Abstract
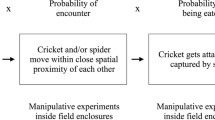
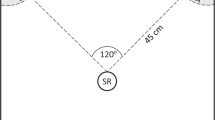
Female mate choice is remarkably complex and has wide-ranging implications for the strength and direction of male trait evolution. Yet mating decisions can be fickle and inconsistent. Here, we explored predation risk as a source of variation in the effort a female is willing to invest in acquiring a preferred mate type (“choosiness”). We did so by comparing phonotaxis behaviors of female eastern gray treefrogs (Hyla versicolor) across trials with and without simulated predators. We tested the behavioral adjustment hypothesis (mate choice is unchanged under predation threat, but mate searching behaviors are modified to reduce conspicuousness) against the mate choice flexibility hypothesis (mate choice becomes indiscriminate under predation threat). Additionally, effectiveness of evasive behaviors may depend on predator attack strategy, so we incorporated two simulated predator cues (bird model vs predatory ranid call). We found support for the behavioral adjustment hypothesis: choosiness was maintained in the presence of predators, but females reduced conspicuousness of mate searching locomotion. Females approached the conspecific male stimuli slower and more cautiously in both predator treatments. In the ranid frog call treatment (stationary cue), females adjusted movements away from predator location. Females also attempted escape more frequently when predator cues were present. We suggest that focusing exclusively on the final mate decision may overlook nuances in mating decisions and hamper our understanding of the remarkable complexity of mate choice.
Significance statement
The presence of predators is an inherent threat to survival. This leads to the general expectation that higher predation risk results in more indiscriminate mate choice decisions and, hence, a weakening of sexual selection. Yet, discriminating mate choice may be maintained if prudent prey change their approach behavior when detecting the presence of a predator. We conducted playback trials with female treefrogs to test whether their willingness to invest in obtaining a more attractive mate (quantified by “choosiness”) differed depending on the presence and type of predation risk. We found that females adjusted their approach behavior in a way that should make them less conspicuous to predators, but that they did not compromise their mate choice decisions. Our results show that strong sexual selection by females’ choice can be maintained in high predation environments.



Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available as supplementary material.
Code availability
Not applicable.
References
Acharya L, McNeil J (1998) Predation risk and mating behavior: the responses of moths to bat-like ultrasound. Behav Ecol 9:552–558. https://doi.org/10.1093/beheco/9.6.552
Ah-King M, Gowaty PA (2016) A conceptual review of mate choice: stochastic demography, within-sex phenotypic plasticity, and individual flexibility. Ecol Evol 6:4607–4642. https://doi.org/10.1002/ece3.2197
Andersson M (1994) Sexual Selection. Princeton University Press, Princeton, NJ
Bailey NW, Zuk M (2008) Acoustic experience shapes female mate choice in field crickets. Proc R Soc Lond B 275:2645–2650. https://doi.org/10.1098/rspb.2008.0859
Bastien B, Farley G, Ge F, Malin JS, Simon-Plumb CL, Pulley DM, Yang C, Baugh AT (2018) The waiting and mating game: condition dependent mate sampling in female gray treefrogs (Hyla versicolor). Front Ecol Evol 6:140. https://doi.org/10.3389/fevo.2018.00140
Bateson P (1983) Mate Choice. Cambridge University Press, Cambridge
Batschelet E (1981) Circular Statistics in Biology. Academic Press, London
Baugh AT, Ryan MJ (2009) Female túngara frogs vary in commitment to mate choice. Behav Ecol 20:1153–1159. https://doi.org/10.1093/beheco/arp120
Baugh AT, Ryan MJ (2010) Ambient light alters temporal-updating behaviour during mate choice in a Neotropical frog. Can J Zool 88:448–453. https://doi.org/10.1139/Z10-018
Baugh AT, Gall MD, Silver SC, Bee MA (2021) Moderately elevated glucocorticoids increase mate choosiness but do not affect sexual proceptivity or preferences in female gray treefrogs. Horm Behav 130:104950. https://doi.org/10.1016/j.yhbeh.2021.104950
Booksmythe I, Detto T, Backwell PRY (2008) Female fiddler crabs settle for less: the travel costs of mate choice. Anim Behav 76:1775–1781. https://doi.org/10.1016/j.anbehav.2008.07.022
Bulbert MW, Page RA, Bernal XE (2015) Danger comes from all fronts: predator-dependent escape tactics of túngara frogs. PLoS ONE 10:e0120546. https://doi.org/10.1371/journal.pone.0120546
Chaine AS, Lyon BE (2008) Adaptive plasticity in female mate choice dampens sexual selection on male ornaments in the lark bunting. Science 319:459–462. https://doi.org/10.1126/science.1149167
Cooper WE, Caldwell J, Vitt LJ (2009) Conspicuousness and vestigial escape behaviour by two dendrobatid frogs, Dendrobates auratus and Oophaga pumilio. Behaviour 146:325–349. https://doi.org/10.1163/156853909X410946
Coyne JA, Orr HA (2004) Speciation. Sinauer Associates, Sunderland, MA
Cronin AD, Ryan MJ, Page RA, Hunter KL, Taylor RC (2019) Environmental heterogeneity alters mate choice behavior for multimodal signals. Behav Ecol Sociobiol 73:43. https://doi.org/10.1007/s00265-019-2654-3
Crowley PH, Travers SE, Linton MC, Cohn SL, Sih A, Sargent RC (1991) Mate density, predation risk, and the seasonal sequence of mate choices: a dynamic game. Am Nat 137:567–596. https://doi.org/10.1086/285184
Darwin C (1871) The descent of man and selection in relation to sex. John Murray Press, London, UK
Domenici P, Blagburn JM, Bacon JP (2011) Animal escapology II: escape trajectory case studies. J Exp Biol 214:2474–2494. https://doi.org/10.1242/jeb.053801
Edomwande C, Barbosa F (2020) The influence of predation risk on mate signaling and mate choice in the lesser waxmoth Achroia grisella. Sci Rep 10:524. https://doi.org/10.1038/s41598-020-57481-1
Eilam D (2005) Die hard: A blend of freezing and fleeing as a dynamic defense—implications for the control of defensive behavior. Neurosci Biobehav Rev 29:1181–1191. https://doi.org/10.1016/j.neubiorev.2005.03.027
Ferrari MCO, Chivers DP (2009) Temporal variability, threat sensitivity and conflicting information about the nature of risk: understanding the dynamics of tadpole antipredator behaviour. Anim Behav 78:11–16. https://doi.org/10.1016/j.anbehav.2009.03.016
Gerhardt HC (1992) Conducting playback experiments and interpreting their results. In: McGregor PK (ed) Playback and studies of animal communication. Plenum Press, New York, pp 59–77
Gerhardt HC, Tanner SD, Corrigan CM, Walton HC (2000) Female preference functions based on call duration in the gray tree frog (Hyla versicolor). Behav Ecol 11:663–669. https://doi.org/10.1093/beheco/11.6.663
Godin J-GJ, Briggs SE (1996) Female mate choice under predation risk in the guppy. Anim Behav 51:117–130. https://doi.org/10.1006/anbe.1996.0010
Gong A, Gibson RM (1996) Reversal of a female preference after visual exposure to a predator in the guppy, Poecilia reticulata. Anim Behav 52:1007–1015. https://doi.org/10.1006/anbe.1996.0248
Gowaty PA, Hubbell SP (2009) Reproductive decisions under ecological constraints: it’s about time. P Natl Acad Sci USA 106:10017–10024. https://doi.org/10.1073/pnas.0901130106
Hedrick AV, Dill LM (1993) Mate choice by female crickets is influenced by predation risk. Anim Behav 46:193–196. https://doi.org/10.1006/anbe.1993.1176
Helfman G (1989) Threat-sensitive predator avoidance in damselfish-trumpetfish interactions. Behav Ecol Sociobiol 24:47–58. https://doi.org/10.1007/BF00300117
Howland HC (1974) Optimal strategies for predator avoidance: the relative importance of speed and maneuverability. J Theor Biol 47:333–350. https://doi.org/10.1016/0022-5193(74)90202-1
Ilany A, Eilam D (2008) Wait before running for your life: defensive tactics of spiny mice (Acomys cahirinus) in evading barn owl (Tyto alba) attack. Behav Ecol Sociobiol 62:923–933. https://doi.org/10.1007/s00265-007-0516-x
Jancowski K, Orchard S (2013) Stomach contents from invasive American bullfrogs Rana catesbeiana (= Lithobates catesbeianus) on southern Vancouver Island, British Columbia, Canada. NeoBiota 16:17–37. https://doi.org/10.3897/neobiota.16.3806
Janetos AC (1980) Strategies of female mate choice: a theoretical analysis. Behav Ecol Sociobiol 7:107–112. https://doi.org/10.1007/BF00299515
Jennions MD, Petrie M (1997) Variation in mate choice and mating preferences: a review of causes and consequences. Biol Rev 72:283–327. https://doi.org/10.1111/j.1469-185X.1997.tb00015.x
Johnson JB, Basolo AL (2003) Predator exposure alters female mate choice in the green swordtail. Behav Ecol 14:619–625. https://doi.org/10.1093/beheco/arg046
Karino K, Kuwamura T, Nakashima Y, Sakai Y (2000) Predation risk and the opportunity for female mate choice in a coral reef fish. J Ethol 18:109–114. https://doi.org/10.1007/s101640070009
Kim TW, Christy JH, Dennenmoser S, Choe JC (2009) The strength of a female mate preference increases with predation risk. Proc R Soc Lond B 276:775–780. https://doi.org/10.1098/rspb.2008.1070
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
Lind J, Kaby U, Jakobsson S (2002) Split-second escape decisions in blue tits (Parus caeruleus). Naturwissenschaften 89:420–423. https://doi.org/10.1007/s00114-002-0345-8
Martín J, Luque-Larena JJ, López P (2005) Factors affecting escape behavior of Iberian green frogs (Rana perezi). Can J Zool 83:1189–1194. https://doi.org/10.1139/z05-114
Milinski M (1997) How to avoid seven deadly sins in the study of behavior. Adv Study Behav 26:159–180. https://doi.org/10.1016/S0065-3454%2808%2960379-4
Nakagawa S, Schielzeth H (2010) Repeatability for Gaussian and non-Gaussian data: a practical guide for biologists. Biol Rev 85:935–956. https://doi.org/10.1111/j.1469-185X.2010.00141.x
Neelon DP, Höbel G (2017) Social plasticity in choosiness in green tree frogs, Hyla cinerea. Behav Ecol 28:1540–1546. https://doi.org/10.1093/beheco/arx103
Neelon DP, Rodríguez RL, Höbel G (2019) On the architecture of mate choice decisions: preference functions and choosiness are distinct traits. Proc R Soc B 286:20182830. https://doi.org/10.1098/rspb.2018.2830
Nishiumi N, Mori A (2015) Distance-dependent switching of anti-predator behavior of frogs from immobility to fleeing. J Ethol 33:117–124. https://doi.org/10.1007/s10164-014-0419-z
Nishiumi N, Mori A (2020) A game of patience between predator and prey: waiting for opponent’s action determines successful capture or escape. Can J Zool 98:351–357. https://doi.org/10.1139/cjz-2019-0164
Partridge L, Halliday T (1984) Mating patterns and mate choice. In: Krebs JR, Davies NB (eds) Behavioural Ecology, 2nd edn. Blackwell Science, Oxford, pp 222–250
Pomiankowski A (1987) The costs of choice in sexual selection. J Theor Biol 128:195–218. https://doi.org/10.1016/s0022-5193(87)80169-8
R Development Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, http://www.R-project.org
Rand AS, Bridarolli ME, Dries L, Ryan MJ (1997) Light levels influence female choice in túngara frogs: predation risk assessment? Copeia 1997:447–450. https://doi.org/10.2307/1447770
Real L (1990) Search theory and mate choice. I. Models of Single-Sex Discrimination Am Nat 136:376–405. https://doi.org/10.1086/285103
Reichert MS, Höbel G (2015) Modality interactions alter the shape of acoustic mate preference functions in gray treefrogs: multimodal preference functions. Evolution 69:2384–2398. https://doi.org/10.1111/evo.12750
Rodríguez RL, Hallett AC, Kilmer JT, Fowler-Finn KD (2013) Curves as traits: genetic and environmental variation in mate preference functions. J Evol Biol 26:434–442. https://doi.org/10.1111/jeb.12061
Rosenthal R (1991) Meta-analytic procedures for social research, revised. Sage Publications, Newbury Park
Rosenthal G (2017) Mate Choice: The evolution of sexual decision making from microbes to humans. Princeton University Press, Princeton, NJ
Ryan MJ (2001) Anuran Communication. Smithsonian Institution Press, Washington DC
Sakaluk SK, Belwood JJ (1984) Gecko phonotaxis to cricket calling song: a case of satellite predation. Anim Behav 32:659–662. https://doi.org/10.1016/S0003-3472(84)80141-4
Schwartz JJ, Bee MA, Tanner SD (2000) A behavioral and neurobiological study of the responses of Gray Treefrogs, Hyla versicolor, to the calls of a predator, Rana catesbeiana. Herpetologica 56:27–37
Shifferman E, Eilam D (2004) Movement and direction of movement of a simulated prey affect the success rate in barn owl Tyto alba attack. J Avian Biol 35:111–116. https://doi.org/10.1111/j.0908-8857.2004.03257.x
Sih A (1994) Predation risk and the evolutionary ecology of reproductive behaviour. J Fish Biol 45:111–130. https://doi.org/10.1111/j.1095-8649.1994.tb01087.x
Sueur J, Aubin T, Simonis C (2008) Seewave, a free modular tool for sound analysis and synthesis. Bioacoustics 18:213–226. https://doi.org/10.1080/09524622.2008.9753600
Sullivan BK, Hinshaw SH (1992) Female choice and selection on male calling behaviour in the grey treefrog Hyla versicolor. Anim Behav 44:733–744. https://doi.org/10.1016/S0003-3472(05)80299-4
Tobler M, Schlupp I, Plath M (2008) Does divergence in female mate choice affect male size distributions in two cave fish populations? Biol Lett 4:452–454. https://doi.org/10.1098/rsbl.2008.0259
Underhill VA, Höbel G (2017) Variation in nocturnal light levels does not alter mate choice behavior in female eastern gray treefrogs (Hyla versicolor). Behav Ecol Sociobiol 71:151. https://doi.org/10.1007/s00265-017-2386-1
Weihs D, Webb PW (1984) Optimal avoidance and evasion tactics in predator-prey interactions. J Theor Biol 106:189–206. https://doi.org/10.1016/0022-5193(84)90019-5
Weissburg M, Smee DL, Ferner MC (2014) The sensory ecology of nonconsumptive predator effects. Am Nat 184:141–157. https://doi.org/10.1086/676644
Werner EE, Wellborn GA, McPeek MA (1995) Diet composition in postmetamorphic bullfrogs and green frogs: implications for interspecific predation and competition. J Herpetol 29:600–607. https://doi.org/10.2307/1564744
West-Eberhard MJ (1983) Sexual selection, social competition, and speciation. Q Rev Biol 58:155–183. https://doi.org/10.1086/413215
Acknowledgements
We thank K. Stratman for his assistance with data collection, N. Byers and M. Byers for access to their property, and staff at the University of Wisconsin’s Field Station for building access and logistical support. We also thank C. Diebold, K. Stratman, H. Warner, and J. Cannizzaro for their insightful feedback on an earlier draft of the manuscript and constructive reviewer comments.
Funding
OSF was supported by the UWM Louise Neitge Mather Scholarship.
Author information
Authors and Affiliations
Contributions
All authors conceptualized the study and experimental design. Methods were executed by OSF. OSF prepared the manuscript drafts, and GH critically reviewed and edited subsequent versions. All authors have read and approved the final manuscript and submission for publication.
Corresponding author
Ethics declarations
Ethics approval
All procedures were approved by the Animal Care and Use Committee of the University of Wisconsin Milwaukee (Protocol Number: 19–20#26).
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by K. Summers
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Feagles, O.S., Höbel, G. Female gray treefrogs maintain mate choice decisions under predation threat, but adjust movements to reduce conspicuousness during mate approach. Behav Ecol Sociobiol 76, 17 (2022). https://doi.org/10.1007/s00265-021-03124-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-021-03124-y