Abstract
Objective
T2-relaxometry could differentiate between physiological and haemorrhagic joint effusion (≥ 5% blood) in vitro. Are quantitative T2-relaxation time measurements of synovial fluid feasible and reproducible in vivo in clinically bleed-free joints of men with haemophilia?
Materials and methods
In this cross-sectional study, we measured T2-relaxation times of synovial fluid in clinically bleed-free ankles, knees or elbows of men with severe haemophilia A using a T2-mapping sequence (duration ≤ 7 min) at 3 Tesla MRI. Manual and circular regions of interest (ROI) were drawn in the synovial fluid of each joint by two independent observers to measure T2-relaxation times. Measurement feasibility was expressed as the success rate of the measurements by both observers. The interobserver and intraobserver reproducibility of the measurements were evaluated by the intraclass correlation coefficient of absolute agreement (ICC) and the limits of agreement (LoA) from Bland Altman analysis.
Results
We evaluated 39 clinically bleed-free joints (11 ankles, 12 knees, 16 elbows) of 39 men (median age, 24 years; range 17–33) with severe haemophilia A. The success rate of the T2-measurements was ≥ 90%. Interobserver reliability was good to excellent (manual ROI: ICC = 0.92, 95% CI 0.76–0.97; circular ROI: ICC = 0.82, 95% CI 0.66–0.91) and interobserver agreement was adequate (manual ROI: LoA = 71 ms; circular ROI: LoA = 146 ms). Intraobserver reliability was good to excellent (manual ROI: ICC = 0.78, 95% CI − 0.06–0.94; circular RO: ICC = 0.99, 95% CI 0.98–0.99) and intraobserver agreement was good (manual ROI: LoA = 63 ms; circular ROI: LoA = 41 ms).
Conclusion
T2-relaxometry of synovial fluid in haemophilia patients is feasible with good interobserver and intraobserver reproducibility.
Similar content being viewed by others
Introduction
Haemarthrosis can occur after trauma, or spontaneously in bleeding disorders such as von Willebrand disease and haemophilia [1, 2]. Haemarthrosis can induce joint damage, even after brief exposure to a small amount of intra-articular blood [3,4,5,6]. Recurrent haemarthroses can lead to irreversible arthropathy [3,4,5], causing pain and impaired joint function that reduce quality of life [1, 3, 7].
Large traumatic haemarthroses are usually diagnosed based on clinical symptoms as pain, swelling and function loss [7]. Additionally, haemarthrosis is characterized by joint effusion on imaging. The haemorrhagic effusion may have a complex appearance, sedimentation of blood cells may be seen, and fluid–fluid levels may be observed with extensive intra-articular soft tissue damage or fractures (lipohaemarthrosis) [8, 9].
Diagnosing small haemarthroses, haemarthroses without observed trauma, and haemarthrosis in joints with arthropathy can be difficult. Clinical symptoms are not specific to haemarthrosis and also occur in other joint conditions such as arthropathy [10,11,12,13]. Joint effusion on imaging is not specific either and may be observed in arthropathy flare-ups and arthritis, as in healthy joints [10, 14,15,16,17]. Furthermore, the nature of smaller effusions may be difficult to determine with conventional imaging methods. Conventional T1 and T2 weighted magnetic resonance imaging (MRI) is not sensitive to detecting early small haemarthroses [18, 19]. Therefore, T2*-weighted gradient echo (GRE) sequences are used to visually assess acute haemarthrosis and synovial haemosiderin deposition after haemarthrosis [20]. However, they cannot quantify or may not identify minor or subclinical haemarthroses.
Differentiation between haemarthrosis and other diagnoses is important because they require different treatment [7, 10, 21, 22]. Specifically, people with bleeding disorders need clotting factor replacement therapy to stop haemarthrosis and prevent progression to arthropathy [7].
The current reference standard is joint aspiration, an invasive procedure with a risk of intra-articular infection and bleeding, particularly in people with bleeding disorders [7]. An induced haemarthrosis would result in a false-positive outcome. Therefore, joint aspiration is an imperfect reference standard and a non-invasive alternative would be preferable.
In an in vitro setting, T2-relaxometry MRI can quantitatively differentiate between small volumes of physiological joint effusion and haemorrhagic joint effusion with blood concentrations of ≥ 5% blood [23]. Differentiation was based on differences in T2-relaxation times caused by the T2-shorting effect of iron-containing blood [24]. However, in vivo validation of the experimental T2-relaxometry method is required before its use in patients. Establishing good feasibility and reproducibility of the T2-relaxometry method in bleed-free joints is the first step in in vivo validation.
The primary study objective was to evaluate the feasibility and reproducibility of experimental MRI T2-relaxometry of joint effusion in vivo in clinically bleed-free joints of men with severe haemophilia A. Second, we tested robustness of the T2-measurments and determined normal values for T2-relaxation of synovial fluid without blood.
Materials and methods
Study design and population
The MRI T2-maps in this study were obtained as part of the Detecting Subclinical Joint BlEedinG and INflammation in Haemophilia study (BEGIN study). The BEGIN study was approved by the institutional medical ethical review board and all study participants gave written informed consent. This cross-sectional study investigated signs of subclinical bleeding and inflammation in adolescent and adult men with severe haemophilia A between December 2019 and March 2022 [25, 26].
In the BEGIN study, haemophilia patients born after 1988 who received prophylactic treatment reducing the (joint) bleeding risk were screened for a joint (elbow, knee or ankle) without a clinical history of bleeding. In the subgroup of patients with clinically bleed-free joints, one life-long clinically bleed-free joint per patient was examined using MRI. Clinical MRI images were scored for the presence of joint effusion, haemosiderin and synovial hypertrophy by a musculoskeletal radiologist (WF) with > 10 years of experience using the additive International Prophylaxis Study Group (IPSG) MRI score for haemophilic arthropathy [27]. The additive IPSG MRI score grades effusion, synovial hypertrophy and haemosiderin on a scale from 0 to 3 (0 = absent, 1 = minimal, 2 = moderate or 3 = large). The effusion grade was determined using previously described cut-off values [14]. In addition to the clinical MRI images, T2-maps were obtained in 40 joints for use in the current study. Figure 1 shows a flowchart summarizing the inclusion of patients in the current study.
Magnetic resonance imaging
MRI examinations were performed on three 3 T MRI systems (Philips Achieva, Best, The Netherlands) using joint-specific coils (8-channel small extremity coil for elbows, 16-channel knee coil, 8-channel ankle coil). All three MRI systems were located at the same hospital. Detailed MRI protocols are available in Table 1. MRI protocols included sequences to assess soft tissue (effusion and synovial tissue) and osteochondral (cartilage and bone) pathology, and a GRE sequence to assess haemosiderin deposition according to the IPSG MRI score; these results have been reported elsewhere [25]. In addition, an experimental multi-slice Turbo Spin Echo (TSE) sequence with 32 echoes and SENSitivity Encoding (SENSE) acceleration was included in the imaging protocol for T2 mapping [23]. T2-mapping sequences contained five slices for the knees and three slices for the elbows and ankles. T2-mapping sequence duration was 7 min in the knees and 4 min 12 s in the elbows and ankles.
T2 mapping and T2 measurements
T2-maps were obtained by image processing in Matlab version R2021a (The MathWorks, Inc. Natick, MA, USA). The signal intensity (S) from the data points was voxel-wise fitted using a Levenberg–Marquardt nonlinear least squares method with the fit function S = S0 (e−TE/T2), where S0 represents the spin density, TE the echo time and T2 the T2-relaxation time [23, 28].
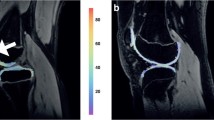
The T2 measurement of synovial fluid was based on the mean T2-relaxation time of a region of interest (ROI) in the synovial fluid. Two types of ROIs were investigated: manual delineation of visible synovial fluid (manual ROI) and placement of a circular ROI within the synovial fluid (circular ROI) as illustrated in Fig. 2. ROIs were placed on the slice of the T2-map showing the most synovial fluid by two independent observers (FL: medical doctor with 4 months training in clinical radiology; JK: medical student with no radiology experience). One observer (JK) placed the ROIs twice in each joint with an interval of 2 weeks between measurements. Minimising the observer’s learning effect of ROI placement on T2-relaxation times, the second round of ratings (JK2) was used to determine interobserver reproducibility and mean values. To verify ROI placement by the relatively inexperienced observers, a musculoskeletal radiologist (WF) reviewed all manual ROIs for correct placement within the synovial fluid.
Statistical analysis
Patient and joint characteristics were reported as median values with ranges for continuous variables and as number with frequencies for dichotomous/categorical variables.
To determine the feasibility of T2 measurements in vivo, we calculated the proportion of joints where both observers successfully performed the measurement (success rate). Corresponding 95% confidence intervals (CI) were calculated using the Clopper-Pearson Exact method [29].
Interobserver and intraobserver reliability of T2 measurements was assessed using the intraclass correlation coefficient of absolute agreement (ICC) [30]. ICC values < 0.5 indicated poor reliability, values between 0.5 and 0.75 indicated moderate reliability, values between 0.75 and 0.9 indicated good reliability, and values > 0.90 indicated excellent reliability. The interobserver and intraobserver limits of agreement (LoA) were determined using Bland Altman analysis [31]. To evaluate interobserver reliability and agreement, the measurements of the first observer (FL) were compared with the second measurements of the second observer (JK2). The smallest detectable change (SDC) was determined to quantify how large the difference between two measurements must be to be detected by the current measurement procedure. The SDC was calculated as SDC = 1.96*√2*SEM agreement. The SEM agreement refers to the standard error of measurement, which equals the square root of the sum of the interobserver/intraobserver variance and the residual variance [30].
Mean T2-relaxation times were calculated from the measurements of the first observer (FL) and the second measurements of the second observer (JK2) and were reported in milliseconds (ms) with standard deviations (sd). The mean T2-relaxation times of the manual and circular ROIs were compared using a paired t-test. To investigate the robustness of the T2-relaxometry method, we investigated whether different joint types and varying amounts of effusion affected the T2 measurements and whether the presence of haemosiderin depositions in the synovial membrane caused significant T2 shortening. Correlations between T2-relaxation time and joint type, the amount of effusion (IPSG MRI scores no/minimal effusion versus moderate/large effusion) and the presence of haemosiderin were determined using multivariate linear regression. All analyses were performed in RStudio version 2022.12.0 + 353 (Posit Software, Boston, MA, USA).
Results
Patients
A flowchart summarizing patient inclusion in the current study is available in Fig. 1. T2-maps were obtained for 40 clinically bleed-free joints. One joint was excluded from evaluation because its MRI data was irreparably damaged during file archiving. Therefore, we ultimately measured T2-relaxation time of synovial fluid in 39 clinically bleed-free joints of 39 patients. All 39 patients had severe haemophilia A and were all male because of the recessive X-linked nature of haemophilia. They all received prophylactic treatment reducing the risk of spontaneous and traumatic (joint) bleeding. The median age was 24 years (range 17–33). Joint characteristics of the evaluated joints are available in Table 2. The 39 joints evaluated included 16 elbows (41%), 12 knees (31%) and 11 ankles (28%). None had a history of overt haemarthrosis. However, 6/39 joints (15%) had haemosiderin deposits in the synovium, indicating previous subclinical bleeding [25]. Concomitant synovial hypertrophy was observed in one ankle with haemosiderin deposits. A physiological amount of synovial fluid (no effusion) was observed in 19/39 joints (49%). There was minimal effusion in 12 joints (31%), moderate effusion in 7 joints (18%), and large effusion in 1 joint (3%).
Feasibility
For all 39 joints, correct placement of the manual ROIs within the synovial fluid was verified by a musculoskeletal radiologist (WF), no ROIs required editing. The success rate of the T2-relaxation measurements was 100% in joints with moderate or large effusion (n = 8/8, CI 69–100%). In joints with no or minimal effusion, the success rate was 87% using the manual ROI (n = 27/31, CI 73–95%) and 94% using the circular ROI (n = 29/31, CI 81–99%). Unsuccessful measurements were due to the absence of joint effusion (n = 1) or minimal joint effusion (n = 3), which hampered placing the ROI.
Reliability
The interobserver and intraobserver reliability results are summarized in Table 3. Interobserver reliability of the T2 measurements was good to excellent (manual ROI ICC = 0.92, CI 0.76–0.97; circular ROI ICC = 0.82, CI 0.66–0.91). Intraobserver reliability was good to excellent (manual ROI ICC = 0.78, CI − 0.06–0.94; circular ROI ICC = 0.99, CI 0.98–0.99).
Agreement
The interobserver and intraobserver agreement results are summarized in Table 3. Figures 3 and 4 show the Bland–Altman plots of interobserver and intraobserver agreement.
Interobserver agreement was adequate (manual ROI LoA = 71 ms; circular ROI LoA = 146 ms), without large systematic interobserver differences (manual ROI mean difference = 23 ms; circular ROI mean difference = 29 ms). The SDC based on the interobserver variance ranged from 84 to 155 ms.
Intraobserver agreement was good (manual ROI LoA = 63 ms; circular ROI LoA = 41 ms). A systematic intraobserver difference (mean difference − 72 ms) was observed for the manual ROI measurements. The second measurements of the second observer (JK2) were systematically lower than the first measurements (JK1) which might indicate a slight learning effect after minimal experience with the T2 measurements. For the circular ROI method, there was no systematic intraobserver difference (mean difference 0 ms). The SDC based on the intraobserver variance ranged from 154 to 40 ms.
T2-relaxation times
Mean T2-relaxation times of the synovial fluid are available in Table 4. Results of the multivariate linear regression on the correlations between T2-relaxation time and joint type, the amount of effusion, and the presence of haemosiderin are available in Supplementary Table 1. Circular ROI measurements of T2 relaxation were higher than manual ROI measurements (p < 0.00). Joint type (elbow, knee, ankle) significantly affected T2 measurements, even when corrected for the amount of effusion in the joint and presence of haemosiderin deposits in the synovial membrane. Figure 5 shows boxplots of T2-relaxation time for different joints. For manual ROIs, T2-relaxation times in the elbows were significantly shorter than T2-relaxation times in the ankles and knees. For circular ROIs, T2-relaxation times were significantly different for all types (p ≤ 0.04). T2-relaxation times were shortest in the elbows, followed by the ankles and knees.
Boxplots showing mean T2-relaxtion times measured with manual and circular regions of interest (ROI) in the different joint types. Asterisk (*) means significant p-values from multivariate linear regression adjusted for amount of effusion and presence of haemosiderin deposits in the synovial membrane. The central thick horizontal lines represent the median values, the boxes contain the 1st to 3rd quartiles. The end of the vertical line represents the minimum and maximum values, excluding outliers. Outliers are shown as dots
Presence of haemosiderin showed a non-significant T2 shortening compared to the absence of haemosiderin (manual ROI − 57 ms, p = 0.20; circular ROI − 74 ms, p = 0.13). T2-relaxation times were not correlated with the amount of effusion (manual ROI p = 0.79; circular ROI p = 0.97).
Discussion
We evaluated the feasibility and reproducibility of MRI T2-relaxometry of non-haemorrhagic joint effusion in vivo using MRI data from 39 clinically bleed-free joints in men with severe haemophilia A. T2-relaxometry measurements were feasible in all joints with moderate or large effusion and ≥ 90% of joints with no or minimal effusion. Both manual and circular ROIs showed good–excellent inter- and intraobserver reliability and agreement. Additionally, we obtained normal values of T2-relaxation time of synovial fluid without blood. Mean T2-relaxation times of synovial fluid were 595 ms (± 102) when effusion was manually delineated and 675 ms (± 125) when a circular ROI was placed within the effusion. T2-relaxation times appeared to vary among different joint types.
Differences in T2-relaxation times for different joints may be due to anatomical difference between joints. For example, knees are larger and have a physiologically larger volume of synovial fluid. This makes it easier to place a ROI in the synovial fluid without including surrounding tissue, making measurements less susceptible to partial volume effects. In addition, it is likely that the volume of the ROI will influence the precision of the measurement and hence the standard deviation. Because of significant differences in T2-relaxation times between joints, joint-specific normal values for synovial fluid T2-relaxation times should be used.
Comparison to previous publications
T2-relaxation times in the current study are consistent with reported T2-relaxation times of synovial fluid in both in vitro and in vivo studies. A previous in vitro study reported a mean T2-relaxation time of 592 ms (± 13) for synovial fluid at 3 Tesla MRI [23], comparable to the mean T2-relaxation times measured with manual ROI (595 ms ± 102) and circular ROI (675 ms ± 125) in the current in vivo study. Two studies measuring T2-relaxation times of synovial fluid in the knees of healthy volunteers reported T2-relaxation times of 767 ms (± 49) [32] and 653 ms (± 113) [33] at 3 Tesla MRI. These are similar to the 684 ms (± 44) in the knees using manual ROI and the 797 ms (± 80) in knees using circular ROI in the current study.
We acknowledge the important work of previous investigators showing conventional T1 and T2 weighted MRI could not qualitatively discriminate physiological synovial fluid or haemorrhagic joint effusion. They report preliminary evidence (mainly ex vivo) that ultrasound is sensitive to small amounts of intra-articular blood [18, 19]. Moreover, GRE sequences are used for visual qualitative differentiation of haemarthrosis in current clinical practice [20]. However, T2-relaxometry may potentially be of interest for detecting small subclinical haemarthrosis in research settings. The previous in vitro results [23] combined with good feasibility and reproducibility of the experimental T2-measurements in vivo suggest the experimental T2-relaxometry method allows quantitative differentiation of physiological synovial fluid from haemorrhagic joint effusion (with low blood concentrations).
Future research and applications
We demonstrated good feasibility and reproducibility of T2-relaxometry in bleed-free joints. T2 measurements failed in a few joints with no or minimal effusion. However, T2-relaxometry will only be relevant in joints suspected of bleeding. This suspicion requires at least some effusion, increasing the probability of a successful measurement. T2 measurements were not significantly affected by haemosiderin deposits. Therefore, T2 measurements remain useful in joints with a history of bleeding.
The 3-slice T2-relaxometry sequence of 4 min 12 s may be feasible in clinical or research settings. The 5-slice sequence for the knee may be shorted by scanning 3 slices at the effusion site only.
Demonstrating good feasibility and reproducibility was the first step in validating the T2-relaxometry method for differentiation between physiological and haemorrhagic joint effusion in vivo. The next step would be confirming T2 shortening of haemorrhagic joint effusion in vivo. This requires a future study including patients with (suspected) haemarthrosis. After confirmation of T2 shortening of haemorrhagic effusion in vivo, T2-relaxometry can serve as a non-invasive alternative to joint aspiration for the diagnosis of haemarthrosis. The T2-relaxation times obtained in the current study can then be used as normal values for synovial fluid without blood. Furthermore, it is known that T2-relaxometry results depend on the magnetic field strength as shown by a previous in vitro study [23]. Future studies are needed to evaluate the test–retest reproducibility of the method and to explore the potential variability of the T2-relaxometry when the measurements are performed in a different institution and/or on different 3 T MR systems from different vendors.
Limitations
Measuring T2-relaxation times of non-haemorrhagic synovial fluid in men with haemophilia could be seen as a limitation, as they do not reflect the healthy general population. However, there is no (patho)physiological indication to assume differences in synovial fluid between our study patients and healthy volunteers. In addition, haemophilia patients are a large part of the target population for non-invasive diagnosis of haemarthrosis. Therefore, it is logical to perform the study in this population.
Lack of joint aspiration as a reference standard for the diagnosis of joint bleeds can be considered another limitation. However, as the patients had no symptoms or history of bleeding in the examined joint, likelihood of haemarthrosis prior to or during MRI is negligible. Furthermore, a false-positive aspiration due to induced haemarthrosis is possible in people with a bleeding disorder and joint aspiration has the risk of inducing intra-articular infection [7]. Therefore, in addition to the ethical concerns, performing joint aspiration in this cohort would have been contraindicated.
ROI placement by relatively inexperienced observers could be seen as a third limitation. However, reliability and agreement were good to excellent despite the lack of experience and a musculoskeletal radiologist verified accurate ROI placement for all joints. Therefore, no prior knowledge or extensive training seems required to perform measurements reliably. Circular ROIs are probably easiest to implement, as circles are easy and quick to place while maintaining good reproducibility. Manual ROIs showed better interrater reproducibility. However, manual delineation is more time-consuming and has a higher risk of interference from the surrounding (haemosiderotic) synovial membrane. Furthermore, its slightly worse intraobserver reproducibility showed potential dependency on observer’s experience, indicating automated joint effusion segmentation can improve the value of manual ROI.
Finally, heterogeneity in joint types, effusion grades and hemosiderin presence in our study population could have confounded results. Yet, only joint type affected T2 measurements significantly. Therefore, we reported joint-specific T2-relaxation times of synovial fluid.
Quantitative MRI T2-relaxometry can be used to measure T2-relaxation times of joint effusions in patients with good inter- and intraobserver reliability and agreement. Both manual delineation of the effusion and placement of a circular ROI within the effusion are feasible and reproducible. Mean T2-relaxation times obtained in this study may be used as reference values for synovial fluid in the elbows, knees and ankles without bleeding.
Data availability
Data is available from the senior author (Dr. W. Foppen, w.foppen@umcutrecht.nl) upon request.
References
Berntorp E, Fischer K, Hart DP, Mancuso ME, Stephensen D, Shapiro AD, et al. Haemophilia. Nat Rev Dis Prim. 2021;7:45.
van Galen KPM, Mauser-Bunschoten EP, Leebeek FWG. Hemophilic arthropathy in patients with von Willebrand disease. Blood Rev. 2012;26:261–6.
Pulles AE, Mastbergen SC, Schutgens REG, Lafeber FPJG, van Vulpen LFD. Pathophysiology of hemophilic arthropathy and potential targets for therapy. Pharmacol Res. 2017;115:192–9.
Jansen NWD, Roosendaal G, Lafeber FPJG. Understanding haemophilic arthropathy: an exploration of current open issues. Br J Haematol. 2008;143:632–40.
Roosendaal G, Lafeber FP. Pathogenesis of haemophilic arthropathy. Haemophilia. 2006;12:117–21.
Jansen NWD, Roosendaal G, Bijlsma JWJ, DeGroot J, Lafeber FPJG. Exposure of human cartilage tissue to low concentrations of blood for a short period of time leads to prolonged cartilage damage: An in vitro study. Arthritis Rheum. 2007;56:199–207.
Srivastava A, Santagostino E, Dougall A, Kitchen S, Sutherland M, Pipe SW, et al. WFH guidelines for the management of hemophilia, 3rd ed. Haemophilia. 2020;26(Suppl 3):1–158.
Costa DN, Cavalcanti CFA, Sernik RA. Sonographic and CT Findings in Lipohemarthrosis. Am J Roentgenol. 2007;188:W389–W389.
Schick C, Mack MG, Marzi I, Vogl TJ. Lipohemarthrosis of the knee: MRI as an alternative to the puncture of the knee joint. Eur Radiol. 2003;13:1185–7.
Timmer MA, Pisters MF, de Kleijn P, de Bie RA, Fischer K, Schutgens RE. Differentiating between signs of intra-articular joint bleeding and chronic arthropathy in haemophilia: A narrative review of the literature. Haemophilia. 2015;21:289–96.
Ceponis A, Wong-Sefidan I, Glass CS, von Drygalski A. Rapid musculoskeletal ultrasound for painful episodes in adult haemophilia patients. Haemophilia. 2013;19:790–8.
Margaretten ME, Kohlwes J, Moore D, Bent S. Does this adult patient have septic arthritis? JAMA. 2007;297(13):1478–88.
Hawker GA, Stewart L, French MR, Cibere J, Jordan JM, March L, et al. Understanding the pain experience in hip and knee osteoarthritis – an OARSI/OMERACT initiative. Osteoarthr Cartil. 2008;16:415–22.
Foppen W, van der Schaaf IC, Witkamp TD, Fischer K. Is joint effusion on MRI specific for haemophilia? Haemophilia. 2014;20:582–6.
Eerdekens M, Peerlinck K, Staes F, Pialat J, Hermans C, Lobet S, et al. Clinical gait features are associated with MRI findings in patients with haemophilic ankle arthropathy. Haemophilia. 2020;26:333–9.
Lundin B, Ljung R, Pettersson H. MRI scores of ankle joints in children with haemophilia - comparison with clinical data. Haemophilia. 2005;11:116–22.
Burke CJ, Alizai H, Beltran LS, Regatte RR. MRI of synovitis and joint fluid. J Magn Reson Imaging. 2019;49:1512–27.
Nguyen S, Lu X, Ma Y, Du J, Chang EY, von Drygalski A. Musculoskeletal ultrasound for intra-articular bleed detection: a highly sensitive imaging modality compared with conventional magnetic resonance imaging. J Thromb Haemost. 2018;16:490–9.
Regi SS, Livingstone RS, Kandagaddala M, Poonnoose P, Gibikote S, Keshava SN, et al. Ultrasound and magnetic resonance imaging for the detection of blood: An ex-vivo study. Haemophilia. 2021;27:488–93.
Doria AS. State-of-the-art imaging techniques for the evaluation of haemophilic arthropathy: present and future. Haemophilia. 2010;16:107–14.
Kolasinski SL, Neogi T, Hochberg MC, Oatis C, Guyatt G, Block J, et al. 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip, and Knee. Arthritis Rheumatol. 2020;72:220–33.
Sharff KA, Richards EP, Townes JM. Clinical Management of Septic Arthritis. Curr Rheumatol Rep. 2013;15:332.
van Leeuwen FHP, Lena B, Zwanenburg JJM, van Vulpen LFD, Bartels LW, Fischer K, et al. Detecting low blood concentrations in joints using T1 and T2 mapping at 1.5, 3, and 7 T: an in vitro study. Eur Radiol Exp. 2021;5:51.
Clark RA, Watanabe AT, Bradley WG, Roberts JD. Acute hematomas: effects of deoxygenation, hematocrit, and fibrin-clot formation and retraction on T2 shortening. Radiology. 1990;175:201–6.
van Leeuwen FHP, van Bergen EDP, Timmer MA, van Vulpen LFD, Schutgens REG, de Jong PA, et al. Magnetic resonance imaging evidence for subclinical joint bleeding in a Dutch population of people with severe hemophilia on prophylaxis. J Thromb Haemost. 2023;21:1156–63.
van Bergen EDP, van Leeuwen FHP, Foppen W, Timmer MA, Schutgens REG, Mastbergen SC, et al. Subclinical synovial proliferation in patients with severe haemophilia A: the value of ultrasound screening and biochemical markers. Haemophilia. 2023;29(6):1580–8.
Lundin B, Manco-Johnson ML, Ignas DM, Moineddin R, Blanchette VS, Dunn AL, et al. An MRI scale for assessment of haemophilic arthropathy from the International Prophylaxis Study Group. Haemophilia. 2012;18:962–70.
Brown RW, Cheng YCN, Haacke EM, Thompson MR, Venkatesan R. 8 Introductory Signal Acquisition Methods: Free Induction Decay, Spin Echoes, Inversion Recovery, and Spectroscopy. Magn Reson imaging Phys Princ Seq Des. 2nd ed. Hoboken: John Wiley & Sons, Inc; 2014. p. 113–36.
Clopper CJ, Pearson ES. The use of confidence or fiducial limits illustrated in the case of the binomial. Biometrika. 1934;26:404–13.
de Vet HCW, Terwee CB, Knol DL, Bouter LM. When to use agreement versus reliability measures. J Clin Epidemiol. 2006;59:1033–9.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet (London, England). 1986;1:307–10.
Gold GE, Han E, Stainsby J, Wright G, Brittain J, Beaulieu C. Musculoskeletal MRI at 3.0 T. Am J Roentgenol. 2004;183:343–51.
Jordan CD, Saranathan M, Bangerter NK, Hargreaves BA, Gold GE. Musculoskeletal MRI at 3.0 T and 7.0 T: A comparison of relaxation times and image contrast. Eur J Radiol. 2013;82:734–9.
Acknowledgements
We would like to thank Niels Blanken for his technical support in adding our experimental T2-mapping sequence to the imaging protocol of the BEGIN-study.
Funding
This study was funded by an independent clinical research grant from NovoNordisk (Access to Insight 2018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This study was funded by an independent clinical research grant from NovoNordisk (Access to Insight 2018) received by W. Foppen which was paid to the institution. M.A. Timmer, and L.F.D. van Vulpen have also received research grants from NovoNordisk. K. Fischer has performed consultancy for NovoNordisk and has received speaker’s fees and research support from NovoNordisk. The other authors declare no financial relationship with NovoNordisk.
In addition, F.H.P. van Leeuwen, B. Lena, E.D.P. van Bergen, J.J. van Klei and C. Bos declare no conflicts of interest. M.A. Timmer has received research grants from SOBI, which were paid to the institution. L.F.D. van Vulpen has received research grants from CSL Behring and Grifols; and has performed consultancy for Sobi, CSL Behring, and Octapharma; all fees paid to the institution. K. Fischer has received speaker’s fees from Bayer, Baxter/Shire, Sobi/Biogen, CSL Behring; has performed consultancy for Bayer, Biogen, CSL Behring, Freeline, Roche and Sobi; and has received research support from Bayer, Baxter/Shire, Pfizer and Biogen; all fees were paid to the institution. P.A. de Jong declares a research collaboration with Vifor Pharma. W. Foppen has received a research grant from Pfizer, which was paid to the institution, and consultancy fees from Pfizer. The Department of Radiology & Nuclear Medicine of the UMC Utrecht has received research support from Philips Healthcare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
van Leeuwen, F.H.P., Lena, B., van Bergen, E.D.P. et al. Quantitative MRI assessment of joint effusion using T2-relaxometry at 3 Tesla: a feasibility and reproducibility study. Skeletal Radiol (2024). https://doi.org/10.1007/s00256-024-04652-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00256-024-04652-0