Abstract
Purpose
Classical brachytherapy of solid malignant tumors is an invasive procedure which often results in an uneven dose distribution, while requiring surgical removal of sealed radioactive seed sources after a certain period of time. To circumvent these issues, we report the synthesis of intrinsically radiolabeled and gum Arabic glycoprotein functionalized [169Yb]Yb2O3 nanoseeds as a novel nanoscale brachytherapy agent, which could directly be administered via intratumoral injection for tumor therapy.
Methods
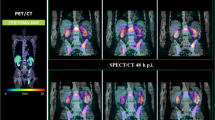
169Yb (T½ = 32 days) was produced by neutron irradiation of enriched (15.2% in 168Yb) Yb2O3 target in a nuclear reactor, radiochemically converted to [169Yb]YbCl3 and used for nanoparticle (NP) synthesis. Intrinsically radiolabeled NP were synthesized by controlled hydrolysis of Yb3+ ions in gum Arabic glycoprotein medium. In vivo SPECT/CT imaging, autoradiography, and biodistribution studies were performed after intratumoral injection of radiolabeled NP in B16F10 tumor bearing C57BL/6 mice. Systematic tumor regression studies and histopathological analyses were performed to demonstrate therapeutic efficacy in the same mice model.
Results
The nanoformulation was a clear solution having high colloidal and radiochemical stability. Uniform distribution and retention of the radiolabeled nanoformulation in the tumor mass were observed via SPECT/CT imaging and autoradiography studies. In a tumor regression study, tumor growth was significantly arrested with different doses of radiolabeled NP compared to the control and the best treatment effect was observed with ~ 27.8 MBq dose. In histopathological analysis, loss of mitotic cells was apparent in tumor tissue of treated groups, whereas no significant damage in kidney, lungs, and liver tissue morphology was observed.
Conclusions
These results hold promise for nanoscale brachytherapy to become a clinically practical treatment modality for unresectable solid cancers.
Graphical Abstract











Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30. https://doi.org/10.3322/caac.21590.
Thun MJ, DeLancey JO, Center MM, Jemal A, Ward EM. The global burden of cancer: priorities for prevention. Carcinogenesis. 2010;31:100–10. https://doi.org/10.1093/carcin/bgp263.
Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17–48. https://doi.org/10.3322/caac.21763.
Cheng T, Peng R, Qu A, Wang H. High-dose rate endorectal brachytherapy for rectal cancer: a state-of-the-art review. Cancer Sci. 2023. https://doi.org/10.1111/cas.15959.
Ladbury C, Harkenrider M, Taunk N, Fisher C, Mayadev J, Venkat P, et al. A practical guide to hybrid interstitial/intracavitary brachytherapy for locally-advanced cervical cancer. Brachytherapy. 2023. https://doi.org/10.1016/j.brachy.2023.07.001.
Mourtada F, Tomiyoshi K, Sims-Mourtada J, Mukai-Sasaki Y, Yagihashi T, Namiki Y, et al. Actinium-225 targeted agents: where are we now? Brachytherapy. 2023;22(6):697–708. https://doi.org/10.1016/j.brachy.2023.06.228.
Wang W, Wang T, Liu Z, He J, Sun X, Zhong W, et al. Practice patterns of adjuvant radiotherapy in women with stage I to II endometrial carcinoma: a real-world multi-institutional analysis in China. BMC Womens Health. 2023;23:417. https://doi.org/10.1186/s12905-023-02548-0.
Seniwal B, Thipe VC, Singh S, Fonseca TCF, Freitas de Freitas L. Recent advances in brachytherapy using radioactive nanoparticles: an alternative to seed-based brachytherapy. Front Oncol. 2021;11:766407. https://doi.org/10.3389/fonc.2021.766407.
Laprise-Pelletier M, Simão T, Fortin MA. Gold nanoparticles in radiotherapy and recent progress in nanobrachytherapy. Adv Healthc Mater. 2018;7:e1701460. https://doi.org/10.1002/adhm.201701460.
Ehlerding EB, Cai W. Smaller agents for larger therapeutic indices: nanoscale brachytherapy with 177Lu-labeled gold nanoparticles. J Nucl Med. 2016;57:834–5. https://doi.org/10.2967/jnumed.116.173278.
Zhang Y, Sheng J, Zhai F, Wang X, Chen L, Shi C, et al. Pioneering iodine-125-labeled nanoscale covalent organic frameworks for brachytherapy. Bioconjug Chem. 2021;32:755–62. https://doi.org/10.1021/acs.bioconjchem.1c00040.
Chakravarty R, Chakraborty S, Guleria A, Shukla R, Kumar C, Vimalnath Nair KV, et al. Facile one-pot synthesis of intrinsically radiolabeled and cyclic RGD conjugated 199Au nanoparticles for potential use in nanoscale brachytherapy. Ind Eng Chem Res. 2018;57:14337–46. https://doi.org/10.1021/acs.iecr.8b02526.
Yook S, Cai Z, Lu Y, Winnik MA, Pignol JP, Reilly RM. Intratumorally injected 177Lu-labeled gold nanoparticles: gold nanoseed brachytherapy with application for neoadjuvant treatment of locally advanced breast cancer. J Nucl Med. 2016;57:936–42. https://doi.org/10.2967/jnumed.115.168906.
Pellico J, Gawne PJ, de Rosales RTM. Radiolabelling of nanomaterials for medical imaging and therapy. Chem Soc Rev. 2021;50:3355–423. https://doi.org/10.1039/d0cs00384k.
Chakravarty R, Goel S, Dash A, Cai W. Radiolabeled inorganic nanoparticles for positron emission tomography imaging of cancer: an overview. Q J Nucl Med Mol Imaging. 2017;61:181–204. https://doi.org/10.23736/s1824-4785.17.02969-7.
Hopfensperger KM, Adams Q, Kim Y, Wu X, Xu W, Patwardhan K, et al. Needle-free cervical cancer treatment using helical multishield intracavitary rotating shield brachytherapy with the 169Yb Isotope. Med Phys. 2020;47:2061–71. https://doi.org/10.1002/mp.14101.
Safaeipour E, Poorbaygi H, Jabbari I, Sheibani S. Evaluation of dosimetric functions for a new 169Yb HDR brachytherapy source. J Appl Clin Med Phys. 2021;22:82–93. https://doi.org/10.1002/acm2.13347.
Taheri ME, Poorbaygi H, Hadadi A, Sheibani S. Dosimetry investigation of a prototype of 169Yb seed brachytherapy for use in circular stapler. Phys Eng Sci Med. 2021;44:525–34. https://doi.org/10.1007/s13246-021-01004-3.
Reynoso FJ, Munro Iii JJ, Cho SH. Technical note: Monte Carlo calculations of the AAPM TG-43 brachytherapy dosimetry parameters for a new titanium-encapsulated Yb-169 source. J Appl Clin Med Phys. 2017;18:193–9. https://doi.org/10.1002/acm2.12111.
Chakravarty R, Guleria A, Jadhav S, Kumar C, Debnath AK, Sarma HD, et al. Bioinspired synthesis of intrinsically 177Lu-labeled hybrid nanoparticles for potential cancer therapy. Ind Eng Chem Res. 2020;59:22492–500. https://doi.org/10.1021/acs.iecr.0c03910.
Chakravarty R, Shetty P, Nair KV, Rajeswari A, Jagadeesan K, Sarma HD, et al. Reactor produced [64Cu] CuCl2 as a PET radiopharmaceutical for cancer imaging: from radiochemistry laboratory to nuclear medicine clinic. Ann Nucl Med. 2020;34:899–910.
Zhang X, Chen F, Turker MZ, Ma K, Zanzonico P, Gallazzi F, et al. Targeted melanoma radiotherapy using ultrasmall 177Lu-labeled α-melanocyte stimulating hormone-functionalized core-shell silica nanoparticles. Biomaterials. 2020;241:119858.
Prasad N, Thombare N, Sharma S, Kumar S. Gum arabic–A versatile natural gum: a review on production, processing, properties and applications. Ind Crops Prod. 2022;187:115304.
Han J, Chen F, Gao C, Zhang Y, Tang X. Environmental stability and curcumin release properties of Pickering emulsion stabilized by chitosan/gum Arabic nanoparticles. Int J Biol Macromol. 2020;157:202–11.
Sharkawy A, Barreiro MF, Rodrigues AE. Preparation of chitosan/gum Arabic nanoparticles and their use as novel stabilizers in oil/water Pickering emulsions. Carbohyd Polym. 2019;224:115190.
Gruber JB, Chirico RD, Westrum EF Jr. Correlation of spectral and heat-capacity Schottky contributions for Dy2O3, Er2O3, and Yb2O3. J Chem Phys. 1982;76:4600–5.
Jinqiu Y, Lei C, Huaqiang H, Shihong Y, Yunsheng H, Hao W. Raman spectra of RE2O3 (RE= Eu, Gd, Dy, Ho, Er, Tm, Yb, Lu, Sc and Y): laser-excited luminescence and trace impurity analysis. J Rare Earths. 2014;32:1–4.
Pandey SD, Samanta K, Singh J, Sharma ND, Bandyopadhyay A. Anharmonic behavior and structural phase transition in Yb2O3. AIP Adv. 2013;3: 122123.
Piz M, Dulian P, Filipek E, Wieczorek-Ciurowa K, Kochmanski P. Characterization of phases in the V2O5–Yb2O3 system obtained by high-energy ball milling and high-temperature treatment. J Mater Sci. 2018;53:13491–500.
Muthulakshmi V, Kumar P, Sundrarajan M. Green synthesis of Ionic liquid mediated Ytterbium oxide nanoparticles by Andrographis Paniculata leaves extract for structural, morphological and biomedical applications. J Environ Chem Eng. 2021;9:105270.
Ibekwe CA, Oyatogun GM, Esan TA, Oluwasegun KM. Synthesis and characterization of chitosan/gum arabic nanoparticles for bone regeneration. Am J Mater Sci Eng. 2017;5:28–36.
Bashir M, Haripriya S. Assessment of physical and structural characteristics of almond gum. Int J Biol Macromol. 2016;93:476–82.
Lu D-Y, Wei X, Cai Q. Mixed valence states of Yb3+/Yb2+ in low-loss (Ba1− xNdx)(Ti1− xYbx) O3 dielectric ceramics. J Alloy Compd. 2021;884: 161049.
Ohno Y. XPS studies of the intermediate valence state of Yb in (YbS) 1.25 CrS2. J Electron Spectros Relat Phenomena. 2008;165:1–4.
Park K-W, Ahn S, Lim S-H, Jin MH, Song J, Yun S-Y, et al. Ytterbium oxide nanodots via block copolymer self-assembly and their efficacy to dye-sensitized solar cells. Appl Surf Sci. 2016;364:573–8.
Smith M, Scudiero L, Espinal J, McEwen J-S, Garcia-Perez M. Improving the deconvolution and interpretation of XPS spectra from chars by ab initio calculations. Carbon. 2016;110:155–71.
Dhenadhayalan N, Mythily R, Kumaran R. Fluorescence spectral studies of gum Arabic: multi-emission of Gum Arabic in aqueous solution. J Lumin. 2014;155:322–9.
Gakamskya D, Gakamskyb A. Intrinsic fluorescence of proteins as a medical diagnostic tool. Spectrosc Eur. 2017;29:6.
Chakravarty R, Chakraborty S. A review of advances in the last decade on targeted cancer therapy using 177Lu: focusing on 177Lu produced by the direct neutron activation route. Am J Nucl Med Mol Imaging. 2021;11:443–75.
Kim J, Bar-Ness D, Si-Mohamed S, Coulon P, Blevis I, Douek P, et al. Assessment of candidate elements for development of spectral photon-counting CT specific contrast agents. Sci Rep. 2018;8:12119. https://doi.org/10.1038/s41598-018-30570-y.
Spahn I, Takács S, Shubin YN, Tárkányi F, Coenen HH, Qaim SM. Cross-section measurement of the 169Tm(p, n) reaction for the production of the therapeutic radionuclide 169Yb and comparison with its reactor-based generation. Appl Radiat Isot. 2005;63:235–9. https://doi.org/10.1016/j.apradiso.2005.03.006.
Tatari M, Goudarzi P, Naik H. Study of the accelerator production of 169Yb radioisotope via various particles nuclear reactions. J Radioanal Nucl Chem. 2021;327:525–32. https://doi.org/10.1007/s10967-020-07515-9.
Manual for reactor produced radioisotopes: international atomic energy agency. 2003 (IAEA-TECDOC-1340) Available online at: https://www.iaea.org/publications/6407/manual-for-reactor-produced-radioisotopes. Accessed 1 Dec 2023.
Gonçalves JP, da Cruz AF, Nunes ÁM, Meneghetti MR, de Barros HR, Borges BS, et al. Biocompatible gum arabic-gold nanorod composite as an effective therapy for mistreated melanomas. Int J Biol Macromol. 2021;185:551–61. https://doi.org/10.1016/j.ijbiomac.2021.06.172.
Hassani A, Azarian MMS, Ibrahim WN, Hussain SA. Preparation, characterization and therapeutic properties of gum arabic-stabilized gallic acid nanoparticles. Sci Rep. 2020;10:17808. https://doi.org/10.1038/s41598-020-71175-8.
Moeendarbari S, Tekade R, Mulgaonkar A, Christensen P, Ramezani S, Hassan G, et al. Theranostic nanoseeds for efficacious internal radiation therapy of unresectable solid tumors. Sci Rep. 2016;6:20614. https://doi.org/10.1038/srep20614.
Shukla R, Chanda N, Zambre A, Upendran A, Katti K, Kulkarni RR, et al. Laminin receptor specific therapeutic gold nanoparticles (198AuNP-EGCg) show efficacy in treating prostate cancer. Proc Natl Acad Sci U S A. 2012;109:12426–31. https://doi.org/10.1073/pnas.1121174109.
Bentivoglio V, Nayak P, Varani M, Lauri C, Signore A. Methods for radiolabeling nanoparticles (part 3): therapeutic use. Biomolecules. 2023;13:241. https://doi.org/10.3390/biom13081241.
Ni D, Jiang D, Ehlerding EB, Huang P, Cai W. Radiolabeling silica-based nanoparticles via coordination chemistry: basic principles, strategies, and applications. Acc Chem Res. 2018;51:778–88. https://doi.org/10.1021/acs.accounts.7b00635.
Price EW, Orvig C. Matching chelators to radiometals for radiopharmaceuticals. Chem Soc Rev. 2014;43:260–90. https://doi.org/10.1039/c3cs60304k.
Di Pasqua AJ, Yuan H, Chung Y, Kim JK, Huckle JE, Li C, et al. Neutron-activatable holmium-containing mesoporous silica nanoparticles as a potential radionuclide therapeutic agent for ovarian cancer. J Nucl Med. 2013;54:111–6. https://doi.org/10.2967/jnumed.112.106609.
Chakravarty R, Valdovinos HF, Chen F, Lewis CM, Ellison PA, Luo H, et al. Intrinsically germanium-69-labeled iron oxide nanoparticles: synthesis and in-vivo dual-modality PET/MR imaging. Adv Mater. 2014;26:5119–23. https://doi.org/10.1002/adma.201401372.
Patra S, Kancharlapalli S, Chakraborty A, Singh K, Kumar C, Guleria A, et al. Chelator-free radiolabeling with theoretical insights and preclinical evaluation of citrate-functionalized hydroxyapatite nanospheres for potential use as radionanomedicine. Ind Eng Chem Res. 2023;62:3194–205. https://doi.org/10.1021/acs.iecr.2c04378.
LuengoMorato Y, Ovejero Paredes K, Lozano Chamizo L, Marciello M, Filice M. Recent advances in multimodal molecular imaging of cancer mediated by hybrid magnetic nanoparticles. Polymers (Basel). 2021;13:2989. https://doi.org/10.3390/polym13172989.
Jennings LE, Long NJ. ‘Two is better than one’--probes for dual-modality molecular imaging. Chem Commun (Camb). 2009;24:3511–24. https://doi.org/10.1039/b821903f.
Li M, Wei W, Barnhart TE, Jiang D, Cao T, Fan K, et al. ImmunoPET/NIRF/Cerenkov multimodality imaging of ICAM-1 in pancreatic ductal adenocarcinoma. Eur J Nucl Med Mol Imaging. 2021;48:2737–48. https://doi.org/10.1007/s00259-021-05216-3.
Rosenkrans ZT, Ferreira CA, Ni D, Cai W. Internally responsive nanomaterials for activatable multimodal imaging of cancer. Adv Healthc Mater. 2021;10:e2000690. https://doi.org/10.1002/adhm.202000690.
Dong YC, Kumar A, Rosario-Berríos DN, Si-Mohamed S, Hsu JC, Nieves LM, et al. Ytterbium nanoparticle contrast agents for conventional and spectral photon-counting CT and their applications for hydrogel imaging. ACS Appl Mater Interfaces. 2022;14:39274–84. https://doi.org/10.1021/acsami.2c12354.
Hu W, Hu J, Guo W, Chen J, Liang S, Qian W, et al. Usage of Yb(OH)CO3 nanoparticles-based computed tomography image in the prediction model of lung biopsy pneumothorax. Cell Mol Biol (Noisy-le-grand). 2022;68:258–69. https://doi.org/10.14715/cmb/2022.68.3.29.
Acknowledgements
The authors are grateful for the financial support from the Bhabha Atomic Research Centre (BARC), the National Institutes of Health (P30 CA014520 and T32 CA009206), and the University of Wisconsin-Madison. The authors from BARC are grateful to Dr. P. K. Mohapatra, Associate Director, Radiochemistry and Isotope Group, BARC, Dr. Sandip Basu, Head, Radiation Medicine Centre (Medical), BARC and Dr. Tapas Das, Head, Radiopharmaceuticals Division, BARC for their support to this work. Dr. A.K. Debnath, Technical Physics Division, BARC, is acknowledged for providing the XPS data. The Sophisticated Analytical Instrumentation Facility of the Indian Institute of Technology Bombay is gratefully acknowledged for the HRTEM analysis of the samples.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval
All animal studies were performed according to the guidelines of the Animal Care and Use Committee of the Bhabha Atomic Research Centre, India.
Conflict of interest
Weibo Cai declares conflict of interest with the following corporations: Actithera, Inc., Rad Source Technologies, Inc., Portrai, Inc., rTR Technovation Corporation, and Four Health Global Pharmaceuticals Inc. All other authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghosh, S., Patra, S., Younis, M.H. et al. Brachytherapy at the nanoscale with protein functionalized and intrinsically radiolabeled [169Yb]Yb2O3 nanoseeds. Eur J Nucl Med Mol Imaging 51, 1558–1573 (2024). https://doi.org/10.1007/s00259-024-06612-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-024-06612-1