Abstract
Background
The association between iodine levels and the risk of papillary thyroid cancer (PTC) has been suggested, but not definitively established. This study is to compare the iodine status of a group of patients with PTC (with and without BRAF V600E) with that of a healthy population cohort.
Methods
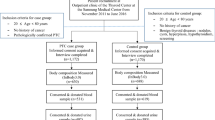
A cohort of patients scheduled for thyroidectomy was enrolled, along with a community-based health-screening cohort with no known history of thyroid disease. Median urinary iodine (UI) levels, creatinine-adjusted median UI levels, and food frequency questionnaire (FFQ) scores (mean ± SD) were compared. In a subgroup analysis, these values were compared between BRAF V600E-positive and BRAF V600E-negative patients in the PTC group.
Results
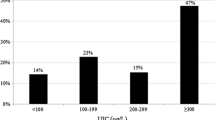
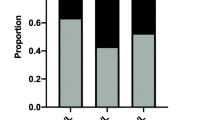
The PTC group consisted of 210 patients, and the control group consisted of 90 healthy individuals. Among the 191 PTC patients whose BRAF V600E mutational status was reported, 169 (88.5%) were revealed positive for the mutation. The median UI levels were significantly higher in the PTC group (786.0 μg/l) than the control group (112.0 μg/l; p < 0.001), as was the case with creatinine-adjusted median UI levels (884.6 μg/g creatinine versus 182.0 μg/g creatinine; p < 0.001) and FFQ scores (66.2 ± 17.5, range 13–114 versus 54.6 ± 21.5, range 16–134; p < 0.001). No significant differences were seen in the subgroup analysis between BRAF V600E-positive and BRAF V600E-negative patients.
Conclusions
Our results indicate that iodine status differs significantly between patients with PTC and healthy controls, suggesting that iodine may be involved in the occurrence of PTC, although the association between iodine levels and BRAF mutational status did not reach statistical significance.

Similar content being viewed by others
References
Pellegriti G, Frasca F, Regalbuto C et al (2013) Worldwide increasing incidence of thyroid cancer: update on epidemiology and risk factors. J Cancer Epidemiol 2013:965212
Ahn HS, Kim HJ, Welch HG (2014) Korea’s thyroid-cancer “epidemic”—screening and overdiagnosis. N Engl J Med 371:1765–1767
Choi YM, Kim TY, Jang EK et al (2014) Standardized thyroid cancer mortality in Korea between 1985 and 2010. Endocrinol Metab (Seoul) 29:530–535
Williams D (2008) Radiation carcinogenesis: lessons from Chernobyl. Oncogene 27(Suppl 2):S9–18
Pearce MS, Salotti JA, Little MP et al (2012) Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: a retrospective cohort study. Lancet 380:499–505
Mazonakis M, Tzedakis A, Damilakis J et al (2007) Thyroid dose from common head and neck CT examinations in children: is there an excess risk for thyroid cancer induction? Eur Radiol 17:1352–1357
Ron E, Lubin JH, Shore RE et al (1995) Thyroid cancer after exposure to external radiation: a pooled analysis of seven studies. Radiat Res 141:259–277
Richardson DB (2009) Exposure to ionizing radiation in adulthood and thyroid cancer incidence. Epidemiology 20:181–187
Pellegriti G, De Vathaire F, Scollo C et al (2009) Papillary thyroid cancer incidence in the volcanic area of Sicily. J Natl Cancer Inst 101:1575–1583
Feldt-Rasmussen U (2001) Iodine and cancer. Thyroid 11:483–486
Dijkstra B, Prichard RS, Lee A et al (2007) Changing patterns of thyroid carcinoma. Ir J Med Sci 176:87–90
Harach HR, Escalante DA, Onativia A et al (1985) Thyroid carcinoma and thyroiditis in an endemic goitre region before and after iodine prophylaxis. Acta Endocrinol (Copenh) 108:55–60
Hwang Y, Lee KE, Park YJ et al (2016) Annual average changes in adult obesity as a risk factor for papillary thyroid cancer: a large-scale case–control study. Medicine (Baltimore) 95:e2893
WHO/UNICEF/ICCIDD (2007) Assessment of iodine deficiency disorders and monitoring their elimination: a guide for programme managers. World Health Organization: http://whqlibdoc.who.int/publications/2007/9789241595827_eng.pdf. Accessed 9789241595820 August 9789241592013)
Guan H, Ji M, Bao R et al (2009) Association of high iodine intake with the T1799A BRAF mutation in papillary thyroid cancer. J Clin Endocrinol Metab 94:1612–1617
Wang F, Wang Y, Wang L et al (2014) Strong association of high urinary iodine with thyroid nodule and papillary thyroid cancer. Tumour Biol 35:11375–11379
Choi J, Kim HS, Hong DJ et al (2012) Urinary iodine and sodium status of urban Korean subjects: a pilot study. Clin Biochem 45:596–598
Hong AR, Lim JA, Kim TH et al (2014) The frequency and clinical implications of the BRAF(V600E) mutation in papillary thyroid cancer patients in Korea over the past two decades. Endocrinol Metab (Seoul) 29:505–513
Viertler C, Groelz D, Gundisch S et al (2012) A new technology for stabilization of biomolecules in tissues for combined histological and molecular analyses. J Mol Diagn JMD 14:458–466
WHO (2013) Urinary iodine concentrations for determining iodine status in populations. World Health Organizations, Geneva
Zimmermann MB, Andersson M (2012) Assessment of iodine nutrition in populations: past, present, and future. Nutr Rev 70:553–570
Kim HK, Lee SY, Lee JI et al (2010) Usefulness of iodine/creatinine ratio from spot-urine samples to evaluate the effectiveness of low-iodine diet preparation for radioiodine therapy. Clin Endocrinol 73:114–118
Lee KE, Park YJ, Cho B et al (2015) Protocol of a thyroid cancer longitudinal study (T-CALOS): a prospective, clinical and epidemiological study in Korea. BMJ Open 5:e007234
Moon SJ, Kim JY, Chung YJ et al (1998) The iodine content in common Korean foods. Korean Nutr Soc 31:7
Mood AM (1954) On the asymptotic efficiency of certain nonparametric two-sample tests. Ann Math Stat 25:8
Laurberg P, Bulow Pedersen I, Knudsen N et al (2001) Environmental iodine intake affects the type of nonmalignant thyroid disease. Thyroid 11:457–469
Todd CH, Allain T, Gomo ZA et al (1995) Increase in thyrotoxicosis associated with iodine supplements in Zimbabwe. Lancet 346:1563–1564
Stanbury JB, Ermans AE, Bourdoux P et al (1998) Iodine-induced hyperthyroidism: occurrence and epidemiology. Thyroid 8:83–100
Michikawa T, Inoue M, Shimazu T et al (2012) Seaweed consumption and the risk of thyroid cancer in women: the Japan Public Health Center-based prospective study. Eur J Cancer Prev 21:254–260
Pagano L, Caputo M, Sama MT et al (2012) Clinical-pathological changes in differentiated thyroid cancer (DTC) over time (1997–2010): data from the University Hospital “Maggiore della Carita” in Novara. Endocrine 42:382–390
Kim JY, Kim KR (2000) Dietary iodine intake and urinary iodine excretion in patients with thyroid diseases. Yonsei Med J 41:22–28
Kim H, Lee H, Park K et al (1985) A study on the urinary iodide excretion in normal subjects and patients with thyroid disease. Korean J Intern Med 29:625–631
Kim JY, Moon SJ, Kim KR et al (1998) Dietary iodine intake and urinary iodine excretion in normal Korean adults. Yonsei Med J 39:355–362
Murray CW, Egan SK, Kim H et al (2008) US Food and Drug Administration’s Total Diet Study: dietary intake of perchlorate and iodine. J Expo Sci Environ Epidemiol 18:571–580
Scientific Committee on Food (2002) Opinion of the scientific committee on food on the tolerable upper intake level of iodine, European Commission. Health & Consumer Protection Directorate-General, Brussels
Expert Group on Vitamins and Minerals (2003) Safe upper levels for vitamins and minerals. Food Standards Agency, London
Sehestedt T, Knudsen N, Perrild H et al (2006) Iodine intake and incidence of thyroid cancer in Denmark. Clin Endocrinol 65:229–233
Xiang J, Wang X, Wang Z et al (2015) Effect of different iodine concentrations on well-differentiated thyroid cancer cell behavior and its inner mechanism. Cell Biochem Biophys 71:299–305
Durante C, Puxeddu E, Ferretti E et al (2007) BRAF mutations in papillary thyroid carcinomas inhibit genes involved in iodine metabolism. J Clin Endocrinol Metab 92:2840–2843
Fuziwara CS, Kimura ET (2014) High iodine blocks a Notch/miR-19 loop activated by the BRAF(V600E) oncoprotein and restores the response to TGFbeta in thyroid follicular cells. Thyroid 24:453–462
Song YS, Lim JA, Park YJ (2015) Mutation profile of well-differentiated thyroid cancer in asians. Endocrinol Metab (Seoul) 30:252–262
Andersen S, Karmisholt J, Pedersen KM et al (2008) Reliability of studies of iodine intake and recommendations for number of samples in groups and in individuals. Br J Nutr 99:813–818
Lim CY, Kim JY, Yoon MJ et al (2015) Effect of a low iodine diet vs. restricted iodine diet on postsurgical preparation for radioiodine ablation therapy in thyroid carcinoma patients. Yonsei Med J 56:1021–1027
Cho YA, Kim J (2015) Dietary factors affecting thyroid cancer risk: a meta-analysis. Nutr Cancer 67:811–817
Bosetti C, Kolonel L, Negri E et al (2001) A pooled analysis of case–control studies of thyroid cancer. VI. Fish and shellfish consumption. Cancer Causes Control CCC 12:375–382
Liu ZT, Lin AH (2014) Dietary factors and thyroid cancer risk: a meta-analysis of observational studies. Nutr Cancer 66:1165–1178
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors involved in this study have actual or potential competing conflict of interests to declare.
Rights and permissions
About this article
Cite this article
Lee, JH., Song, RY., Yi, J.W. et al. Case–Control Study of Papillary Thyroid Carcinoma on Urinary and Dietary Iodine Status in South Korea. World J Surg 42, 1424–1431 (2018). https://doi.org/10.1007/s00268-017-4287-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-017-4287-x