Abstract
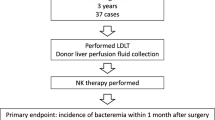
Liver transplantation (LT) is a viable treatment option for cirrhosis patients with hepatocellular carcinoma (HCC). However, recurrence is the rate-limiting factor of long-term survival. To prevent this, we conducted the phase I study of the adoptive transfer of deceased donor liver-derived natural killer (NK) cells. Liver NK cells were extracted from donor liver graft perfusate and were stimulated in vitro with IL-2. The patient received an intravenous infusion of NK cells 3–5 days after LT. Eighteen LT recipients were treated. There were no severe cell infusion-related adverse events or acute rejection episodes. One patient withdrew from the study because the pathological observation revealed sarcoma instead of HCC. All patients who received this immunotherapy completed the follow-up for at least 2 years without evidence of HCC recurrence (median follow-up, 96 months [range, 17–121 months]). Considering that 9 (52.9%) of the 17 patients pathologically exceeded the Milan criteria, liver NK cell infusion is likely to be useful for preventing HCC recurrence after LT. This is the first-in-human immunotherapy study using deceased donor liver-derived NK cells to prevent HCC recurrence after LT. This treatment was well tolerated and resulted in no HCC recurrence after LT.
Clinical trial registration www.clinicaltrials.gov; NCT01147380; registration date: June 17, 2010.




Similar content being viewed by others
Abbreviations
- CIK:
-
Cytokine-induced killer
- CTCAE:
-
Common Terminology Criteria for Adverse Events
- DDLT:
-
Deceased donor liver transplantation
- FCM:
-
Flow cytometry
- GVHD:
-
Graft-versus-host disease
- HCC:
-
Hepatocellular carcinoma
- HLA:
-
Human leukocyte antigen
- KIR:
-
Killer cell immunoglobulin-like receptor
- LDLT:
-
Living donor liver transplantation
- LMNCs:
-
Liver mononuclear cells
- LT:
-
Liver transplantation
- MELD:
-
The Model for End-Stage Liver Disease
- NK:
-
Natural killer
- PBMCs:
-
Peripheral blood mononuclear cells
- TRAIL:
-
Tumor necrosis factor-related apoptosis-inducing ligand
- UNOS:
-
The United Network for Organ Sharing
References
Mazzaferro V, Regalia E, Doci R, Andreola S, Pulvirenti A, Bozzetti F et al (1996) Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med 334(11):693–699. https://doi.org/10.1056/NEJM199603143341104
Ioannou GN, Perkins JD, Carithers RL Jr (2008) Liver transplantation for hepatocellular carcinoma: impact of the MELD allocation system and predictors of survival. Gastroenterology 134(5):1342–1351. https://doi.org/10.1053/j.gastro.2008.02.013
Saab S, Yeganeh M, Nguyen K, Durazo F, Han S, Yersiz H et al (2009) Recurrence of hepatocellular carcinoma and hepatitis B reinfection in hepatitis B surface antigen-positive patients after liver transplantation. Liver Transpl 15(11):1525–1534. https://doi.org/10.1002/lt.21882
Levi DM, Tzakis AG, Martin P, Nishida S, Island E, Moon J et al (2010) Liver transplantation for hepatocellular carcinoma in the model for end-stage liver disease era. J Am Coll Surg 210(5):727–734. https://doi.org/10.1016/j.jamcollsurg.2010.01.007
Horn M, Phebus C, Blatt J (1990) Cancer chemotherapy after solid organ transplantation. Cancer 66(7):1468–1471. https://doi.org/10.1002/1097-0142(19901001)66:7%3c1468::aid-cncr2820660705%3e3.0.co;2-b
Vivarelli M, Cucchetti A, La Barba G, Ravaioli M, Del Gaudio M, Lauro A et al (2008) Liver transplantation for hepatocellular carcinoma under calcineurin inhibitors: reassessment of risk factors for tumor recurrence. Ann Surg 248(5):857–862. https://doi.org/10.1097/SLA.0b013e3181896278
Yokoyama I, Carr B, Saitsu H, Iwatsuki S, Starzl TE (1991) Accelerated growth rates of recurrent hepatocellular carcinoma after liver transplantation. Cancer 68(10):2095–2100. https://doi.org/10.1002/1097-0142(19911115)68:10%3c2095::aid-cncr2820681002%3e3.0.co;2-y
Soderdahl G, Backman L, Isoniemi H, Cahlin C, Hockerstedt K, Broome U et al (2006) A prospective, randomized, multi-centre trial of systemic adjuvant chemotherapy versus no additional treatment in liver transplantation for hepatocellular carcinoma. Transpl Int 19(4):288–294. https://doi.org/10.1111/j.1432-2277.2006.00279.x
Zimmerman, M. A., Ghobrial, R. M., Tong, M. J., Hiatt, J. R., Cameron, A. M., Hong, J., et al. (2008). Recurrence of hepatocellular carcinoma following liver transplantation: a review of preoperative and postoperative prognostic indicators. Arch Surg 143(2):182–188; discussion 188. https://doi.org/10.1001/archsurg.2007.39
Ljunggren HG, Karre K (1990) In search of the “missing self”: MHC molecules and NK cell recognition. Immunol Today 11(7):237–244. https://doi.org/10.1016/0167-5699(90)90097-s
Karre K (2002) NK cells, MHC class I molecules and the missing self. Scand J Immunol 55(3):221–228. https://doi.org/10.1046/j.1365-3083.2002.01053.x
Balch CM, Tilden AB, Dougherty PA, Cloud GA, Abo T (1984) Heterogeneity of natural killer lymphocyte abnormalities in colon cancer patients. Surgery 95(1):63–70
Cai L, Zhang Z, Zhou L, Wang H, Fu J, Zhang S et al (2008) Functional impairment in circulating and intrahepatic NK cells and relative mechanism in hepatocellular carcinoma patients. Clin Immunol 129(3):428–437. https://doi.org/10.1016/j.clim.2008.08.012
Ishiyama K, Ohdan H, Ohira M, Mitsuta H, Arihiro K, Asahara T (2006) Difference in cytotoxicity against hepatocellular carcinoma between liver and periphery natural killer cells in humans. Hepatology 43(2):362–372. https://doi.org/10.1002/hep.21035
Harada N, Shimada M, Okano S, Suehiro T, Soejima Y, Tomita Y et al (2004) IL-12 gene therapy is an effective therapeutic strategy for hepatocellular carcinoma in immunosuppressed mice. J Immunol 173(11):6635–6644. https://doi.org/10.4049/jimmunol.173.11.6635
Hirata M, Kita Y, Saito S, Nishimura M, Ito M, Mizuta K et al (1998) Increase in natural killer cell activity following living-related liver transplantation. Transpl Int 11(Suppl 1):S185-188. https://doi.org/10.1007/s001470050457
Miller JS, Soignier Y, Panoskaltsis-Mortari A, McNearney SA, Yun GH, Fautsch SK et al (2005) Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in patients with cancer. Blood 105(8):3051–3057. https://doi.org/10.1182/blood-2004-07-2974
Ohira M, Ishiyama K, Tanaka Y, Doskali M, Igarashi Y, Tashiro H et al (2009) Adoptive immunotherapy with liver allograft-derived lymphocytes induces anti-HCV activity after liver transplantation in humans and humanized mice. J Clin Invest 119(11):3226–3235. https://doi.org/10.1172/JCI38374
Ohira M, Ohdan H, Mitsuta H, Ishiyama K, Tanaka Y, Igarashi Y et al (2006) Adoptive transfer of TRAIL-expressing natural killer cells prevents recurrence of hepatocellular carcinoma after partial hepatectomy. Transplantation 82(12):1712–1719. https://doi.org/10.1097/01.tp.0000250935.41034.2d
Xu L, Wang J, Kim Y, Shuang ZY, Zhang YJ, Lao XM et al (2016) A randomized controlled trial on patients with or without adjuvant autologous cytokine-induced killer cells after curative resection for hepatocellular carcinoma. Oncoimmunology 5(3):e1083671. https://doi.org/10.1080/2162402X.2015.1083671
Li X, Dai D, Song X, Liu J, Zhu L, Xu W (2014) A meta-analysis of cytokine-induced killer cells therapy in combination with minimally invasive treatment for hepatocellular carcinoma. Clin Res Hepatol Gastroenterol 38(5):583–591. https://doi.org/10.1016/j.clinre.2014.04.010
Lee JH, Lee JH, Lim YS, Yeon JE, Song TJ, Yu SJ et al (2015) Adjuvant immunotherapy with autologous cytokine-induced killer cells for hepatocellular carcinoma. Gastroenterology 148(7):1383-1391 e1386. https://doi.org/10.1053/j.gastro.2015.02.055
Rubnitz JE, Inaba H, Ribeiro RC, Pounds S, Rooney B, Bell T et al (2010) NKAML: a pilot study to determine the safety and feasibility of haploidentical natural killer cell transplantation in childhood acute myeloid leukemia. J Clin Oncol 28(6):955–959. https://doi.org/10.1200/JCO.2009.24.4590
Brand JM, Meller B, Von Hof K, Luhm J, Bahre M, Kirchner H et al (2004) Kinetics and organ distribution of allogeneic natural killer lymphocytes transfused into patients suffering from renal cell carcinoma. Stem Cells Dev 13(3):307–314. https://doi.org/10.1089/154732804323099235
Tanimine N, Tanaka Y, Kobayashi T, Tashiro H, Miki D, Imamura M et al (2014) Quantitative effect of natural killer-cell licensing on hepatocellular carcinoma recurrence after curative hepatectomy. Cancer Immunol Res 2(12):1142–1147. https://doi.org/10.1158/2326-6066.CIR-14-0091
Ohira M, Nishida S, Matsuura T, Muraoka I, Tryphonopoulos P, Fan J et al (2013) Comparative analysis of T-cell depletion method for clinical immunotherapy-anti-hepatitis c effects of natural killer cells via interferon-gamma production. Transplant Proc 45(5):2045–2050. https://doi.org/10.1016/j.transproceed.2013.01.046
Ohira M, Nishida S, Tryphonopoulos P, Tekin A, Selvaggi G, Moon J et al (2012) Clinical-scale isolation of interleukin-2-stimulated liver natural killer cells for treatment of liver transplantation with hepatocellular carcinoma. Cell Transplant 21(7):1397–1406. https://doi.org/10.3727/096368911X627589
Kumar A, Le DT (2016) Hepatocellular Carcinoma Regression After Cessation of Immunosuppressive Therapy. J Clin Oncol 34(10):e90-92. https://doi.org/10.1200/JCO.2013.51.4067
Shimizu S, Tanaka Y, Tazawa H, Verma S, Onoe T, Ishiyama K et al (2016) Fc-gamma receptor polymorphisms predispose patients to infectious complications after liver transplantation. Am J Transplant 16(2):625–633. https://doi.org/10.1111/ajt.13492
Frohn C, Doehn C, Durek C, Bohle A, Schlenke P, Jocham D et al (2000) Feasibility of the adoptive transfusion of allogenic human leukocyte antigen-matched natural killer cells in patients with renal cell carcinoma. J Immunother 23(4):499–504. https://doi.org/10.1097/00002371-200007000-00014
Schulze A, Schirutschke H, Oelschlagel U, Schmitz M, Fussel M, Wassmuth R et al (2008) Altered phenotype of natural killer cell subsets after haploidentical stem cell transplantation. Exp Hematol 36(4):378–389. https://doi.org/10.1016/j.exphem.2007.12.006
Passweg JR, Tichelli A, Meyer-Monard S, Heim D, Stern M, Kuhne T et al (2004) Purified donor NK-lymphocyte infusion to consolidate engraftment after haploidentical stem cell transplantation. Leukemia 18(11):1835–1838. https://doi.org/10.1038/sj.leu.2403524
Sakamoto N, Ishikawa T, Kokura S, Okayama T, Oka K, Ideno M et al (2015) Phase I clinical trial of autologous NK cell therapy using novel expansion method in patients with advanced digestive cancer. J Transl Med 13:277. https://doi.org/10.1186/s12967-015-0632-8
Doskali M, Tanaka Y, Ohira M, Ishiyama K, Tashiro H, Chayama K et al (2011) Possibility of adoptive immunotherapy with peripheral blood-derived CD3(-)CD56+ and CD3+CD56+ cells for inducing antihepatocellular carcinoma and antihepatitis C virus activity. J Immunother 34(2):129–138. https://doi.org/10.1097/CJI.0b013e3182048c4e
Pan G, O’Rourke K, Chinnaiyan AM, Gentz R, Ebner R, Ni J et al (1997) The receptor for the cytotoxic ligand TRAIL. Science 276(5309):111–113. https://doi.org/10.1126/science.276.5309.111
Sheridan JP, Marsters SA, Pitti RM, Gurney A, Skubatch M, Baldwin D et al (1997) Control of TRAIL-induced apoptosis by a family of signaling and decoy receptors. Science 277(5327):818–821. https://doi.org/10.1126/science.277.5327.818
Mikulak J, Bruni E, Oriolo F, Di Vito C, Mavilio D (2019) Hepatic Natural Killer Cells: Organ-Specific Sentinels of Liver Immune Homeostasis and Physiopathology. Front Immunol 10:946. https://doi.org/10.3389/fimmu.2019.00946
Acknowledgements
We would like to thank Oliver Umland for supporting flow cytometry analysis, Drs. Ryosuke Misawa, Taizo Hibi, Koichiro Uchida, Takehiko Dohi, Izumi Carpenter, Ji Fan, David Grant, Panagiotis Tryphonopoulos, Bonnie Blomberg, and Gary Kleiner for study support, and Editage (www.editage.com) for English language editing.
Funding
This study was supported by the research funding the Grant No. 1BG-08 from the Florida Department of Health and the Bankhead-Coley Cancer Research Program (Seigo Nishida), AMED under Grant Number JP21fk0210051 (Hideki Ohdan), and JSPS KAKENHI Grant Number JP20K09104 (Masahiro Ohira).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors of this manuscript have no conflict of interest to disclose as described by the Cancer Immunology, Immunotherapy.
Ethical approval
This phase I clinical trial was approved by the Institutional Review Board of the University of Miami Miller School of Medicine (IRB#20100344) and the Food and Drug Administration, and was registered with ClinicalTrials.gov (NCT01147380). The trial was designed and conducted according to the Declaration of Helsinki.
Informed consent
All patients provided written informed consent before enrolling in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ohira, M., Hotta, R., Tanaka, Y. et al. Pilot study to determine the safety and feasibility of deceased donor liver natural killer cell infusion to liver transplant recipients with hepatocellular carcinoma. Cancer Immunol Immunother 71, 589–599 (2022). https://doi.org/10.1007/s00262-021-03005-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-021-03005-3