Abstract
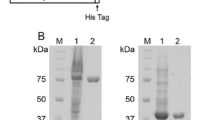
Immediately (17 bp) upstream of the Clostridium paraputrificum chiB gene [J. Bacteriol. 179: 7306–7314 (1997)], we found another chitinase gene chiA encoding chitinase A (ChiA). The chiA gene consists of an open reading frame of 2496 nucleotides and encodes 832 amino acids with a deduced molecular mass of 92 585 Da. The mature ChiA is a modular enzyme composed of a family-18 catalytic domain responsible for chitinase activity, two cadherin-like domains, and a chitin-binding domain. The domain organization of ChiA is fundamentally identical to that of ChiB and the overall sequence identity between them is 35.4%. ChiA was purified from the periplasm fraction of Escherichia coli harboring the chiA gene. The molecular mass of purified ChiA (89 000 Da), determined by sodium dodecyl sulfate/polyacrylamide gel electrophoresis analysis, was in good agreement with the value (89 119 Da) calculated from the deduced amino acid sequence, excluding the signal peptide. Immunological and N-terminal amino acid sequence analyses revealed that ChiA and ChiB are major chitinases of C. paraputrificum and their production is inducible by ball-milled chitin. Northern blot analysis indicated that the chiA and chiB genes constitute a polycistronic operon. Primer-extension analysis confirmed that the transcription of this operon starts upstream of chiA.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 12 August 1998 / Received revision: 16 October 1998 / Accepted: 7 November 1998
Rights and permissions
About this article
Cite this article
Morimoto, K., Karita, S., Kimura, T. et al. Sequencing, expression, and transcription analysis of the Clostridium paraputrificum chiA gene encoding chitinase ChiA. Appl Microbiol Biotechnol 51, 340–347 (1999). https://doi.org/10.1007/s002530051400
Issue Date:
DOI: https://doi.org/10.1007/s002530051400