Abstract
Purpose
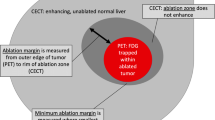
To evaluate 18F-fluorodeoxyglucose (FDG) perfusion PET during FDG PET/CT-guided liver tumor microwave ablation procedures for assessing the ablation margin and correlating minimum margin measurements with local progression.
Methods
This IRB-approved, HIPAA-compliant study included 20 adult patients (11 M, 9 F; mean age 65) undergoing FDG PET/CT-guided liver microwave ablation to treat 31 FDG-avid tumors. Intraprocedural FDG perfusion PET was performed to assess the ablation margin. Intraprocedural decisions regarding overlapping ablations were recorded. Two readers retrospectively interpreted intraprocedural perfusion PET and postprocedural contrast-enhanced MRI. Assessability of the ablation margin and minimum margin measurements were recorded. Imaging follow-up for local progression ranged from 30 to 574 days (mean 310). Regression modeling of minimum margin measurements was performed. Hazard ratios were calculated to correlate an ablation margin threshold of 5 mm with outcomes.
Results
Intraprocedural perfusion PET prompted additional overlapping ablations of two tumors, neither of which progressed. Incomplete ablation or local progression occurred in 8/31 (26%) tumors. With repeat ablation, secondary efficacy was 26 (84%) of 31. Both study readers deemed ablation margins fully assessable more often using perfusion PET than MRI (OR 69.7; CI 6.0, 806.6; p = 0.001). Minimum ablation margins ≥ 5 mm on perfusion PET correlated with a low risk of incomplete ablation/local progression by both study readers (HR 0.08 and 0.02, p < 0.001).
Conclusion
Intraprocedural FDG perfusion PET consistently enabled complete liver tumor microwave ablation margin assessments, and the perfusion PET minimum ablation margin measurements correlated well with local outcomes.
Clinical trial registration clinicaltrials.gov (NCT02018107).





Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are not publicly available as the data are contained within our hospital electronic medical record but are available from the corresponding author on reasonable request.
Code availability (Software application or custom code)
Not applicable.
References
Weinstein JL, Ahmed M. Percutaneous ablation for hepatocellular carcinoma. AJR Am J Roentgenol 2018;210(6):1368-1375 doi: https://doi.org/10.2214/AJR.17.18695
Meijerink MR, Puijk RS, vanTilborg AAJM, Henningsen KH, Fernandez LG, Neyt M, Heymans J, Frankema JS, de Jong KP, Richel DJ, Prevoo W, Vlayen J. Radiofrequency and microwave ablation compared to systemic chemotherapy and to partial hepatectomy in the treatment of colorectal liver metastases: a systematic review and meta-analysis. Cardiovasc Intervent Radiol 2018;41(8):1189-1204 doi: https://doi.org/10.1007/s00270-018-1959-3
Winkelmann MT, Clasen S, Pereira PL, Hoffmann R. Local treatment of oligometastatic disease: current role. Br J Radiol 2019:92(1100):20180835 doi: https://doi.org/10.1259/bjr.20180835
Shady W, Petre EN, Gonen M, et al. Percutaneous radiofrequency ablation of colorectal cancer liver metastasis: Factors affecting outcomes – A 10 -year experience at a single center. Radiology 2016;278(2):601-611 doi: https://doi.org/10.1148/radiol.2015142489
Hocquelet A, Trillaud H, Frulio N, Papadopoulos P, Balageas P, Salut C, Meyer M, Blanc JF, Montaudon M, Denis de Senneville B. Three-dimensional measurement of hepatocellular carcinoma ablation zones and margins for predicting local tumor progression. J Vasc Interv Radiol 2016;27(7):1038-1045 doi: https://doi.org/10.1016/j.jvir.2016.02.031
Yoon JH, Lee JM, Klotz E, Woo H, Yu MH, Joo I, Lee ES, Han JK. Prediction of local tumor progression after radiofrequency ablation (RFA) of hepatocellular carcinoma by assessment of ablative margin using preRFA MRI and post-RFA CT registration. Korean J Radiol 2018;19(6):1053-1065 doi: https://doi.org/10.3348/kjr.2018.19.6.1053
Shady W, Petre EN, Do KG, Gonen M, Yarmohammadi H, Brown KT, Kemeny NE, D’Angelica M, Kingham PT, Solomon SB, Sofocleous CT. Percutaneous microwave versus radiofrequency ablation of colorectal liver metastases: ablation with clear margins (A0) provides the best local control. J Vasc Interv Radiol 2018;29(2):268-275 doi: https://doi.org/10.1016/j.jvir.2017.08.021
Makino Y, Imai Y, Igura T et al. Feasibility of extracted-overlay fusion imaging for intra-operative treatment evaluation of radiofrequency ablation for hepatocellular carcinoma. Liver Cancer 2016;5(4):269-279 doi: https://doi.org/10.1159/000449338
Li K, Su ZZ, Xu EJ, Ju JX, Meng XC, Zheng RQ. Improvement of ablative margins by the intraoperative use of CEUS-CT/MR image fusion in hepatocellular carcinoma. BMC Cancer 2016;16:277 doi: https://doi.org/10.1186/s12885-016-2306-1
Kim YS, Lee WJ, Rhim H, Lim HK, Choi D, Lee JY. The minimal ablative margin of radiofrequency ablation of hepatocellular carcinoma (> 2 and <5 cm) needed to prevent local tumor progression: 3D quantitative assessment using CT image fusion). AJR AM J Roentgenol 2010;195(3):758-765 doi: https://doi.org/10.2214/AJR.09.2954
Kim SM, Shin SS, Lee BC, Kim JW, Heo SH, Lim HS, Jeong YY. Imaging evaluation of ablative margin and index tumor immediately after radiofrequency ablation for hepatocellular carcinoma: comparison between multidetector-row CT and MR imaging. Abdom Radiol 2017;42(10):2527-2537 doi: https://doi.org/10.1007/s00261-017-1146-z
Schaible J, Pregler B, Baumler WW, Einspieler I, Jung EM, Stroszczynski C, Beyer LP. Safety margin assessment after microwave ablation of liver tumors: inter- and intrareader variability. Radiol Oncol. 2020;54(1):57-61 doi:https://doi.org/10.2478/raon-2020-0004
Fukuda K, Mori K, Hasegawa N, Nasu K, Ishige K, Okamoto Y, Shiigai M, Abei M, Minami M, Hyodo I. Safety margin of radiofrequency ablation for hepatocellular carcinoma: a prospective study using magnetic resonance imaging with superparamagnetic iron oxide. Jpn J Radiol 2019:37(7):555-563 doi: https://doi.org/10.1007/s11604-019-00843-1
Solbiati M, Muglia R, Goldberg SN, Ierace T, Rotilio A, Passera KM, Marre I, Solbiati L. A novel software platform for volumetric assessment of ablation completeness. Int J Hyperthermia 2019;36(1):337-343 doi: https://doi.org/10.1080/02656736.2019.1569267
Calandri M, Mauri G, Yevich S, Gazzera C, Basile D, Gatti M, Veltri A, Fonio P. Fusion imaging and virtual navigation to guide percutaneous thermal ablation of hepatocellular carcinoma: a review of the literature. Cardiovasc Intervent Radiol 2019;42(5):639-647 doi: https://doi.org/10.1007/s00270-019-02167-z
Bing F, Vappou J, Breton E, Enescu I, Garmon J, Gangi A. Accuracy of CT-Ultrasound Fusion Imaging Guidance System Used for hepatic percutaneous procedures. J Vasc Interv Radiol 2019;30(7):1013-1020 doi: https://doi.org/10.1016/j.jvir.2018.11.034
Kaye EA, Cornelis FH, Petre EN, Tyagi N, Shady W, Shi W, Zhang Z, Solomon SB, Sofocleous CT, Durack JC. Volumetric 3D assessment of ablation zones after thermal ablation of colorectal liver metastases to improve prediction of local tumor progression. Eur Radiol 2019;29(5):2698-2705 doi: https://doi.org/10.1007/s00330-018-5809-0
Sainani NI, Shyn PB, Tatli S, Morrison PR, Tuncali K, Silverman SG. PET/CT-guided radiofrequency and cryoablation: is tumor fluorine-18 fluorodeoxyglucose activity, dissipated by thermal ablation? J Vasc Interv Radiol 2011;22(3):354–360 doi: https://doi.org/10.1016/j.jvir.2010.11.027
Shyn PB, Casadaban LC, Sainani NI, Sadow CA, Bunch PM, Levesque VM, Kim CK, Gerbaudo VH, Silverman SG. Intraprocedural ablation margin assessment by using ammonia perfusion PET during FDG PET/CT-guided liver tumor ablation: a pilot study. Radiology 2018;288:138–145 doi: https://doi.org/10.1148/radiol.2018172108
Cornelis FH, Petre EN, Vakiani E, Klimstra D, Durack JC, Gonen M, Osborne J, Solomon SB, Sofocleous CT. Immediate postablation 18F-FDG injection and corresponding SUV are surrogate biomarkers of local tumor progression after thermal ablation of colorectal carcinoma liver metastases. J Nucl Med 2018;59(9):1360-1365 doi: https://doi.org/10.2967/jnumed.117.194506
Brace CL, Diaz TA, Hinshaw JL, Lee FT. Tissue contraction caused by radiofrequency and microwave ablation: a laboratory study in liver and lung. J Vasc Interv Radiol 2010;21(8):1280–1286 doi: https://doi.org/10.1016/j.jvir.2010.02.038
Lee JK, Siripongsakun S, Bahrami S, Raman SS, Sayre J, Lu DS. Microwave ablation of liver tumors: degree of tissue contraction as compared to RF ablation. Abdom Radiol 2016;41(4):659-66 doi: https://doi.org/10.1007/s00261-016-0725-8
Kim SM, Koda M, Tokunaga S, Okamoto T, Hodozuka M, Miyoshi K, Kishina M, Fuise Y, Kato J, Matono T, Sugihara T, Oyama K, Hosho K, Okano J, Murawaki Y, Kakite S, Yamashita E. Clinical usefulness of the ablative margin assessed by magnetic resonance imaging with Gd-EOB-DTPA for radiofrequency ablation of hepatocellular carcinoma. J Hepatol 2015;63(6):1360-7 doi: https://doi.org/10.1016/j.jhep.2015.07.023
Funding
Research funding was provided by Siemens Healthineers.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
The Institutional Review Board (IRB) reviewed and approved the study including review of health/medical records. The study was conducted in compliance with the Health Insurance Portability and Accountability Act.
Informed consent
Informed consent was obtained from all study participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shyn, P.B., Cubre, A.J., Catalano, P.J. et al. F-18 FDG perfusion PET: intraprocedural assessment of the liver tumor ablation margin. Abdom Radiol 46, 3437–3447 (2021). https://doi.org/10.1007/s00261-021-02970-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-02970-8