Abstract
Purpose
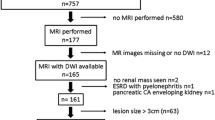
To evaluate magnetic resonance imaging (MRI) criteria of solid renal lesions lower-equal to 2 cm to differentiate benign and malignant tumors, using histopathology as gold standard.
Methods
Three radiologists independently evaluated objective and subjective MRI criteria of focal renal lesions. A total of 105 nodules of patients who had MRI and histopathological results in our institution were included. Subjective criteria evaluated were signal on T2-weighted imaging, presence of microscopic and macroscopic fat, hemosiderin, hemorrhage, central scar, segmented inversion enhancement and enhancement type; objective criteria were gender, ADC value, heterogeneity on T2-weighted imaging and proportion of enhancement in late post-contrast phases. Finally, the readers classified the lesions in probably benign or malignant. Interobserver agreement was evaluated by the Gwet method, and the quantitative variables by intraclass correlation coefficients. To adjust the predictive model, the logistic regression model was used considering the benignity variable as outcome.
Results
A total of 26 nodules (24.5%) were benign and 79 (75.2%) were malignant, with size ranging from 7 to 20 mm (median: 14 mm). The most frequent subtype was papillary renal cell carcinoma (RCC) (35.2%), followed by clear-cell RCC (24.8%) and oncocytoma (12.4%). The univariate and multivariate analysis showed, among all categories evaluated, that microscopic fat (p: 0.072), intermediate (p: 0.004) and hyper-enhancement (p: 0.031) and female sex (p: 0.0047) had the best outcome for benignity, within odds ratios of 4.29, 5.75, 4.07 and 2.86, respectively.
Conclusion
In small solid renal lesions lower-equal to 2 cm, microscopic fat, moderate and hyper-enhancement and female sex were associated with benignity.
Graphic abstract




Similar content being viewed by others
References
Capitanio U, Bensalah K, Bex A, et al. Epidemiology of Renal Cell Carcinoma. Eur Urol 2019;75:74–84
Turner RM II, Morgan TM, Jacobs BL. Epidemiology of the Small Renal Mass and the Treatment Disconnect Phenomenon. Urol Clin North Am 2017;44:147–154
Klaassen Z, Sayyid RK, Wallis CJD. Lessons Learned from the Global Epidemiology of Kidney Cancer: A Refresher in Epidemiology 101. Eur Urol 2019;75:85–87
Zagoria RJ. Imaging of small renal masses: a medical success story. AJR 2000;175:945–955
Cooperberg MR, Mallin K, Ritchey J, Villalta JD, Carroll PR, Kane CJ. Decreasing Size at Diagnosis of Stage 1 Renal Cell Carcinoma: Analysis From the National Cancer Data Base, 1993 to 2004. J Urol 2008;179:2131–2135
Zaid HB, Parker WP, Lohse CM, et al. Patient factors associated with 30-day complications after partial nephrectomy_ A contemporary update. Urol Oncol 2017;35:153.e1–153.e6
Lowrance WT, Yee DS, Savage C, et al. Complications After Radical and Partial Nephrectomy as a Function of Age. J Urol 2010;183:1725–1730
Srougi V, Kato RB, Salvatore FA, Ayres PPM, Dall'Oglio MF, Srougi M. Incidence of benign lesions according to tumor size in solid renal masses. Int Braz J Urol 2009;35:427–431
Frank I, Blute ML, Cheville JC, Lohse CM, Weaver AL, Zincke H. Solid Renal Tumors: An Analysis of Pathological Features Related to Tumor Size. J Urol 2003;170:2217–2220
Soulen MC. Small Renal Masses: Challenges for the Radiologist. Radiology 2018;288:91–92
Thompson RH, Kurta JM, Kaag M, et al. Tumor Size is Associated With Malignant Potential in Renal Cell Carcinoma Cases. J Urol 2009;181:2033–2036
Schieda N, Lim RS, McInnes MDF, et al. Characterization of small (<4 cm) solid renal masses by computed tomography and magnetic resonance imaging: Current evidence and further development. Diag Interv Imaging 2018;99:443–455
Cornelis F, Grenier N. Multiparametric Magnetic Resonance Imaging of Solid Renal Tumors_ A Practical Algorithm. Semin Ultrasound CT MRI 2017;38:47–58
Akdogan B, Gudeloglu A, Inci K, Gunay LM, Koni A, Ozen H. Prevalence and Predictors of Benign Lesions in Renal Masses Smaller Than 7 cm Presumed to be Renal Cell Carcinoma. Clin Genitourin Cancer 2012;10:121–125
Woo S, Cho JY. Imaging Findings of Common Benign Renal Tumors in the Era of Small Renal Masses: Differential Diagnosis from Small Renal Cell Carcinoma: Current Status and Future Perspectives. Korean J Radiol 2015;16:99–115
Leão RRN, Richard PO, Jewett MAS. The role of biopsy for small renal masses. Int J Surg 2016;36:513–517
He Q, Wang H, Kenyon J, et al. Accuracy of Percutaneous Core Biopsy in the Diagnosis of Small Renal Masses (≤ 4.0 cm): A Meta-analysis. Int Braz J Urol 2015;41:15–25
Kay FU, Pedrosa I. Imaging of Solid Renal Masses. Radiol Clin N Am 2017;55:243–258
Wang ZJ, Westphalen AC, Zagoria RJ. CT and MRI of small renal masses. Brit J Radiol 2018;91:20180131.
van Oostenbrugge TJ, Fütterer JJ, Mulders PFA. Diagnostic Imaging for Solid Renal Tumors: A Pictorial Review. KCA 2018;2:79–93
Mauermann J, de Martino M, Waldert M, et al. Gender differences in benign renal masses. World J Urol 2013;31:1051–1057
Acknowledgements
We thank Gisele Cristine Eugenio for helping with the statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
IRB approved and waived of formal consent was acquired.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mussi, T.C., Martins, T., Yamauchi, F.I. et al. Which criteria can be used to predict benignity in solid renal lesions lower-equal to 2 cm?. Abdom Radiol 46, 4873–4880 (2021). https://doi.org/10.1007/s00261-021-03158-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03158-w



