol Malig Rep 12(6):557–567. https://doi.org/10.1007/s11899-017-0419-5
Ruan GR, Li JL, Qin YZ, Li LD, Xie M, Chang Y, Zhang Y, Liu YR, Jiang B, Chen SS (2009) Nucleophosmin mutations in Chinese adults with acute myelogenous leukemia. Ann Hematol 88(2):159–166. https://doi.org/10.1007/s00277-008-0591-8
Yazhen Q, Honghu Z, Bin J, Jinlan L, Xijing L, Lingdi L, Guorui R, Yanrong L, Shanshan C, Xiaojun H (2009) Expression patterns of WT1 and PRAME in acute myeloid leukemia patients and their usefulness for monitoring minimal residual disease. Leuk Res 33(3):384–390. https://doi.org/10.1016/j.leukres.2008.08.026
Qin Y-Z, Zhao T, Zhu H-H, Wang J, Jia J-S, Lai Y-Y, Zhao X-S, Shi H-X, Liu Y-R, Jiang H, Huang X-J, Jiang Q (2018) High EVI1 expression predicts poor outcomes in adult acute myeloid leukemia patients with intermediate cytogenetic risk receiving chemotherapy. Med Sci Monit 24:758–767. https://doi.org/10.12659/msm.905903
Liu YR, Zhu HH, Ruan GR, Qin YZ, Shi HX, Lai YY, Chang Y, Wang YZ, Lu D, Hao L, Li JL, Li LD, Jiang B, Huang XJ (2013) NPM1-mutated acute myeloid leukemia of monocytic or myeloid origin exhibit distinct immunophenotypes. Leuk Res 37(7):737–741. https://doi.org/10.1016/j.leukres.2013.03.009
Syampurnawati M, Tatsumi E, Ardianto B, Takenokuchi M, Nakamachi Y, Kawano S, Kumagai S, Saigo K, Matsui T, Takahashi T, Nagai K, Gunadi, Nishio H, Yabe H, Kondo S, Hayashi Y (2008) DR negativity is a distinctive feature of M1/M2 AML cases with NPM1 mutation. Leuk Res 32(7):1141–1143. https://doi.org/10.1016/j.leukres.2007.11.017
Chen W, Konoplev S, Medeiros LJ, Koeppen H, Leventaki V, Vadhan-Raj S, Jones D, Kantarjian HM, Falini B, Bueso-Ramos CE (2009) Cuplike nuclei (prominent nuclear invaginations) in acute myeloid leukemia are highly associated with FLT3 internal tandem duplication and NPM1 mutation. Cancer 115(23):5481–5489. https://doi.org/10.1002/cncr.24610
Weisser M, Kern W, Rauhut S, Schoch C, Hiddemann W, Haferlach T, Schnittger S (2005) Prognostic impact of RT-PCR-based quantification of WT1 gene expression during MRD monitoring of acute myeloid leukemia. Leukemia 19(8):1416–1423. https://doi.org/10.1038/sj.leu.2403809
Trka J, Kalinova M, Hrusak O, Zuna J, Krejci O, Madzo J, Sedlacek P, Vavra V, Michalova K, Jarosova M, Stary J (2002) Real-time quantitative PCR detection of WT1 gene expression in children with AML: prognostic significance, correlation with disease status and residual disease detection by flow cytometry. Leukemia 16(7):1381–1389. https://doi.org/10.1038/sj.leu.2402512
van Dijk JP, Knops GH, van de Locht LTF, Menke AL, Jansen JH, Mensink EJBM, Raymakers RAP, de Witte T (2002) Abnormal WT1 expression in the CD34-negative compartment in myelodysplastic bone marrow. Br J Haematol 118:1027–1033
Mason EF, Kuo FC, Hasserjian RP, Seegmiller AC, Pozdnyakova O (2018) A distinct immunophenotype identifies a subset of NPM1-mutated AML with TET2 or IDH1/2 mutations and improved outcome. Am J Hematol 93(4):504–510. https://doi.org/10.1002/ajh.25018
Taussig DC, Vargaftig J, Miraki-Moud F, Griessinger E, Sharrock K, Luke T, Lillington D, Oakervee H, Cavenagh J, Agrawal SG, Lister TA, Gribben JG, Bonnet D (2010) Leukemia-initiating cells from some acute myeloid leukemia patients with mutated nucleophosmin reside in the CD34(-) fraction. Blood 115(10):1976–1984. https://doi.org/10.1182/blood-2009-02-206565
Papaemmanuil E, Gerstung M, Bullinger L, Gaidzik VI, Paschka P, Roberts ND, Potter NE, Heuser M, Thol F, Bolli N, Gundem G, Van Loo P, Martincorena I, Ganly P, Mudie L, McLaren S, O’Meara S, Raine K, Jones DR, Teague JW, Butler AP, Greaves MF, Ganser A, Dohner K, Schlenk RF, Dohner H, Campbell PJ (2016) Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med 374(23):2209–2221. https://doi.org/10.1056/NEJMoa1516192
Hosen N, Shirakata T, Nishida S, Yanagihara M, Tsuboi A, Kawakami M, Oji Y, Oka Y, Okabe M, Tan B, Sugiyama H, Weissman IL (2007) The Wilms’ tumor gene WT1-GFP knock-in mouse reveals the dynamic regulation of WT1 expression in normal and leukemic hematopoiesis. Leukemia 21(8):1783–1791. https://doi.org/10.1038/sj.leu.2404752
Bras AE, de Haas V, van Stigt A, Jongen-Lavrencic M, Beverloo HB, Te Marvelde JG, Zwaan CM, van Dongen JJM, Leusen JHW, van der Velden VHJ (2019) CD123 expression levels in 846 acute leukemia patients based on standardized immunophenotyping. Cytometry B Clin Cytom 96(2):134–142. https://doi.org/10.1002/cyto.b.21745
Acknowledgments
The authors thank Wei-Hua Shi, Lin-Lin He, Le Hao, and Jing Wu for their assistance.
Author information
Authors and Affiliations
Contributions
Xiao-Rui Wang and Yan-Rong Liu designed, wrote, and interpreted data. Yan Chang, Xiao-Ying Yuan, and Ya-Zhe Wang performed experiments and provided technical support. Ya-Zhen Qin, Guo-Rui Ruan, and Yue-Yun Lai performed experiments and analyzed data. All authors approved the manuscript. Yan-Rong Liu supervised the study.
Corresponding author
Ethics declarations
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (the Ethics Committee of Peking University People’s Hospital) and with the Helsinki Declaration of 1975, as revised in 2008. Since this is a retrospective study, formal informed consent is not required.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, XR., Chang, Y., Yuan, XY. et al. Overexpressed WT1 exhibits a specific immunophenotype in intermediate and poor cytogenetic risk acute myeloid leukemia. Ann Hematol 99, 215–221 (2020). https://doi.org/10.1007/s00277-019-03808-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-019-03808-6