Abstract
Purpose
An international survey was conducted by the Cardiovascular Interventional Radiological Society of Europe (CIRSE) to evaluate radioembolization practice and capture opinions on real-world clinical and technical aspects of this therapy.
Materials and Methods
A survey with 32 multiple choice questions was sent as an email to CIRSE members between November and December 2022. CIRSE group member and sister societies promoted the survey to their local members. The dataset was cleaned of duplicates and entries with missing data, and the resulting anonymized dataset was analysed. Data were presented using descriptive statistics.
Results
The survey was completed by 133 sites, from 30 countries, spanning 6 continents. Most responses were from European centres (87/133, 65%), followed by centres from the Americas (22/133, 17%). Responding sites had been performing radioembolization for 10 years on average and had completed a total of 20,140 procedures over the last 5 years. Hepatocellular carcinoma treatments constituted 56% of this total, colorectal liver metastasis 17% and cholangiocarcinoma 14%. New sites had opened every year for the past 20 years, indicating the high demand for this therapy. Results showed a trend towards individualized treatment, with 79% of responders reporting use of personalized dosimetry for treatment planning and 97% reporting routine assessment of microsphere distribution post-treatment. Interventional radiologists played an important role in referrals, being present in the referring multi-disciplinary team in 91% of responding centres.
Conclusion
This survey provides insight into the current state of radioembolization practice globally. The results reveal the increasing significance placed on dosimetry, evolving interventional techniques and increased technology integration.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Radioembolization is a complex, multi-disciplinary and multi-step intervention [1]. The treatment pathway typically begins with CT/MRI assessment [2], followed by tests to determine the clinical and biochemical status. Subsequently, a diagnostic hepatic angiography and scintigraphy using either technetium 99 m (99mTc) macroaggregated albumin (MAA) or 166Ho scout dose is conducted, for assessment of lung shunt and intra-/extrahepatic deposition. The therapeutic procedure then takes place in a second hepatic angiography where microspheres are administered. Lastly, post-treatment imaging may be conducted to verify distribution, derive quantitative dose estimates and assess tumour response [3].
At each of the outlined steps, a multitude of decisions (regarding imaging, intent, technique, etc.) must be made, and therefore, despite being a well-established treatment option, standardization in approach is lacking [4]. Addressing variability in the field is a key challenge, especially in the context of multi-centre collaborations and clinical trials. Several studies have already been conducted to assess practice variations in Europe [5,6,7]. This study will expand on these previous works by obtaining a worldwide perspective that encompasses a wide variety of radioembolization centres.
The objective of this international survey was to evaluate radioembolization practice around the world and to capture a snapshot of interventional radiologist’s opinions on technical and clinical aspects of radioembolization.
Material and Methods
Survey Design
A survey was jointly developed by our centre and the Cardiovascular Interventional Society of Europe (CIRSE), with specific contribution from CIRSE’s Contract Research Organization Next Research. The survey consisted of 32 multiple choice questions (Table 1) in the following categories: treatment workup, treatment planning and dosimetry, intervention, follow-up and innovations. Previous surveys conducted in 2012 [6] and 2018 [5] were used as a basis when constructing the questions. Themes from the previous two iterations were maintained in the current version to allow for trending of results. Existing questions were supplemented with new topics and concepts reflecting contemporary practice, which were derived following an extensive literature review.
Participant Recruitment
To reach an international audience and maximize participants, the survey was emailed to members of CIRSE and the Society of Interventional Radiology (SIR). CIRSE group member societies were asked to promote the survey amongst their local members. The survey was launched on the 3/11/22 and closed on the 24/12/22, and three reminder emails were sent in the interim period. Members received an email with a link to the website where the survey was hosted. Prospective responders were asked to consent to have their responses pooled, analysed and reported in a scientific publication. In the absence of consent, participants were disqualified from completing the survey.
Data Cleaning
To allow for data extraction, the database was cleaned of duplicates, entry errors were identified and entries with missing information were removed. In case of duplicate responses, where answers differed between participants from the same centre, the response which was deemed most complete (i.e. more in-depth responses in free text fields) was selected.
Data Analysis
Descriptive statistics were calculated for pooled responses across all sites and were used to put results into context. For multiple choice questions, where more than one response had been selected, all responses were included, which meant percentages could exceed 100%. Processing of the data was performed centrally using Tableau (ver2023.1).
Results
General Overview
A total of 502 responses were collected. After correcting for disqualified (7), partial (348) and duplicate (14) entries, 133 responses were used for further analysis.
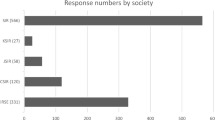
The 133 responding sites were distributed across 30 countries, spanning 6 continents (Fig. 1). The majority of responses were from European centres 65%, followed by sites from the Americas 17%, Asia 13%, Oceania 4% and Africa 2%.
Cumulatively, a total of 20,140 procedures had been completed over the last 5 years across all responding sites. The total number of procedures performed per year across all sites increased by approximately 50% from 2790 in 2017 to 4165 in 2022. The average number of procedures completed by each site per year increased from 21 (range 0–230) per year in 2017 to 31 (range 0–150) in 2022. The data also revealed a substantial increase in the number of high-volume centres, defined as those performing on average at least one radioembolization procedure per week. There was triple the number of sites performing 40–60 procedures per year in 2022 compared to 2017.
Of the 20,140 procedures completed from 2017 to 2022, hepatocellular carcinoma treatments constituted 56% of this total, colorectal liver metastasis 17% and cholangiocarcinoma 14%. Lesser treated indications included breast liver metastasis 5%, neuroendocrine liver metastasis 4% and other indications 4%. Resin yttrium-90 (90Y)-microspheres (SIR-Spheres®, Sirtex) were used for 51% of procedures, glass 90Y-microspheres (TheraSphere®, Boston Scientific Corporation) 43% and holmium-166 (166Ho)-microspheres (QuiremSpheres®, Terumo) 5% (Table 2).
Pre-treatment Workup
Interventional radiologists played an important role in referrals, being present in the referring multi-disciplinary team in 91% of responding centres. Prophylactic antiemetics (22%) and proton pump inhibitors (21%) were frequently prescribed before radioembolization, whereas opiates (7%) were rarely used (Fig. 2).
Image-based liver function assessment was performed by 63% of centres as part of the workup, with contrast-enhanced MRI using liver specific agents being the preferred method (26%).
Treatment Planning and Dosimetry
The imaging modality of choice for the pre-treatment scintigraphy or ‘workup’ procedure was SPECT/CT (68%) (Fig. 3). 79% of responders utilized the workup procedure for personalized dosimetry, and specifically, to determine whether tumour dose met a pre-defined dose threshold. 59% of responders indicated they would exclude a patient from treatment if the workup procedure indicated poor tumour MAA-targeting. 99mTc-MAA (i.e. the generally used scout agent prior to 90Y-microspheres) was considered to be reliable for intrahepatic dosimetry by a significant majority of responders (92% in favour vs. 8% against). The favoured calculation method used to determine injected activity for all radioembolization products was multi-compartment dosimetry using the MIRD (medical internal radiation dose) schema. Specifically, 51% of resin users, 52% of glass users and 61% of 166Ho users selected this method. Median lung shunt and lung dose values of 20.0% and 30.0 Gy, respectively, were considered a contraindication to treatment. A minority of centres (4.5%) did not consider lung shunting a contraindication.
The majority of responders (60%) used software to support dosimetry calculations, the most commonly used package was Simplicit90Y™ (Mirada Medical) (22%), followed by SurePlan™ MRT (MIM Software Inc) (13%). Routine coil embolization of non-target vessels was rare. Overall, the right gastric artery and gastroduodenal artery were the most frequently embolized vessels (Fig. 4).
Intervention
A large majority of responders utilized intra-procedural CT imaging (e.g. cone-beam CT or Angio-CT) (84%), and only a small number of users did not use it (9%) or lacked access to it (7%) (Fig. 5). Most centres reported using intra-procedural CT to confirm adequate tumour coverage (77%). The majority of users considered standard catheters (as opposed to antireflux or other catheters) sufficient for administration of microspheres (87%). A femoral access route was used in 75% of cases, radial was used in 14%, and a large proportion of sites (33%) used both femoral and radial access options.
For patients with multiple tumours and/or bilobar tumour manifestation, 65% of centres opted for a sequential left/right session with an interval, and only 4% performed a whole liver (bilobar) infusion in a single session via the proper hepatic artery (Supplemental Fig. 1).
Tailoring the number of injected microspheres for different clinical indications is a method that has gained prominence in recent years [8]. Results indicated that 61% of responders did consider the number of microspheres when planning treatments. Among those using glass microspheres, the ‘late week 1’ order option was favoured by the majority (35%). For resin microspheres, the ‘1-day pre-calibration’ order option was the preferred selection (39%) (supplemental Fig. 1).
Regarding the duration of patient stay (0,1 or ≥ 2 nights) following radioembolization, the average number of patients staying 0, 1 and ≥ 2 nights was 46, 61 and 44%, respectively.
Follow-Up
The majority of centres performed post-treatment imaging (97%) to visually evaluate whether the microsphere distribution was as planned, SPECT/CT was the most commonly used modality for this procedure (Fig. 6). A quantitative evaluation of post-treatment imaging to determine delivered dose was performed by 62% of responders. The reported incidence of complications was low, with REILD (radioembolization induced liver disease) (6%) and gastrointestinal complications (5%) the highest reported across all centres.
(Upper) The percentage of users that perform a quantitative evaluation of post-treatment imaging via assessment of absorbed dose. (Lower) The percentage usage of imaging modalities for post-treatment imaging. A The percentage usage of various administration techniques in cases of bilobar tumour manifestation. B The percentage usage of the calibration date options for 90Y glass and flexdose options for 90Y resin
Innovations
Centres showed most enthusiasm for improved dose calculation methods, real-time imaging feedback on the dose distribution and novel scout agents, with 79, 77 and 70% of responders indicating they either agreed or strongly agreed these potential developments could improve radioembolization practice, respectively (Table 3) (Supplemental Fig. 2).
Highlighted innovations that may improve radioembolization practice varied from possible synergistic effects with immuno-oncology agents, to MRI guided administration of microspheres and temporary embolization material to redirect blood flow.
Discussion
A comprehensive database on the real-life clinical application of radioembolization has been collated as part of this global CIRSE survey. The extent to which results accurately represent reality is dependent on the number of responders, how well the questions were understood [9], the propensity for centres involved in research to be more likely to respond, among other factors. Despite these limitations, the 133 responding centres included sites with various backgrounds and spanned the majority of geographical regions where radioembolization is conducted, giving these data a broad basis to represent the field as a whole.
Our data demonstrated an almost 50% increase in the number of patients treated with radioembolization over the last 5 years. This has persisted despite challenges associated with reimbursement [10, 11], the outcome of several negative trials [12,13,14,15,16,17] and the global COVID-19 pandemic [18,19,20]. European surveys in 2011 and 2018 reported data from 28 and 71 responding centres, respectively. The number of participating centres steadily increased and culminated in this work, for which 502 responses were received and 133 were analysed. Products from all three major microspheres vendors (Sirtex, Boston Scientific and Terumo) were considered. This marked a change from previous surveys that focussed solely on resin and glass microspheres. Resin remained the predominantly used microsphere.
Trends towards individualized treatment and personalized dosimetry that have been widely reported in the literature [18] were clearly evident in this survey. For example, an emerging technique gaining prominence is the trans-radial administration of microspheres, as an alternative to the standard trans-femoral route. This approach has been shown to decrease risk of entry site complications, improve post-procedural comfort levels [21, 22] and provide flexibility to treat patients with inaccessible groins/uncorrectable coagulopathy. A third of responders (33%) indicated that they now utilize both femoral and radial access routes in their centre. In addition, there was a trend towards more selective administrations to tumour-bearing segments of the liver [23, 24]. Results demonstrated reduced prophylactic coil embolization rates, which reflects the increased drive for more selective treatments.
Many of the highlighted procedural innovations are underpinned by technological developments. The precise visualization of vasculature [25] offered by intra-procedural CT imaging (e.g. cone-beam CT or Angio-CT) enables accurate detection of tumour-feeding vessels and precise targeting of lesions, facilitating the super-selective microsphere administration techniques that are increasingly observed. Results indicated high percentage usage of this modality and growth over the last 5 years; however, there were a minority (9%) of responders who indicated they have access to c-arm CT but do not use it. This potentially reveals a role for educational initiatives, to emphasize the impact of this technology on patient outcomes and the necessity for its incorporation by all centres. The increased sensitivity and specificity of 99mTc-MAA SPECT/CT in the detection of extrahepatic arterial shunting [26] compared to conventional planar imaging have resulted in a shift towards this modality for the pre-treatment workup. The present results indicated that more than two-thirds of responders utilize SPECT/CT to evaluate the scintigraphy workup procedure.
While some centres still utilized semi-empirical dosimetry planning methods for radioembolization, these represented a minority (e.g. BSA dosimetry usage was < 15% for all microsphere products), and over the past five years there has been a shift in the community towards MIRD dosimetry methods that have been used in several landmark trials [27,28,29]. This survey demonstrated that MIRD dosimetry methods were unanimously favoured across all products, which is a change from a previous (2018) European survey [5] that indicated the partition model was "rarely" used. Consequently, it seems reasonable to assume that activity planning is now evolving along two paths: MIRD single and MIRD multi-compartment modelling. In general, dosimetry has taken on greater significance and now constitutes a central practice in the treatment workup. Our results indicated that responders frequently use dosimetric data, including projected tumour and liver doses to assist clinical decision-making.
The increasing evidence supporting personalized treatment planning [27, 30,31,32] has resulted in a rise in the use of dosimetry software packages. The widespread adoption of these packages will help to standardize dosimetry planning methods and improve consistency. Comparable performance that allows for reliable comparison of quantitative dose metrics between centres is essential, and harmonization strategies are the subject of ongoing research [33, 34].
A clear consensus among centres was the low incidence of complications, with average reported incidences ranging from 0 to 6%. Gastrointestinal complications and REILD were most commonly encountered, which is in line with previously reported results [5]. It should be noted that there is no clear consensus on definition of REILD and therefore responders may have interpreted what constitutes REILD differently. An example response in this survey called for ‘better predictive capabilities of who may develop REILD, as lab values can give a false impression of a normal functioning liver’. This may explain why an increasing number of centres are integrating image-based liver function tests into the pre-treatment workup, results indicating that 63% of centres now perform these tests as standard.
For this survey, there were challenges in reaching responders resulting in an irregular response rate between countries. The uneven global distribution of responses can be attributed to the lead role of CIRSE, with sister organizations being comparatively less involved. Efforts to enhance global participation should be considered for future surveys. A challenge of conducting a qualitative survey is the reliance on self-reported data; the absence of validation introduces the potential for reporting bias. Additionally, a substantial volume of data was collected and only a curated subset may be presented in this manuscript. Consequently, greater emphasis should be placed on discerning trends rather than exact numerical estimates.
This survey highlighted several prospective advancements in radioembolization which may improve practice. Notably, adaptive planning, involving intraoperative re-optimization, garnered widespread approval. Current developments including radio-opaque microspheres [35] and a novel imaging modality IXSI [36], could facilitate real-time feedback on the dose distribution, which is necessary to make adaptive planning a reality. Respondents also underscored the treatment optimization potential of personalized dosimetry (79% of responders agreed this would improve practice), suggesting a probable expansion of personalized dosimetry adoption.
Conclusion
Radioembolization is a rapidly growing and evolving treatment modality; the scale of growth warrants consideration as to how standardization in practice may be achieved. This survey has revealed the increasing significance placed on dosimetry, evolving interventional techniques and increased technology integration. The findings underscore the value of sharing knowledge and best practice.
Availability of Data and Materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CT :
-
Computed tomography
- MRI :
-
Magnetic resonance imaging
- 99m Tc -MAA :
-
Technetium-99 macroaggregated albumin
- 166Ho :
-
Holmium-166
- CIRSE :
-
Cardiovascular Interventional Radiological Society of Europe
- SIR :
-
Society of Interventional Radiology
- 90Y :
-
Yttrium-90
- MIRD :
-
Medical Internal Radiation Dose
- REILD :
-
Radioembolization induced liver disease
References
Gregory J, Tselikas L, Allimant C, de Baere T, Bargellini I, Bell J, Bilbao JI, Bouvier A, Chapiro J, Chiesa C, Decaens T, Denys A, Duran R, Edeline J, Garin E, Ghelfi J, Helmberger T, Irani F, Lam M, Lewandowski R, Liu D, Loffroy R, Madoff DC, Mastier C, Salem R, Sangro B, Sze D, Vilgrain V, Vouche M, Guiu B, Ronot M. Defining textbook outcome for selective internal radiation therapy of hepatocellular carcinoma: an international expert study. Eur J Nucl Med Mol Imaging. 2023;50(3):921–8. https://doi.org/10.1007/s00259-022-06002-5.
Braat AJAT, Smits MLJ, Braat MNGJA, van den Hoven AF, Prince JF, de Jong HWAM, van den Bosch MAAJ, Lam MGEH. 90Y Hepatic radioembolization: an update on current practice and recent developments. J Nucl Med. 2015;56(7):1079–87. https://doi.org/10.2967/jnumed.115.157446.
Salem R, Padia SA, Lam M, Bell J, Chiesa C, Fowers K, et al. Clinical and dosimetric considerations for Y90: recommendations from an international multidisciplinary working group. Eur J Nucl Med Mol Imaging. 2019;46:1695–704. https://doi.org/10.1007/s00259-019-04340-5.
Keane G, de Jong HWAM, Lam MGEH. Beyond the MAA-Y90 paradigm: the evolution of radioembolization dosimetry approaches and scout particles. Semin Interv Radiol. 2021;38(5):542–53. https://doi.org/10.1055/s-0041-1736660.
Reinders MTM, Mees E, Powerski MJ, Bruijnen RCG, van den Bosch MAAJ, Lam MGEH, Smits MLJ. Radioembolisation in Europe: a survey amongst CIRSE members. Cardiovasc Intervent Radiol. 2018;41(10):1579–89. https://doi.org/10.1007/s00270-018-1982-4.
Powerski MJ, Scheurig-Munkler C, Banzer J, Schnapauff D, Hamm B, Gebauer B. Clinical practice in radioembolization of hepatic malignancies: a survey among interventional centres in Europe. Eur J Radiol. 2012;81(7):e804–11. https://doi.org/10.1016/j.ejrad.2012.04.004.
Helmberger T, Golfieri R, Pech M, Pfammatter T, Arnold D, Cianni R, Maleux G, Munneke G, Pellerin O, Peynircioglu B, Sangro B, Schaefer N, de Jong N, Bilbao JI. Clinical application of trans-arterial radioembolization in hepatic malignancies in Europe: first results from the prospective multicentre observational study CIRSE registry for SIR-spheres therapy (CIRT). Cardiovasc Intervent Radiol. 2021;44(1):21–35. https://doi.org/10.1007/s00270-020-02642-y.
Kim HC, Lee M, Lee JH, Paeng JC, Kim YJ, Chung JW. Combination of 1st and 2nd week dosing of glass yttrium-90 microspheres for superselective radioembolization. in Vivo. 2020;34(5):2763–8. https://doi.org/10.21873/invivo.12100.
Sjögreen Gleisner K, Spezi E, Solny P, et al. Variations in the practice of molecular radiotherapy and implementation of dosimetry: results from a European survey. EJNMMI Phys. 2017;4:28. https://doi.org/10.1186/s40658-017-0193-4.
Rojas B, Hooker C, McGowan DR, Guy MJ, Tipping J. Eight years of growth and change in UK molecular radiotherapy with implications for the future: internal dosimetry users group survey results from 2007 to 2015. Nucl Med Commun. 2017;38(3):201–4. https://doi.org/10.1097/MNM.0000000000000642.
Alonso JC, Casans I, González FM, Fuster D, Rodríguez A, Sánchez N, Oyagüez I, Burgos R, Williams AO, Espinoza N. Economic evaluations of radioembolization with Itrium-90 microspheres in hepatocellular carcinoma: a systematic review. BMC Gastroenterol. 2022;22:326. https://doi.org/10.1186/s12876-022-02396-6.
Vilgrain V, Pereira H, Assenat E, Guiu B, Ilonca AD, Pageaux GP, et al. Efficacy and safety of selective internal radiotherapy with yttrium-90 resin microspheres compared with sorafenib in locally advanced and inoperable hepatocellular carcinoma (SARAH): an open-label randomised controlled phase 3 trial. Lancet Oncol. 2017;18(12):1624–36. https://doi.org/10.1016/S1470-2045(17)30683-6.
Chow PKH, Gandhi M, Tan SB, Khin MW, Khasbazar A, Ong J, et al. SIRveNIB: Selective internal radiation therapy versus sorafenib in Asia-Pacific patients with hepatocellular carcinoma. J Clin Oncol. 2018;36(19):1913–21. https://doi.org/10.1200/JCO.2017.76.0892.
Ricke J, Klümpen HJ, Amthauer H, Bargellini I, Bartenstein P, de Toni EN, Gasbarrini A, Pech M, Peck-Radosavljevic M, Popovič P, Rosmorduc O, Schott E, Seidensticker M, Verslype C, Sangro B, Malfertheiner P. Impact of combined selective internal radiation therapy and sorafenib on survival in advanced hepatocellular carcinoma. J Hepatol. 2019;71(6):1164–74. https://doi.org/10.1016/j.jhep.2019.08.006.
van Hazel GA, Heinemann V, Sharma NK, Findlay MP, Ricke J, Peeters M, et al. SIRFLOX: randomized phase iii trial comparing first-line mfolfox6 (plus or minus bevacizumab) versus mfolfox6 (plus or minus bevacizumab) plus selective internal radiation therapy in patients with metastatic colorectal cancer. J Clin Oncol. 2016;34(15):1723–31. https://doi.org/10.1200/JCO.2015.66.1181.
Dutton SJ, Kenealy N, Love SB, Wasan HS, Sharma RA. FOXFIRE protocol: an open-label, randomised, phase III trial of 5-fluorouracil, oxaliplatin and folinic acid (OxMdG) with or without interventional Selective Internal Radiation Therapy (SIRT) as first-line treatment for patients with unresectable liver-only or liver-dominant metastatic colorectal cancer. BMC Cancer. 2014;14:497. https://doi.org/10.1186/1471-2407-14-497.
Hermann AL, Dieudonné A, Ronot M, Sanchez M, Pereira H, Chatellier G, Garin E, Castera L, Lebtahi R, Vilgrain V. Relationship of tumor radiation-absorbed dose to survival and response in hepatocellular carcinoma treated with transarterial radioembolization with 90Y in the SARAH study. Radiology. 2020;296(3):673–84. https://doi.org/10.1148/radiol.2020191606.
de Baere T, Crocetti L. Radioembolization: same player shoots again. Cardiovasc Intervent Radiol. 2022;45:1577–8. https://doi.org/10.1007/s00270-022-03269-x.
Rojas B, McGowan DR, Gear J, Smith AL, Scott C, Craig AJ, Scuffham J, Towey D, Aldridge M, Tipping J. Nearly double the patients and dramatic changes over 14 years of UK MRT: Internal Dosimetry Users Group survey results from 2007 to 2021. Nucl Med Commun. 2023. https://doi.org/10.1097/MNM.0000000000001780.
Cutler CS, Bailey E, Kumar V, Schwarz SW, Bom HS, Hatazawa J, Paez D, Orellana P, Louw L, Mut F, Kato H, Chiti A, Frangos S, Fahey F, Dillehay G, Oh SJ, Lee DS, Lee ST, Nunez-Miller R, Bandhopadhyaya G, Pradhan PK, Scott AM. Global issues of radiopharmaceutical access and availability: a nuclear medicine global initiative project. J Nucl Med. 2021;62(3):422–30. https://doi.org/10.2967/jnumed.120.247197.
Ghosh A, Zhang J, Akhter NM. Transradial versus transfemoral arterial access in Yttrium-90 microspheres radioembolization for hepatocellular carcinoma. J Clin Imaging Sci. 2022;12(12):27. https://doi.org/10.25259/JCIS_213_2021.
Liu LB, Cedillo MA, Bishay V, Ranade M, Patel RS, Kim E, Nowakowski SF, Lookstein RA, Fischman AM. Patient experience and preference in transradial versus transfemoral access during transarterial radioembolization: a randomized single-center trial. J Vasc Interv Radiol. 2019;30(3):414–20. https://doi.org/10.1016/j.jvir.2018.10.005.
Salem R, Johnson GE, Kim E, Riaz A, Bishay V, Boucher E, Fowers K, Lewandowski R, Padia SA. Yttrium-90 Radioembolization for the Treatment of Solitary, unrespectable HCC: the LEGACY study. Hepatology. 2021;74(5):2342–52. https://doi.org/10.1002/hep.31819.
Kim E, Sher A, Abboud G, Schwartz M, Facciuto M, Tabrizian P, Knešaurek K, Fischman A, Patel R, Nowakowski S, Llovet J, Taouli B, Lookstein R. Radiation segmentectomy for curative intent of unresectable very early to early stage hepatocellular carcinoma (RASER): a single-centre, single-arm study. Lancet Gastroenterol Hepatol. 2022;7(9):843–50. https://doi.org/10.1016/S2468-1253(22)00091-7.
Lionberg A, Nijhawan K, Navuluri R, Zangan S, Van Ha T, Funaki B, Ahmed O. Hybrid angiography-CT for transarterial radioembolization: a pictorial essay. Abdominal Radiol (NY). 2021;46(6):2850–4. https://doi.org/10.1007/s00261-020-02914-8.
Hamami ME, Poeppel TD, Müller S, Heusner T, Bockisch A, Hilgard P, Antoch G. SPECT/CT with 99mTc-MAA in radioembolization with 90Y microspheres in patients with hepatocellular cancer. J Nucl Med. 2009;50(5):688–92. https://doi.org/10.2967/jnumed.108.058347.
Garin E, Tselikas L, Guiu B, Chalaye J, Edeline J, de Baere T, et al. Personalised versus standard dosimetry approach of selective internal radiation in patients with locally advanced hepatocellular carcinoma (DOSISPHERE-01): a randomised, multicentre, open-label phase 2 trial. Lancet Gastroenterol Hepatol. 2021;6(1):17–29. https://doi.org/10.1016/S2468-1253(20)-9.
van Roekel C, Bastiaannet R, Smits MLJ, Bruijnen RC, Braat AJAT, de Jong HWAM, Elias SG, Lam MGEH. Dose-effect relationships of 166Ho radioembolization in colorectal Cancer. J Nucl Med. 2021;62(2):272–9. https://doi.org/10.2967/jnumed.120.243832.
Mahvash A, Chartier S, Turco M, et al. A prospective, multicenter, open-label, single-arm clinical trial design to evaluate the safety and efficacy of 90Y resin microspheres for the treatment of unresectable HCC: the DOORwaY90 (Duration Of Objective Response with arterial Ytrrium-90) study. BMC Gastroenterol. 2022;22:151. https://doi.org/10.1186/s12876-022-02204-1.
Ilhan H, Goritschan A, Paprottka P, et al. Predictive value of 99mTc-MAA SPECT for 90Y-labeled resin microsphere distribution in radioembolization of primary and secondary hepatic tumors. J Nucl Med. 2015;56(11):1654–60.
Smits MLJ, Dassen MG, Prince JF, et al. The superior predictive value of 166Ho-scout compared with 99mTc-macroaggregated albumin prior to 166Ho-microspheres radioembolization in patients with liver metastases. Eur J Nucl Med Mol Imaging. 2020;47(04):798–806.
Smits ML, Elschot M, van den Bosch MA, et al. In vivo dosimetry based on SPECT and MR imaging of 166Ho-microspheres for treatment of liver malignancies. J Nucl Med. 2013;54(12):2093–100.
AAPM. Standardizing Imaging and Reconstruction Protocols for Quantitative SPECT/CT Post Yttrium-90 Microspheres Delivery (SIRP. [Online]. American Association of Physicists in Medicine. 2022; Available at: https://www.aapm.org/GrandChallenge/SIRPRISE/ [Accessed 1 May 2023].
Lam M, Garin E, Maccauro M, Kappadath SC, Sze DY, Turkmen C, et al. A global evaluation of advanced dosimetry in transarterial radioembolization of hepatocellular carcinoma with Yttrium-90: the TARGET study. Eur J Nucl Med Mol Imaging. 2022. https://doi.org/10.1007/s00259-022-05774-0.
Abraham R, Lewandowski R, Gandhi R, et al. What’s New in Y-90? Technical and procedural innovations in Y-90 radioembolization including existing and investigational next-generation technologies. Interv Oncol. 2019;18(10):49–60.
van der Velden S, Kunnen B, Koppert WJC, Steenbergen JHL, Dietze MMA, Beijst C, Viergever MA, Lam MGEH, de Jong HWAM. A dual-layer detector for simultaneous fluoroscopic and nuclear imaging. Radiology. 2019;290(3):833–8. https://doi.org/10.1148/radiol.2018180796.
Acknowledgements
We would like to thank all participating interventional radiologists who took part in this survey. We are very grateful to the Cardiovascular Interventional Radiology Society of Europe (CIRSE) for facilitating the distribution of the survey and in particular Next Research.
Funding
The study was funded by research support from Boston Scientific.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Grace Keane is a consultant for Boston Scientific. She is paid via a research collaboration with Boston Scientific. Marnix Lam, MD, PhD, is a consultant for Boston Scientific, Terumo and Quirem Medical. He receives research support from Boston Scientific, Terumo and Quirem Medical. Arthur Braat, MD, PhD, is a consultant for Boston Scientific, Terumo, and receives research support from Ariceum Therapeutics. Nathalie Kaufmann works for Next Research. Hugo de Jong, PhD, is a consultant for Boston Scientific and Quirem Medical. Maarten Smits, MD, PhD, is a consultant for Philips, Terumo and Quirem Medical. He is a speaker for Medtronic. The UMC Utrecht receives royalties from Quirem Medical.
Ethics approval
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Keane, G., Lam, M., Braat, A. et al. Transarterial Radioembolization (TARE) Global Practice Patterns: An International Survey by the Cardiovascular and Interventional Radiology Society of Europe (CIRSE). Cardiovasc Intervent Radiol (2024). https://doi.org/10.1007/s00270-024-03768-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00270-024-03768-z