Abstract
Purpose
To evaluate the Sclerograft™ procedure, which is an image-guided, minimally invasive approach of chemical sclerotherapy followed by bone grafting of unicameral bone cysts (UBC).
Materials and Methods
A retrospective evaluation from August 2018 through August 2023 was performed at a single institution on patients that underwent the Sclerograft™ procedure for UBCs. Radiographic healing was evaluated utilizing the Modified Neer Classification. Two different regenerative grafts, CaSO4–CaPO4 and HA-CaSO4 were utilized. A total of 50 patients were evaluated with 41 patients grafted with CaSO4–CaPO4 and 9 patients grafted with HA-CaSO4.
Results
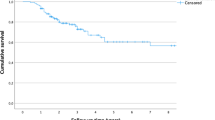
The average age of the patient was 12.1 years with an average radiographic follow-up of 14.5 months. Average cyst size was 5.5 cm in the largest dimension and average cyst volume was 20.2 cc. 42 out of 50 (84%) showed healed cysts (Modified Neer Class 1) on the most recent radiograph or MRI. Recurrences occurred on average at 7.2 months. Activity restrictions were lifted at 3–4.5 months post-procedure. Cyst stratification by size did not show a difference in recurrence rates (p = 0.707). There was no significant difference in recurrence rate between lesions abutting the physis compared to those that were not abutting the physis (p = 0.643). There were no major complications.
Conclusions
The Sclerograft™ procedure is an image-guided approach to treating unicameral bone cysts, utilizing chemical sclerosis and regenerative bone grafting. The radiographic healing of cysts compares favorably to open curettage and grafting as determined utilizing previously published trials.
Graphical Abstract




Similar content being viewed by others
References
Kadhim M, Thacker M, Kadhim A, Holmes L Jr. Treatment of unicameral bone cyst: systematic review and meta analysis. J Child Orthop. 2014;8:171–91. https://doi.org/10.1007/s11832-014-0566-3.
Pretell-Mazzini J, Murphy RF, Kushare I, et al. Unicameral bone cysts: general characteristics and management controversies. J Am Acad Orthop Surg. 2014;22:295–303. https://doi.org/10.5435/JAAOS-22-05-295.
Mascard E, Gomez-Brouchet A, Lambot K. Bone cysts: unicameral and aneurysmal bone cyst. Orthop Traumatol Surg Res. 2015;101(1 Suppl):S119–27.
Weinman J, Servaes S, Anupindi SA. Treated unicameral bone cysts. Clin Radiol. 2013;68(6):636–42. https://doi.org/10.1016/j.crad.2012.11.020.
Chigira M, Maehara S, Arita S, Udagawa E. The aetiology and treatment of simple bone cysts. J Bone Joint Surg Br. 1983;65(5):633–7. https://doi.org/10.1302/0301-620X.65B5.6643570.
Noordin S, Allana S, Umer M, Jamil M, Hilal K, Uddin N. Unicameral bone cysts: Current concepts. Ann Med Surg (Lond). 2018;34:43–9. https://doi.org/10.1016/j.amsu.2018.06.005.
Zhao JG, Wang J, Huang WJ, Zhang P, Ding N, Shang J. Interventions for treating simple bone cysts in the long bones of children. Cochrane Database Syst Rev. 2017. https://doi.org/10.1002/14651858.CD010847.pub3.
Farr S, Balacó IMS, Martínez-Alvarez S, Hahne J, Bae DS. Current trends and variations in the treatment of unicameral bone cysts of the humerus: a Survey of EPOS and POSNA members. J Pediatr Orthop. 2020;40(1):e68–76. https://doi.org/10.1097/BPO.0000000000001376.
Bukva B, Vrgoč G, Abramović D, Dučić S, Brkić I, Čengić T. Treatment of unicameral bone cysts in children: a comparative study. Acta Clin Croat. 2019;58(3):403–9. https://doi.org/10.20471/acc.2019.58.03.01.
Sung AD, Anderson ME, Zurakowski D, Hornicek FJ, Gebhardt MC. Unicameral bone cyst: a retrospective study of three surgical treatments. Clin Orthop Relat Res. 2008;466(10):2519–26. https://doi.org/10.1007/s11999-008-0407-0.
Hou HY, Wu K, Wang CT, Chang SM, Lin WH, Yang RS. Treatment of unicameral bone cyst: a comparative study of selected techniques. J Bone Joint Surg Am. 2010;92(4):855–62. https://doi.org/10.2106/JBJS.I.00607.
Canavese F, Wright JG, Cole WG, Hopyan S. Unicameral bone cysts: comparison of percutaneous curettage, steroid, and autologous bone marrow injections. J Pediatr Orthop. 2011;31(1):50–5. https://doi.org/10.1097/BPO.0b013e3181ff7510.
Shiels WE 2nd, Mayerson JL. Percutaneous doxycycline treatment of aneurysmal bone cysts with low recurrence rate: a preliminary report. Clin Orthop Relat Res. 2013;471(8):2675–83. https://doi.org/10.1007/s11999-013-3043-2.
Rajeswaran S, Khan A, Samet JD, Donaldson J, Attar S, Peabody T, Green J. Minimally invasive treatment for unicameral bone cysts with chemical sclerosis and bone graft substitute: a preliminary report. Cardiovasc Intervent Radiol. 2022;45:190–6. https://doi.org/10.1007/s00270-021-02945-8.
Neer CS II, Francis KC, Marcove RC, Terz J, Carbonara PN. Treatment of unicameral bone cyst. a follow-up study of one hundred seventy-five cases. J Bone Joint Surg Am. 1966;48:731–45.
Liu Q, He H, Zeng H, et al. Active unicameral bone cysts: control firstly, cure secondly. J Orthop Surg Res. 2019;1418(1):275. https://doi.org/10.1186/s13018-019-1326-3.
Gentile JV, Weinert CRS, JA. Treatment of unicameral bone cysts in pediatric patients with an injectable regenerative graft. J Pediatr Orthop. 2013;33(3):254–61. https://doi.org/10.1097/BPO.0b013e318285c56c.
Funding
This study was not supported by any funding.
Author information
Authors and Affiliations
Contributions
SM: Boston Scientific Consultant, Siemens Corp research funding, SR: Owner of trademark Sclerograft™ and has not received any financial compensation for this trademark, AR: Boston Scientific consultant.
Corresponding author
Ethics declarations
Conflict of interest
The authors do not have any conflicts of interest related to the submission of this manuscript.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. This study was approved by the Institutional Review Board at Lurie Children’s Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rajeswaran, S., Wiese, M., Baker, J. et al. Treatment of Unicameral Bone Cysts Utilizing the Sclerograft™ Technique. Cardiovasc Intervent Radiol 47, 346–353 (2024). https://doi.org/10.1007/s00270-024-03671-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-024-03671-7