Abstract
Introduction
Numerous randomized controlled trials (RCTs) have investigated PD-1/PD-L1 inhibitor-based combination therapies. The debate surrounding the potential additive clinical benefits of combination of two immune-oncology (IO) therapies for cancer patients persists.
Methods
Both published and grey sources of randomized clinical trials that compared anti-PD-1/PD-L1-based immunotherapy combinations with monotherapy in patients with advanced or metastatic solid tumors were encompassed. The primary outcome was progression-free survival (PFS), and secondary outcomes included objective response rate (ORR), overall survival (OS) and treatment-related adverse events (TRAEs).
Results
Our analysis encompassed 31 studies comprising 10,341 patients, which covered 12 distinct immune-oncology combination regimens. Across all patients, the immunotherapy combinations exhibited the capability to enhance the ORR (OR = 1.23 [95% CI 1.13–1.34]) and extend PFS (HR = 0.91 [95% CI 0.87–0.95]). However, the observed enhancement in OS (HR = 0.96 [95% CI 0.91–1.01]) was of no significance. Greater benefits in terms of PFS (HR = 0.82 [95% CI 0.72 to 0.93]) and OS (HR = 0.85 [95% CI 0.73 to 0.99]) may be particularly pronounced in cases where PD-L1 expression is negative. Notably, despite a heightened risk of any-grade TRAEs (OR = 1.72 [95% CI 1.40–2.11]) and grade greater than or equal to 3 TRAEs (OR = 2.01 [95% CI 1.67–2.43]), toxicity was generally manageable.
Conclusions
This study suggests that incorporating an additional immunotherapy agent with PD-1/PD-L1 inhibitors can elevate the response rate and reduce the risk of disease progression, all while maintaining manageable toxicity. However, there remains a challenge in translating these primary clinical benefits into extended overall survival.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Therapeutic options for patients with inoperable tumors were historically limited to chemotherapy, which often carried significant adverse events and frequent instances of relapse [1]. In the pursuit of enhanced efficacy and improved tolerability, extensive research has brought up novel therapeutic agents and regimens with multifaceted functions, particularly within the realm of cancer immunotherapy which has ushered in a new era of cancer treatment over the past decade [2]. The promising advancements in immune checkpoint inhibitors (ICBs), especially those targeting CTLA-4 or PD-1/PD-L1, have garnered substantial attention due to their favorable clinical outcomes across various malignancies, supported by an array of ongoing trials that are diligently exploring their potential across different indications [3]. However, ICB monotherapy only confers prolonged lifespan to a subset of patients, primarily due to innate and acquired resistance. This limitation underscores the pressing need for alternative strategies that can deliver improved performance and outcomes [4].
The concept of combined immunotherapies targeting multiple immune pathways holds the potential for establishing a durable antitumor immune response and expanding the patient population that could benefit from immunotherapy [5]. Combing PD-1/PD-L1 inhibitor with CTLA-4 inhibitor was demonstrated to amplify antitumor responses synergistically [6,7,8,9], and the promising outcomes has led to the approval of the combination therapy for a broad spectrum of malignancies [10].
However, controversies persist over the additional value of IO combinations including synergy of PD-1/PD-L1 inhibitor with another immune checkpoint inhibitor or novel IO drug. Although certain clinical trials have yielded unsatisfactory results with no discernible additional benefit from dual immune checkpoint blockades [11, 12], predefined secondary analyses within the DANUBE study have lent support to the hypothesis that combining durvalumab with tremelimumab might lead to increased activity compared to durvalumab alone. This effect was particularly pronounced in the biomarker-positive population, underscoring the crucial role of precise patient selection in achieving optimal therapeutic benefits [13]. Similarly, clinical trials exploring other immunotherapy combination regimens yielded no certain additional benefit over monotherapy [14, 15]. What’s more, numerous meta-analyses [16,17,18,19,20] consistently indicated a noteworthy rise in the frequency of toxicity incidents when anti-PD-1/PD-L1 and anti-CTLA-4 blockades are administered in tandem.
The discrepancies in clinical efficacy results and concerns regarding toxicity have raised additional doubts about the utilization of IO combinations. Thus, we determined to conduct this meta-analysis aimed at investigating whether the combination of PD-1/PD-L1 blockade with another immunotherapy could offer greater survival benefits to a larger number of patients across various solid tumors while maintaining an acceptable safety profile, as compared to PD-1/PD-L1 blockade alone. The detailed toxicity profiles of adverse events with a potential immunologic cause were also aggregated to provide a clinical reference.
Materials and methods
This meta-analysis was designed to identify trials employing combination therapies consisting of PD-1/PD-L1-inhibitor-based dual IO therapies that reported efficacy and safety outcomes. It was performed under the guidance of the PRISMA statement [21] and a prospective protocol was registered as INPLASY202370063 (https://inplasy.com).
Data sources and search strategy
We systematically searched PubMed, EMBASE, Scopus, Cochrane Library databases for relevant trials conducted and reported until November 18, 2023. The ClinicalTrials.gov and abstract books of annual conferences that took place from 2018 to 2023 were also screened in order to include the updated outcomes and unpublished trials, including American Society of Clinical Oncology, European Society of Medical Oncology, Chinese Society of Clinical Oncology, American Association of Cancer Research, and World Conference of Lung Cancer. The keywords for the literature search were “randomized clinical trial, cancer, PD-1, PD-L1, pembrolizumab, atezolizumab, nivolumab, durvalumab, camrelizumab, tislelizumab, sintilimab, toripalimab and serplulimab”.
Selection criteria
Published and gray sources which met the following criteria were included in this meta-analysis: RCTs enrolling patients with advanced or metastatic solid tumor confirmed either histologically or cytologically; RCTs that used PD-1/PD-L1 inhibitor combining another immunotherapy in either first-line or later treatment settings; RCTs comparing PD-1/PD-L1 inhibitor plus another IO therapy with anti-PD-1/PD-L1 monotherapy for patients with advanced or metastatic solid tumor; phase II or phase III trials reporting at least one of the following clinical outcomes: progression-free survival (PFS), overall survival (OS), objective response rate (ORR), treatment-related Adverse events (TRAEs) of any-grade or grade greater than or equal to 3 TRAEs.
The exclusion criteria were as follows: RCTs that enrolled patients with hematologic malignancies; RCTs comparing dual IO combinations with immunologic monotherapy combined with chemotherapy, tyrosine kinase inhibitor or other treatment modality.
To determine the eligibility, titles and abstracts were screened before assessing full texts. All the included trials were double-checked online to make sure that the most recent data was included.
Data extraction and quality assessment
Basic information about the trial name, first author, publication year, publication source, National Clinical Trials identification number, cancer type, sample size, patients’ age and sex distribution, names of PD-1 or PD-L1 inhibitor, names of combination agents were extracted. The clinical outcomes we extracted encompassed ORR and hazard ratios (HRs) with corresponding 95% confidence intervals (95% CIs) for PFS and OS. We also recorded the incidence of any-grade TRAEs and grade greater than or equal to 3 TRAEs, along with the safety profile of events experienced by patients who received IO combinations. Part of included trials only provide information about any-grade TRAEs reported in at least 5%, 10% or 15% patients [12, 15, 22,23,24,25,26]. Consequently, any-grade events experienced by ≥ 5% as well as grade ≥ 3 events experienced by ≥ 1% patients in the combination arms were listed after extraction and calculation. Profile of immune-mediated adverse events (imAEs) or adverse events of special interest (AESIs) with a potential immunologic mechanism were also extracted from published reports.
Data extraction was independently done by two investigators (Y.C. and J.C.) and any discrepancies in this process were resolved through discussion with the other investigator (H.H.). The Cochrane Risk of Bias Tool (2.0) for RCTs was applied to check the quality of the included studies.
Statistical analysis
The primary outcome was PFS, while the secondary outcomes included ORR, OS, any-grade TRAEs, grade greater than or equal to 3 TRAEs and AESIs. HRs with corresponding 95% CIs were utilized to quantify the effect sizes for PFS and OS. On the other hand, ORs with 95% CIs were employed to measure the effect sizes for ORR, any-grade TRAEs, and grade greater than or equal to 3 TRAEs. The risks of each event were measured by risk ratios (RRs) with 95%CIs. Moreover, the statistic inconsistency index (I2) was employed to assess the heterogeneity among studies. An I2 value exceeding 50% is conventionally considered indicative of substantial heterogeneity, prompting the application of a random-effects model with a logit transformation and subsequent sensitivity analysis. Otherwise, the fixed-effects model was applied. The models were fitted using restricted maximum likelihood estimation, incorporating a classic continuity correction of 0.5 for cells with zero values along with their corresponding sample sizes.
Subgroup analyses were performed based on the type of combination agent, PD-L1 expression status, cancer type and line of treatment. In the subgroup analysis of clinical outcomes for patients with varying PD-L1 expression status, only PFS and OS could be estimated. Given that only two trials reported clinical outcomes for patients with PD-L1 expression exceeding 25%, the patients were categorized into the following groups: less than 1%, greater than or equal to 1%, less than 25%, and greater than or equal to 50%.
Some three-arm trials were initially designed to compare immunotherapy combined with chemotherapy, along with evaluating PD-1/PD-L1 blockade monotherapy against chemotherapy. These trials reported median PFS/OS data and event counts for each group. We used these data to calculate HRs and their corresponding 95% CIs for anti-PD-1/PD-L1-based combinations in comparison to PD-1/PD-L1 monotherapy [27,28,29]. For studies presenting only Kaplan–Meier curves, the IPDfromKM method [30] was employed to reconstruct individual patient data and conduct survival analysis.
Publication bias was evaluated using the Egger’s regression test in conjunction with a funnel plot. A p value of less than 0.10 was deemed significant, suggesting the presence of asymmetry and publication bias. All analytical procedures were conducted using STATA software (version 17) and SPSS statistical software (version 26.0).
Results
Systematic review and characteristics of the included studies
We initially identified a total of 16,639 records across the 4 databases. After removing duplicates and non-pertinent articles through abstract screening, 521 records were deemed eligible for full-text review. Additionally, 13 studies were sourced from ClinicalTrials.gov and conference proceedings. Ultimately, 32 randomized controlled trials were found to meet our eligibility criteria (Fig. 1). Detailed information and baseline characteristics can be found in the provided table (Table 1). Detailed assessment of each randomized controlled trials on the risk of bias is presented in supplementary materials (Supplementary Fig. S1).
Comparison of PFS, OS and ORR
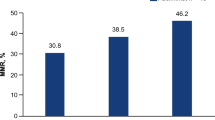
21 studies included in this meta-analysis provide information about PFS. Generally, patients receiving PD-1/PD-L1-inhibitor-based IO combinations were prone to longer survival without progression (HR = 0.91 [95% CI 0.87–0.95]). Among the 32 trials included in this meta-analysis, 31 reported ORR, with some of them additionally providing information for comparing OS. In general, combination therapies based on PD-1/PD-L1 blockade exhibited a higher response rate (OR = 1.24 [95% CI 1.13–1.35]). When considering OS, IO combinations conferred greater benefit compared to PD-1 or PD-L1 inhibitor monotherapy (HR = 0.96 [95% CI 0.91–1.01]), although the observed differences in OS were not statistically significant (Fig. 2).
Comparisons of any-grade TRAEs and grade ≥ 3 TRAEs
Safety and toxicity were evaluated based on the incidence of any-grade TRAEs and grade greater than or equal to 3 TRAEs. Specifically, 16 and 22 studies were respectively incorporated to assess any-grade TRAEs and grade greater than or equal to 3 TRAEs.
The combination of two immunotherapeutic agents exhibited a significant increase in toxicity (OR = 1.72 [95% CI 1.40–2.11]), particularly in relation to the elevated risk of higher-grade adverse events (OR = 2.01 [95% CI 1.67–2.43]) (Fig. 2). Among the total of 17 clinical trials that provided detailed information about any-grade TRAEs in their articles or supplementary materials, the most frequently reported adverse events for IO combinations encompassed fatigue, chills, pruritus, diarrhea, and pyrexia. Notably, special attention is warranted for infusion-related rash (RR = 17.78 [95% CI 1.05–300.08]), chills (RR = 6.31 [95% CI 3.49–11.42]) and pyrexia (RR = 4.08 [95% CI 3.03–5.48]), all of which displayed significantly elevated risk ratios. When considering grade greater than or equal to 3 TRAEs, clinicians are advised to exercise vigilance regarding anemia (RR = 4.10 [95% CI 1.17–14.34]), thrombocytopenia (RR = 4.73 [95% CI 2.32–9.65]), diarrhea (RR = 3.37 [95% CI 2.13–5.34]), and colitis (RR = 3.93 [95% CI 1.92–8.08]) (Supplementary Table 2).
Among adverse events of special interest (AESIs), a heightened focus may be required on sarcoidosis (RR = 8.96 [95% CI 0.48–166.13]), hypopituitarism (RR = 6.04 [95% CI 0.73–49.99]), increases in ALT (RR = 4.58 [95% CI 2.50–8.39]) or AST (RR = 4.00 [95% CI 2.17–7.38]), autoimmune hepatitis (RR = 3.99 [95% CI 0.45–35.51]), and colitis (RR = 3.78 [95% CI 2.47–5.80]). Notably, greater attention should be directed towards grade ≥ 3 adverse events with a potential inflammatory or immunologic cause, including pruritus (RR = 17.07 [95% CI 0.99–295.31]), hypopituitarism (RR = 9.05 [95% CI 0.491–67.81]), increases in ALT (RR = 6.50 [95% CI 2.30–18.41]) or AST (RR = 6.33 [95% CI 1.89–21.19]), and type 1 diabetes mellitus (RR = 5.51 [95% CI 0.70–45.75]) (Supplementary Table 3).
Subgroup analysis
On the basis of combination agent
Given the approved usage of dual immune checkpoint inhibitors targeting PD-1/PD-L1 and CTLA-4 across a range of malignancies, we categorized the included trials into two distinct groups: “anti-PD-1/PD-L1 plus anti-CTLA-4” and “anti-PD-1/PD-L1 plus another IO therapy.” In comparison to PD-1/PD-L1 inhibitor monotherapy, the addition of a CTLA-4 inhibitor demonstrated an increase in the response rate (OR = 1.29 [95% CI 1.15–1.44]) and potential extension of survival without progression (HR = 0.91 [95% CI 0.86–0.96]). A subtle variation in OS was also observed (HR = 0.96 [95% CI 0.90–1.02]). Correspondingly, the combined therapy exhibited an elevated incidence of adverse events. The odds ratio was 1.52 [95% CI 1.19–1.94] for any-grade TRAEs and 2.11 [95% CI 1.74–2.57] for grade greater than or equal to 3 TRAEs (Supplementary Fig. S2).
Furthermore, the combination of anti-PD-1/PD-L1 with a different type of immunotherapy demonstrated enhancements in the ORR (OR = 1.15 [95% CI 1.01–1.32]) and PFS (HR = 0.91 [95% CI 0.83–1.00]). However, these novel combinatorial regimens were comparatively less effective than the traditional pairing of PD-1/PD-L1 inhibitor and CTLA-4 inhibitor. Notably, there were minimal differences between combination therapy and monotherapy concerning OS (HR = 0.98 [95% CI 0.88–1.09]). The risk of any-grade (OR = 2.25 [95% CI 1.50–3.38]) and grade equal–or higher than 3 TRAEs (OR = 1.88 [95% CI 1.25–2.83]) were both significantly higher than PD-1/PD-L1 blockade alone (Supplementary Fig. S2).
On the basis of PD-L1 status
The most pronounced trend toward enhanced PFS (HR = 0.82 [95% CI 0.72–0.93]) was evident among patients with a lower PD-L1 expression (less than 1%) accompanied by improved OS (HR = 0.85 [95% CI 0.73–0.99]). Among patients with PD-L1 expression greater than or equal to 1%, the combination therapy were capable of prolonging survival without progression (HR = 0.95 [95% CI 0.87–1.04]). However, when compared to PD-1/PD-L1 inhibitor monotherapy, the addition of another immunotherapy did not yield notable benefits in terms of OS (HR = 1.00 [95% CI 0.90–1.11]) (Supplementary Fig. S3).
Regarding patients with PD-L1 expression less than 25%, the results were comparable, demonstrating an improvement in PFS (HR = 0.95 [95% CI 0.84–1.09]) and minimal deviation in OS (HR = 0.99 [95% CI 0.86–1.14]). For patients with PD-L1 expression greater than or equal to 50%, the combination of PD-1/PD-L1 blockade and another immunotherapy also demonstrated a potential reduction in the risk of disease progression (HR = 0.90 [95% CI 0.80–1.02]) and death (HR = 0.99 [95% CI 0.87–1.13]). These results implied a potential link between PD-L1 status and efficacy (Supplementary Fig. S3).
On the basis of cancer type
To evaluate clinical efficacy and safety based on cancer type, patients were categorized into four groups: non-small-cell lung cancer (NSCLC), head and neck squamous cell cancer (HNSCC), melanoma, and other.
For patients with NSCLC, the combination of two IO therapies demonstrated improvements in both PFS (HR = 0.90 [95% CI 0.83–0.98]) and OS (HR = 0.95 [95% CI 0.87–1.04]), as deduced from data extracted from eight trials analyzed in the subgroup analysis. Patients with melanoma exhibited a comparable trend, with extended PFS (HR = 0.90 [95% CI 0.83–0.98]) and OS (HR = 0.92 [95% CI 0.82–1.02]). However, the outcomes for patients with HNSCC were less encouraging, indicating a potential lack of benefit in terms of PFS (HR = 0.99 [95% CI 0.89–1.11]) and OS (HR = 1.09 [95% CI 0.98–1.21]). Conversely, among patients with other types of cancer, there appeared to be a lower risk of disease progression (HR = 0.87 [95% CI 0.78–0.96]) as well as the inspiriting improvement in OS (HR = 0.90 [95% CI 0.80–1.01]) (Supplementary Fig. S4).
Turning to odds ratios for any-grade TRAEs, other types of cancer displayed the highest incidence (OR = 2.60 [95% CI 2.09–3.23]), followed by melanoma (OR = 2.12 [95% CI 1.34–3.35]) and NSCLC (OR = 1.63 [95% CI 1.19–2.24]). Notably, patients with NSCLC were more prone to experiencing grade greater than or equal to 3 TRAEs (OR = 2.31 [95% CI 1.64–3.26]). Pooling available data, the collective outcomes indicated that combination immunologic therapies were generally well-tolerated in patients with HNSCC, as evidenced by odds ratios for any-grade and grade greater than or equal to 3 TRAEs of 1.13 [95% CI 0.82– 1.56] and 1.77 [95% CI 1.32–2.39], respectively (Supplementary Fig. S4).
On the basis of treatment setting
Immunotherapy combination seems to be more advantageous over PD-1/PD-L1 inhibitor alone to improve response rate in the post-line treatment setting (OR = 1.33 [95%CI 1.09–1.62]), but is more beneficial to prolong overall survival in the first-line treatment (OR = 0.95 [95%CI 0.89–1.01]). The increased incidence of adverse events while receiving IO combination regimens was more pronounced in first-line application than in non-first-line application. (Supplementary Fig. S5).
Heterogeneity analysis and publication bias
To validate the consistency of our findings, sensitivity analyses were performed by systematically omitting one study at a time to evaluate if the aggregated outcomes were significantly influenced by any particular trial. Encouragingly, the results indicated that the overall estimates remained stable throughout these evaluations. A marginal to moderate level of heterogeneity across the included trials was identified. Meta-regression analysis was conducted to delve into the potential sources of heterogeneity in the pooled outcomes of any-grade and grade higher than or equal to 3 TRAEs. Trial phase, line of treatment, cancer type, type of PD-1/PD-L1 inhibitor, and target of the combination agent were considered as categorial coefficients while the sources of heterogeneity remained challenging to fully elucidate and explain (Supplementary Table 4). No significant evidence of publication bias was observed, as the funnel plots displayed no discernible asymmetry (Supplementary Fig. S6). This observation was further confirmed through the application of Egger’s test (p = 0.673) (Supplementary Table 5).
Discussion
Immunotherapy has changed the landscape of cancer treatment, especially the encouraging performance of ICBs, and combination therapies arose with the aim of restoring immune function. However, whether the combination could outperform monotherapy remains controversial. To our knowledge, this meta-analysis is the first to directly compare clinical efficacy and safety of PD-1/PD-L1 inhibitor-based IO combinations with single PD-1/PD-L1 inhibitor. The pooled results indicate that combining anti-PD-1/PD-L1 with another immunotherapeutic agent is able to augment both response rates and progression-free survival, all while maintaining a manageable level of treatment-related adverse events which can be resolved with the help of therapeutic agents.
Combination therapies may bolster antitumor immune response and broaden patient population who might benefit from immunotherapy [5]. In contrast to CTLA-4, which operates within lymph nodes to dampen T-cell activation, PD-1 plays a role in hindering T effector cells and promoting Treg differentiation in peripheral tissues [1]. The spatial segregation of functions, along with their complementary and non-redundant roles in effectively targeting both CD8+T cells and CD4 + T cells, underscore the rationale for combination therapy [31]. Other PD-1/PD-L1 inhibitor-based strategies have also been hypothesized to synergistically enhancing T-cell responses and overcoming PD-L1-related immune evasion within the intricate tumor microenvironment [14, 32]. Beyond the realm of immune checkpoint molecules, targeting co-stimulatory pathways play a pivotal role in bolstering T cell activity [33] and revitalizing antitumor immune responses [34]. Moreover, a spectrum of immune and non-immune cell types, along with the myriad factors they secrete, has been identified within the tumor microenvironment (TME). These elements collectively establish a chronic inflammatory, immunosuppressive, and pro-angiogenic intertumoral milieu [35]. Cytokines orchestrating critical interactions between immune and non-immune cells, such as IL-2, IL-12, and IL-15, have been harnessed [36] and demonstrated the potential to rejuvenate exhausted T cells [37], decrease regulatory T cells (Treg) in the TME, or reprogram macrophage polarization [36], thus enhancing tumor control in synergy with immune checkpoint inhibitors in preclinical models and early-phase clinical trials [38,39,40,41]. A series of clinical investigations involving novel immunotherapeutic agents are underway [42,43,44,45]. Furthermore, preclinical investigations and early-phase clinical trials have demonstrated enhanced anti-tumor efficacy and safety of bispecific antibodies compared to traditional multi-target therapies, including combination approaches [37, 46,47,48,49,50,51]. Notably, the improvement in OS was of no statistical significance. This may be explained by acquired drug resistance, and the mechanisms behind include acquisition of mutations in essential pathways of immunotherapy response, the loss of tumor neoantigens, co-inhibition molecules in the tumor microenvironment and so on [52]. Given the complicated and dynamic TME, further studies exploring how to translate benefit in tumor response and progression-free survival into prolonged overall survival are needed.
The intriguing observations in our study provoke inquiries into the reliability of PD-L1 as a dependable biomarker for patient selection at low cutoffs and within the realm of combination therapies. An interesting observation emerged when examining the PD-L1 subgroups. Specifically, the group with PD-L1 expression < 1% exhibited a more favorable response to combination therapy compared with monotherapy, as opposed to the PD-L1 ≥ 1% group. This intriguing phenomenon could potentially be attributed to the distinctive immune effects of anti-PD-1 and anti-CTLA-4 agents, which might hold particular significance in PD-L1-negative tumors [23]. Thus, the expression level of PD-L1 may not accurately represent the PD-1/PD-L1 interaction state and anti-tumor immunity [53]. The reduced reliance on PD-L1 expression in the context of IO combinations opened avenues for in-depth exploration into the intricate mechanisms at play and paved the way for the identification of more dependable biomarkers guiding patient selection. Notably, tumor mutational burden (TMB) was proposed to serve as a more robust biomarker independent of PD-L1 expression level for immunotherapy combinations [54]. There is growing recognition of other conceivable biomarkers encompassing microsatellite instability [55] as well as signatures closely tied to tumor inflammation, including those derived from analyses of inflammatory gene expression profiles, which hold promise as additional potential indicators [56]. Given the inherent tumor heterogeneity, the creation of a comprehensive framework that amalgamates PD-L1 expression status, tumor mutational burden (TMB), personalized immune profiling, genomic, transcriptomic, and microbiome data could substantially enhance predictive accuracy and facilitate future interventions [4, 10].
The most frequently reported adverse events for IO combinations in our study encompassed fatigue, chills, pruritus, diarrhea, and pyrexia, generally in line with previous study focused on treatment-related adverse events [3]. Special attention is warranted for infusion-related rash, chills and pyrexia, all of which displayed significantly elevated risk ratios. Notably, the occurrence of infusion-related rash was solely documented in 2 trials [57], possibly attributable to pegilodecakin. Given that profiles of imAEs or AESI with a potential immunologic mechanism was available in only 10 studies, we aggregated and analyzed this data to provide a comprehensive overview of events warranting particular attention. Lung cancer displayed a lower risk of all-grade and grade 3 or higher TRAEs compared to melanoma and urinary system tumors in the prior investigation [3], which was contrary to our results. This discrepancy may be attributed to the limited sample size of each cancer type in our study to some extent. What’s more, it was speculated that the prior history of chemotherapy might induce a lower baseline immunoactivity and potentially shield patients from immune system over activation. This is due to the immunosuppressive effect exhibited by chemotherapy agents, arising from their anti-proliferative impact on immune cells [58]. The influence of pretreatment may also explain the discrepancies observed to some degree, highlighting the need for further exploration and validation through additional studies. Subgroup analyses of melanoma patients revealed significant heterogeneity in clinical efficacy and safety compared to other cancer types. The heterogeneity observed may arise from the diverse baseline patient characteristics and therapeutic agents with distinct target molecules and mechanisms of action.
There are several limitations to this study. Firstly, limited by number of clinical trials directly comparing IO combinations with PD-1/PD-L1 inhibitor alone, the trials included were diverse so that the heterogeneity was hard to be fully elucidated. Not enough follow-up duration of some trials led to the limited dataset for PFS and OS statistics which could potentially constrain comprehensive investigations as well as adding to the heterogeneity of pooled results. Consequently, future randomized controlled trials are essential to suffice the pooled outcomes and more efforts should be made to explore efficient treatment combinations. Secondly, while the included studies encompassed various PD-1/PD-L1 inhibitor-based combination regimens, not all of them reported outcomes for patients with different PD-L1 expression statuses, which may hinder further analyses based on PD-L1 expression levels. Thirdly, it is important to note that some of the studies included were not explicitly designed for a formal statistical comparison between the two groups: single PD-1/PD-L1 inhibitor and PD-1/PD-L1 inhibitor-based IO combinations, calling for secondary data processing. Furthermore, future studies focusing on specific cancer types are imperative. This is particularly important considering the distinct immunogenicity profiles exhibited by different malignancies [59].
In conclusion, safety and toxicity assessments unveiled that the addition of another I-O therapy led to increased incidence of treatment-related adverse events as well as adverse events with a potential immunologic mechanism. Nevertheless, the toxicity profiles were generally manageable. Clinical efficacy was predominantly gauged by progression-free survival, overall survival, and objective response rate. The introduction of an anti-CTLA-4 inhibitor or other I-O therapy was associated with heightened objective response rates, translating to reduced risks of disease progression and mortality. The incorporation of combination therapies may also confer improvements in overall survival, although the difference was slight and statistical significance for this outcome was not achieved. Notably, the augmentation of clinical efficacy appears more pronounced in patients exhibiting high PD-L1 expression levels. Consequently, a pressing unmet need persists for identifying an optimal treatment combination that extends the lifespan of individuals with advanced or metastatic cancer, while minimizing the burden of heightened drug-related toxic effects.
References
Rotte A (2019) Combination of CTLA-4 and PD-1 blockers for treatment of cancer. J Exp Clin Cancer Res 38:255. https://doi.org/10.1186/s13046-019-1259-z
Zhou Y, Penny HL, Kroenke MA, Bautista B, Hainline K, Chea LS, Parnes J, Mytych DT (2022) Immunogenicity assessment of bispecific antibody-based immunotherapy in oncology. J Immunother Cancer. https://doi.org/10.1136/jitc-2021-004225
Zhou X, Yao Z, Bai H et al (2021) Treatment-related adverse events of PD-1 and PD-L1 inhibitor-based combination therapies in clinical trials: a systematic review and meta-analysis. Lancet Oncol 22:1265–1274. https://doi.org/10.1016/s1470-2045(21)00333-8
Chowell D, Yoo SK, Valero C et al (2022) Improved prediction of immune checkpoint blockade efficacy across multiple cancer types. Nat Biotechnol 40:499–506. https://doi.org/10.1038/s41587-021-01070-8
Ott PA, Hodi FS, Kaufman HL, Wigginton JM, Wolchok JD (2017) Combination immunotherapy: a road map. J Immunother Cancer 5:16. https://doi.org/10.1186/s40425-017-0218-5
Curran MA, Montalvo W, Yagita H, Allison JP (2010) PD-1 and CTLA-4 combination blockade expands infiltrating T cells and reduces regulatory T and myeloid cells within B16 melanoma tumors. Proc Natl Acad Sci U S A 107:4275–4280. https://doi.org/10.1073/pnas.0915174107
Selby M, Engelhardt J, Lu LS, Quigley M, Wang C, Chen B, Korman AJ (2013) Antitumor activity of concurrent blockade of immune checkpoint molecules CTLA-4 and PD-1 in preclinical models. J Clin Oncol. https://doi.org/10.1371/journal.pone.0161779
Duraiswamy J, Kaluza KM, Freeman GJ, Coukos G (2013) Dual blockade of PD-1 and CTLA-4 combined with tumor vaccine effectively restores T-cell rejection function in tumors. Cancer Res 73:3591–3603. https://doi.org/10.1158/0008-5472.Can-12-4100
Selby MJ, Engelhardt JJ, Johnston RJ et al (2016) Preclinical development of ipilimumab and nivolumab combination immunotherapy: mouse tumor models, in vitro functional studies, and cynomolgus macaque toxicology. PLoS ONE 11:e0161779. https://doi.org/10.1371/journal.pone.0161779
Yi M, Zheng X, Niu M, Zhu S, Ge H, Wu K (2022) Combination strategies with PD-1/PD-L1 blockade: current advances and future directions. Mol Cancer 21:28. https://doi.org/10.1186/s12943-021-01489-2
Goldman JW, Dvorkin M, Chen Y et al (2021) Durvalumab, with or without tremelimumab, plus platinum-etoposide versus platinum-etoposide alone in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): updated results from a randomised, controlled, open-label, phase 3 trial. Lancet Oncol 22:51–65. https://doi.org/10.1016/s1470-2045(20)30539-8
Ferris RL, Haddad R, Even C et al (2020) Durvalumab with or without tremelimumab in patients with recurrent or metastatic head and neck squamous cell carcinoma: EAGLE, a randomized, open-label phase III study. Ann Oncol 31:942–950. https://doi.org/10.1016/j.annonc.2020.04.001
Powles T, van der Heijden MS, Castellano D et al (2020) Durvalumab alone and durvalumab plus tremelimumab versus chemotherapy in previously untreated patients with unresectable, locally advanced or metastatic urothelial carcinoma (DANUBE): a randomised, open-label, multicentre, phase 3 trial. Lancet Oncol 21:1574–1588. https://doi.org/10.1016/S1470-2045(20)30541-6
Long GV, Dummer R, Hamid O et al (2019) Epacadostat plus pembrolizumab versus placebo plus pembrolizumab in patients with unresectable or metastatic melanoma (ECHO-301/KEYNOTE-252): a phase 3, randomised, double-blind study. Lancet Oncol 20:1083–1097. https://doi.org/10.1016/S1470-2045(19)30274-8
Cho BC, Abreu DR, Hussein M et al (2022) Tiragolumab plus atezolizumab versus placebo plus atezolizumab as a first-line treatment for PD-L1-selected non-small-cell lung cancer (CITYSCAPE): primary and follow-up analyses of a randomised, double-blind, phase 2 study. Lancet Oncol 23:781–792. https://doi.org/10.1016/s1470-2045(22)00226-1
Yang Y, Jin G, Pang Y et al (2020) Comparative efficacy and safety of nivolumab and nivolumab plus ipilimumab in advanced cancer: a systematic review and meta-analysis. Front Pharmacol 11:40. https://doi.org/10.3389/fphar.2020.00040
Da L, Teng Y, Wang N, Zaguirre K, Liu Y, Qi Y, Song F (2019) Organ-specific immune-related adverse events associated with immune checkpoint inhibitor monotherapy versus combination therapy in cancer: a meta-analysis of randomized controlled trials. Front Pharmacol 10:1671. https://doi.org/10.3389/fphar.2019.01671
Park R, Lopes L, Cristancho CR, Riano IM, Saeed A (2020) Treatment-related adverse events of combination immune checkpoint inhibitors: systematic review and meta-analysis. Front Oncol 10:258. https://doi.org/10.3389/fonc.2020.00258
Almutairi AR, McBride A, Slack M, Erstad BL, Abraham I (2020) Potential immune-related adverse events associated with monotherapy and combination therapy of ipilimumab, nivolumab, and pembrolizumab for advanced melanoma: a systematic review and meta-analysis. Front Oncol 10:91. https://doi.org/10.3389/fonc.2020.00091
Gu L, Khadaroo PA, Su H et al (2019) The safety and tolerability of combined immune checkpoint inhibitors (anti-PD-1/PD-L1 plus anti-CTLA-4): a systematic review and meta-analysis. BMC Cancer 19:559. https://doi.org/10.1186/s12885-019-5785-z
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Larkin J, Chiarion-Sileni V, Gonzalez R et al (2015) Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med 373:23–34. https://doi.org/10.1056/NEJMoa1504030
Hellmann MD, Paz-Ares L, Bernabe Caro R et al (2019) Nivolumab plus ipilimumab in advanced non-small-cell lung cancer. N Engl J Med 381:2020–2031. https://doi.org/10.1056/NEJMoa1910231
Gettinger SN, Redman MW, Bazhenova L et al (2021) Nivolumab plus ipilimumab vs nivolumab for previously treated patients with stage IV squamous cell lung cancer: the lung-MAP S1400I phase 3 randomized clinical trial. JAMA Oncol 7:1368–1377. https://doi.org/10.1001/jamaoncol.2021.2209
Boyer M, Sendur MAN, Rodriguez-Abreu D et al (2021) Pembrolizumab plus ipilimumab or placebo for metastatic non-small-cell lung cancer with PD-L1 tumor proportion score >/= 50%: randomized, double-blind phase III KEYNOTE-598 study. J Clin Oncol 39:2327–2338. https://doi.org/10.1200/JCO.20.03579
Tawbi HA, Schadendorf D, Lipson EJ et al (2022) Relatlimab and nivolumab versus nivolumab in untreated advanced melanoma. N Engl J Med 386:24–34. https://doi.org/10.1056/NEJMoa2109970
Cortés J, González JA, Campbell MJ, Cobo E (2014) A hazard ratio was estimated by a ratio of median survival times, but with considerable uncertainty. J Clin Epidemiol 67:1172–1177. https://doi.org/10.1016/j.jclinepi.2014.05.017
Hackshaw A (2009) Statistical formulae for calculating some 95% confidence intervals. In: A concise guide to clinical trials. pp 205–207
Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR (2007) Practical methods for incorporating summary time-to-event data into meta-analysis. Trials 8:16. https://doi.org/10.1186/1745-6215-8-16
Liu N, Zhou Y, Lee JJ (2021) IPDfromKM: reconstruct individual patient data from published Kaplan-Meier survival curves. BMC Med Res Methodol 21:111. https://doi.org/10.1186/s12874-021-01308-8
Wei SC, Levine JH, Cogdill AP et al (2017) Distinct cellular mechanisms underlie anti-CTLA-4 and anti-PD-1 checkpoint blockade. Cell 170:1120–33.e17. https://doi.org/10.1016/j.cell.2017.07.024
Chesney JA, Ribas A, Long GV et al (2023) Randomized, double-blind, placebo-controlled, global phase III trial of talimogene laherparepvec combined with pembrolizumab for advanced melanoma. J Clin Oncol 41:528–540. https://doi.org/10.1200/JCO.22.00343
Marin-Acevedo JA, Kimbrough EO, Manochakian R, Zhao Y, Lou Y (2021) Immunotherapies targeting stimulatory pathways and beyond. J Hematol Oncol 14:78. https://doi.org/10.1186/s13045-021-01085-3
Pourakbari R, Hajizadeh F, Parhizkar F, Aghebati-Maleki A, Mansouri S, Aghebati-Maleki L (2021) Co-stimulatory agonists: an insight into the immunotherapy of cancer. Excli J 20:1055–1085. https://doi.org/10.17179/excli2021-3522
Pitt JM, Marabelle A, Eggermont A, Soria JC, Kroemer G, Zitvogel L (2016) Targeting the tumor microenvironment: removing obstruction to anticancer immune responses and immunotherapy. Ann Oncol 27:1482–1492. https://doi.org/10.1093/annonc/mdw168
Tsou P, Lin WY, Lee MN, Chen SC (2022) Combination of recombinant murine IL-12 and anti-PD-1 induced synergistic anti-tumor response. Cancer Res 82:5515. https://doi.org/10.1158/1538-7445.AM2022-5515
Hsiao HW, Hsiao CL, Koni P, Chen HK (2022) A bifunctional anti-PDL-1 antibody/IL-10 fusion protein targeting exhausted T cells for cancer immunotherapy. Cancer Res 82:4169. https://doi.org/10.1158/1538-7445.AM2022-4169
Nakao S, Arai Y, Tasaki M et al (2020) Intratumoral expression of IL-7 and IL-12 using an oncolytic virus increases systemic sensitivity to immune checkpoint blockade. Sci Transl Med. 12:eaax7992. https://doi.org/10.1126/scitranslmed.aax7992
Diab A, Tannir NM, Bentebibel SE et al (2020) Bempegaldesleukin (NKTR-214) plus nivolumab in patients with advanced solid tumors: phase I dose-escalation study of safety, efficacy, and immune activation (PIVOT-02). Cancer Discov 10:1158–1173. https://doi.org/10.1158/2159-8290.Cd-19-1510
Wrangle JM, Velcheti V, Patel MR et al (2018) ALT-803, an IL-15 superagonist, in combination with nivolumab in patients with metastatic non-small cell lung cancer: a non-randomised, open-label, phase 1b trial. Lancet Oncol 19:694–704. https://doi.org/10.1016/s1470-2045(18)30148-7
Wrangle JM, Awad MM, Badin FB, Rubinstein MP, Bhar P, Garner C, Reddy SK, Soon-Shiong P (2021) Preliminary data from QUILT 3.055: a phase 2 multi-cohort study of N803 (IL-15 superagonist) in combination with checkpoint inhibitors (CPI). Journal of Clinical Oncology. 39:2596. https://doi.org/10.1200/JCO.2021.39.15_suppl.2596
Patel MR, Naing A, Burris HA et al (2021) A phase 1/2 open-label study of KY1044, an anti-ICOS antibody with dual mechanism of action, as single agent and in combination with atezolizumab, in adult patients with advanced malignancies. J Clin Oncol 39:2624. https://doi.org/10.1200/JCO.2021.39.15_suppl.2624
Weiss SA, Djureinovic D, Jessel S et al (2021) A phase I study of apx005m and cabiralizumab with or without nivolumab in patients with melanoma, kidney cancer, or non-small cell lung cancer resistant to anti-PD-1/PD-L1. Clin Cancer Res 27:4757–4767. https://doi.org/10.1158/1078-0432.Ccr-21-0903
Tolcher AW, Sznol M, Hu-Lieskovan S et al (2017) Phase Ib study of utomilumab (PF-05082566), a 4–1BB/CD137 agonist, in combination with pembrolizumab (MK-3475) in patients with advanced solid tumors. Clin Cancer Res 23:5349–5357. https://doi.org/10.1158/1078-0432.Ccr-17-1243
Piha-Paul SA, Geva R, Tan TJ et al (2021) First-in-human phase I/Ib open-label dose-escalation study of GWN323 (anti-GITR) as a single agent and in combination with spartalizumab (anti-PD-1) in patients with advanced solid tumors and lymphomas. J Immunother Cancer. https://doi.org/10.1136/jitc-2021-002863
Muik A, Garralda E, Altintas I et al (2022) Preclinical Characterization and Phase I Trial Results of a Bispecific Antibody Targeting PD-L1 and 4–1BB (GEN1046) in Patients with Advanced Refractory Solid Tumors. Cancer Discov 12:1248–1265. https://doi.org/10.1158/2159-8290.Cd-21-1345
Huang Z, Pang X, Zhong T et al (2022) AK129, an anti-PD1/LAG-3 bi-specific antibody for cancer therapy. Cancer Res 82:5520. https://doi.org/10.1158/1538-7445.AM2022-5520
Dai S, Huang W, Yuan Z et al (2022) An Fc-competent bispecific antibody targeting PD-L1 and TIGIT induces strong immune responses and potent anti-tumor efficacy. Cancer Res 82:5525. https://doi.org/10.1158/1538-7445.AM2022-5525
Luo H, Meng Z, Rong J et al (2022) Development of a novel PD-L1xCD40 bispecific antibody with excellent efficacy and safety profile for cancer therapy. Cancer Res 82:5559. https://doi.org/10.1158/1538-7445.AM2022-5559
Revuri V, Bhatnagar S, Schultz J, Larson P, Ferguson DM, Panyam J (2022) STING and TLR 7/8 Agonist combination can improve immune checkpoint blockade therapy efficiency. Cancer Res 82:5587. https://doi.org/10.1158/1538-7445.AM2022-5587
Yi M, Zheng X, Niu M, Zhu S, Ge H, Wu K (2022) Combination strategies with PD-1/PD-L1 blockade: current advances and future directions. Molecular Cancer. 21:8. https://doi.org/10.1186/s12943-021-01489-2
Bagchi S, Yuan R, Engleman EG (2021) Immune checkpoint inhibitors for the treatment of cancer: clinical impact and mechanisms of response and resistance. Annu Rev Pathol 16:223–249. https://doi.org/10.1146/annurev-pathol-042020-042741
Sánchez-Magraner L, Gumuzio J, Miles J et al (2023) Functional engagement of the PD-1/PD-L1 complex but not PD-L1 expression is highly predictive of patient response to immunotherapy in non-small-cell lung cancer. J Clin Oncol 41:2561–2570. https://doi.org/10.1200/jco.22.01748
Rizvi NA, Cho BC, Reinmuth N et al (2020) Durvalumab with or without tremelimumab vs standard chemotherapy in first-line treatment of metastatic non-small cell lung cancer: the MYSTIC phase 3 randomized clinical trial. JAMA Oncol 6:661–674. https://doi.org/10.1001/jamaoncol.2020.0237
Le DT, Uram JN, Wang H et al (2015) PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med 372:2509–2520. https://doi.org/10.1056/NEJMoa1500596
Ayers M, Lunceford J, Nebozhyn M et al (2017) IFN-γ-related mRNA profile predicts clinical response to PD-1 blockade. J Clin Invest 127:2930–2940. https://doi.org/10.1172/jci91190
Spigel D, Jotte R, Nemunaitis J et al (2021) Randomized phase 2 studies of checkpoint inhibitors alone or in combination with pegilodecakin in patients with metastatic NSCLC (CYPRESS 1 and CYPRESS 2). J Thorac Oncol 16:327–333. https://doi.org/10.1016/j.jtho.2020.10.001
Shurin MR, Naiditch H, Gutkin DW, Umansky V, Shurin GV (2012) ChemoImmunoModulation: immune regulation by the antineoplastic chemotherapeutic agents. Curr Med Chem 19:1792–1803. https://doi.org/10.2174/092986712800099785
Wolchok JD (2022) Immunotherapy for melanoma: checkpoint blockade combinations. Cancer Immunol Res 10:IA22. https://doi.org/10.1158/2326-6074.TUMIMM21-IA22
Funding
This work was supported by grants from National Natural Science Foundation of China (#8217272, #82370096), Natural Science Foundation of Jiangsu Province (No. BK20210146) and Jinling Hospital Management Project (#22LCYY-LH3, 22LCYY-QH7).
Author information
Authors and Affiliations
Contributions
Yueying Chen: Conceptualization, Methodology, Visualization, Writing—original draft, Writing—review & editing. Hedong Han: Conceptualization, Methodology, Visualization, Writing—original draft, Writing—review & editing. Jing Cheng: Methodology, Visualization, Writing—original draft, Writing—review & editing. Qinpei Cheng: Methodology, Writing—review & editing. Suhua Zhu: Methodology, Writing—review & editing. Ping Zhan: Conceptualization, Writing—review & editing. Hongbing Liu: Conceptualization, Writing—review & editing. Yong Song: Conceptualization, Writing—review & editing. Tangfeng Lv: Conceptualization, Writing—review & editing, Supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Y., Han, H., Cheng, J. et al. Efficacy and safety of anti-PD-1/PD-L1-based dual immunotherapies versus PD-1/PD-L1 inhibitor alone in patients with advanced solid tumor: a systematic review and meta-analysis. Cancer Immunol Immunother 73, 155 (2024). https://doi.org/10.1007/s00262-024-03734-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00262-024-03734-1