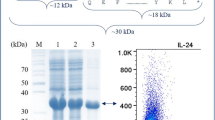
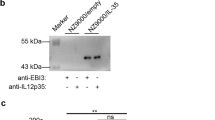
Abstract
Interleukin-18 binding protein (IL-18BP) is a natural IL-18 inhibitor in vivo, which can effectively neutralize IL-18 and inhibit the inflammatory signaling pathway induced by IL-18, thus playing an anti-inflammatory role. Traditional production methods primarily rely on eukaryotic animal cell expression systems, which often entail complex processes, lower yields, and increase production costs. In this study, we present a novel approach for expressing IL-18BP fusion protein using the Escherichia coli (E. coli) system. The N-terminal segment of IL-18BP was fused with the small ubiquitin-related modifier (SUMO) tag, enabling soluble expression, while the C-terminal segment was fused with the human IgG1 Fc fragment to prolong its in vivo lifespan. Through screening, we obtained a high-expression engineering strain from a single colony and developed optimized protocols for fermentation and purification of the recombinant SUMO-IL-18BP-Fc protein. The SUMO tag was subsequently cleaved using SUMO protease, and the purified recombinant human IL-18BP-Fc (rhIL-18BP-Fc) exhibited a purity exceeding 90% with a yield of 1 g per liter of bacterial solution. The biological activities and underlying mechanisms of rhIL-18BP-Fc were evaluated using cell lines and a mouse model. Our results demonstrated that rhIL-18BP-Fc effectively inhibited IL-18-stimulated IFN-γ production in KG-1a cells in vitro and ameliorated DSS-induced ulcerative colitis in mice. In conclusion, we successfully employed the SUMO fusion system to achieve high-level production, soluble expression, and prolonged activity of rhIL-18BP-Fc in E. coli. These findings lay the groundwork for future large-scale industrial production and pharmaceutical development of rhIL-18BP-Fc protein.
Key points
• Effective expression, fermentation, and purification of bioactive rhIL-18BP-Fc protein in E. coli.
• The rhIL-18BP-Fc protein has a great potential for the therapy of ulcerative colitis by inhibiting the expression of inflammatory factors.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Baker KJ, Houston A, Brint E (2019) IL-1 family members in cancer; two sides to every story. Front Immunol 10:1197. https://doi.org/10.3389/fimmu.2019.01197
Dinarello CA, Novick D, Kim S, Kaplanski G (2013) Interleukin-18 and IL-18 binding protein. Front Immunol 4:289. https://doi.org/10.3389/fimmu.2013.00289
Faggioni R, Cattley RC, Guo J, Flores S, Brown H, Qi M, Yin S, Hill D, Scully S, Chen C, Brankow D, Lewis J, Baikalov C, Yamane H, Meng T, Martin F, Hu S, Boone T, Senaldi G (2001) IL-18-binding protein protects against lipopolysaccharide-induced lethality and prevents the development of Fas/Fas ligand-mediated models of liver disease in mice. J Immunol 167:5913–5920. https://doi.org/10.4049/jimmunol.167.10.5913
Gabay C, Fautrel B, Rech J, Spertini F, Feist E, Kötter I, Hachulla E, Morel J, Schaeverbeke T, Hamidou MA, Martin T, Hellmich B, Lamprecht P, Schulze-Koops H, Courvoisier DS, Sleight A, Schiffrin EJ (2018) Open-label, multicentre, dose-escalating phase II clinical trial on the safety and efficacy of tadekinig alfa (IL-18BP) in adult-onset Still’s disease. Ann Rheum Dis 77:840–847. https://doi.org/10.1136/annrheumdis-2017-212608
Ghayur T, Banerjee S, Hugunin M, Butler D, Herzog L, Carter A, Quintal L, Sekut L, Talanian R, Paskind M, Wong W, Kamen R, Tracey D, Allen H (1997) Caspase-1 processes IFN-gamma-inducing factor and regulates LPS-induced IFN-gamma production. Nature 386:619–623. https://doi.org/10.1038/386619a0
González L, Rivera K, Andia ME, Martínez Rodriguez G (2022) The IL-1 family and its role in atherosclerosis. Int J Mol Sci 24:17. https://doi.org/10.3390/ijms24010017
Guo C, Ran Q, Sun C, Zhou T, Yang X, Zhang J, Pang S, Xiao Y (2020) Loss of FGFR3 delays acute myeloid leukemogenesis by programming weakly pathogenic CD117-positive leukemia stem-like cells. Front Pharmacol 11:632809. https://doi.org/10.3389/fphar.2020.632809
Harper S, Speicher DW (2011) Purification of proteins fused to glutathione S-transferase. Methods Mol Biol 681:259–280. https://doi.org/10.1007/978-1-60761-913-0_14
Hodson R (2016) Inflammatory bowel disease. Nature 540:S97. https://doi.org/10.1038/540S97a
Kaplanski G (2018) Interleukin-18: biological properties and role in disease pathogenesis. Immunol Rev 281:138–153. https://doi.org/10.1111/imr.12616
Kim JJ, Shajib MS, Manocha MM, Khan WI (2012) Investigating intestinal inflammation in DSS-induced model of IBD. J Vis Exp:3678. https://doi.org/10.3791/3678
Kimura T, Kato Z, Ohnishi H, Tochio H, Shirakawa M, Kondo N (2008) Expression, purification and structural analysis of human IL-18 binding protein: a potent therapeutic molecule for allergy. Allergol Int 57:367–376. https://doi.org/10.2332/allergolint.O-08-546
Krawisz JE, Sharon P, Stenson WF (1984) Quantitative assay for acute intestinal inflammation based on myeloperoxidase activity. Assessment of inflammation in rat and hamster models. Gastroenterology 87:1344–1350
Lee CD, Sun HC, Hu SM, Chiu CF, Homhuan A, Liang SM, Leng CH, Wang TF (2008) An improved SUMO fusion protein system for effective production of native proteins. Protein Sci 17:1241–1248. https://doi.org/10.1110/ps.035188.108
Luo YP, Jiang L, Kang K, Fei DS, Meng XL, Nan CC, Pan SH, Zhao MR, Zhao MY (2014) Hemin inhibits NLRP3 inflammasome activation in sepsis-induced acute lung injury, involving heme oxygenase-1. Int Immunopharmacol 20:24–32. https://doi.org/10.1016/j.intimp.2014.02.017
Mallat Z, Corbaz A, Scoazec A, Besnard S, Lesèche G, Chvatchko Y, Tedgui A (2001) Expression of interleukin-18 in human atherosclerotic plaques and relation to plaque instability. Circulation 104:1598–1603. https://doi.org/10.1161/hc3901.096721
Mazodier K, Marin V, Novick D, Farnarier C, Robitail S, Schleinitz N, Veit V, Paul P, Rubinstein M, Dinarello CA, Harlé JR, Kaplanski G (2005) Severe imbalance of IL-18/IL-18BP in patients with secondary hemophagocytic syndrome. Blood 106:3483–3489. https://doi.org/10.1182/blood-2005-05-1980
McElwain L, Phair K, Kealey C, Brady D (2022) Current trends in biopharmaceuticals production in Escherichia coli. Biotechnol Lett 44:917–931. https://doi.org/10.1007/s10529-022-03276-5
Min HK, Kim S, Lee JY, Kim KW, Lee SH, Kim HR (2021) IL-18 binding protein suppresses IL-17-induced osteoclastogenesis and rectifies type 17 helper T cell/regulatory T cell imbalance in rheumatoid arthritis. J Transl Med 19:392. https://doi.org/10.1186/s12967-021-03071-2
Nakamura K, Okamura H, Nagata K, Komatsu T, Tamura T (1993) Purification of a factor which provides a costimulatory signal for gamma interferon production. Infect Immun 61:64–70. https://doi.org/10.1128/iai.61.1.64-70.1993
Novick D, Kim SH, Fantuzzi G, Reznikov LL, Dinarello CA, Rubinstein M (1999) Interleukin-18 binding protein: a novel modulator of the Th1 cytokine response. Immunity 10:127–136. https://doi.org/10.1016/s1074-7613(00)80013-8
Nowarski R, Jackson R, Gagliani N, de Zoete MR, Palm NW, Bailis W, Low JS, Harman CC, Graham M, Elinav E, Flavell RA (2015) Epithelial IL-18 equilibrium controls barrier function in colitis. Cell 163:1444–1456. https://doi.org/10.1016/j.cell.2015.10.072
Okamura H, Tsutsi H, Komatsu T, Yutsudo M, Hakura A, Tanimoto T, Torigoe K, Okura T, Nukada Y, Hattori K, Akita K, Namba M, Tanabe F, Konishi K, Fukuda S, Kurimoto M (1995) Cloning of a new cytokine that induces IFN-gamma production by T cells. Nature 378:88–91. https://doi.org/10.1038/378088a0
Park SY, Hisham Y, Shin HM, Yeom SC, Kim S (2022) Interleukin-18 binding protein in immune regulation and autoimmune diseases. Biomedicines 10:1750. https://doi.org/10.3390/biomedicines10071750
Raran-Kurussi S, Waugh DS (2017) Expression and purification of recombinant proteins in Escherichia coli with a His(6) or dual His(6)-MBP tag. Methods Mol Biol 1607:1–15. https://doi.org/10.1007/978-1-4939-7000-1_1
Rosano GL, Morales ES, Ceccarelli EA (2019) New tools for recombinant protein production in Escherichia coli: a 5-year update. Protein Sci 28:1412–1422. https://doi.org/10.1002/pro.3668
Samuel RVM, Farrukh SY, Rehmat S, Hanif MU, Ahmed SS, Musharraf SG, Durrani FG, Saleem M, Gul R (2018) Soluble production of human recombinant VEGF-A121 by using SUMO fusion technology in Escherichia coli. Mol Biotechnol 60:585–594. https://doi.org/10.1007/s12033-018-0094-3
Sedimbi SK, Hägglöf T, Karlsson MC (2013) IL-18 in inflammatory and autoimmune disease. Cell Mol Life Sci 70:4795–4808. https://doi.org/10.1007/s00018-013-1425-y
Sirota JC, Walcher A, Faubel S, Jani A, McFann K, Devarajan P, Davis CL, Edelstein CL (2013) Urine IL-18, NGAL, IL-8 and serum IL-8 are biomarkers of acute kidney injury following liver transplantation. BMC Nephrol 14:17. https://doi.org/10.1186/1471-2369-14-17
Suda T, Zoshima T, Takeji A, Suzuki Y, Mizushima I, Yamada K, Nakashima A, Yachie A, Kawano M (2020) Elderly-onset Still’s disease complicated by macrophage activation syndrome: a case report and review of the literature. Intern Med 59:721–728. https://doi.org/10.2169/internalmedicine.3727-19
Williams MA, O'Callaghan A, Corr SC (2019) IL-33 and IL-18 in inflammatory bowel disease etiology and microbial interactions. Front Immunol 10:1091. https://doi.org/10.3389/fimmu.2019.01091
Wirtz S, Popp V, Kindermann M, Gerlach K, Weigmann B, Fichtner-Feigl S, Neurath MF (2017) Chemically induced mouse models of acute and chronic intestinal inflammation. Nat Protoc 12:1295–1309. https://doi.org/10.1038/nprot.2017.044
Wynn JL, Wilson CS, Hawiger J, Scumpia PO, Marshall AF, Liu JH, Zharkikh I, Wong HR, Lahni P, Benjamin JT, Plosa EJ, Weitkamp JH, Sherwood ER, Moldawer LL, Ungaro R, Baker HV, Lopez MC, McElroy SJ, Colliou N et al (2016) Targeting IL-17A attenuates neonatal sepsis mortality induced by IL-18. Proc Natl Acad Sci USA 113:E2627–E2635. https://doi.org/10.1073/pnas.1515793113
Yang YC, Chen SN, Gan Z, Huang L, Li N, Wang KL, Nie P (2023) Functional characterization of IL-18 receptor subunits, IL-18Rα and IL-18Rβ, and its natural inhibitor, IL-18 binding protein (IL-18BP) in rainbow trout Oncorhynchus mykiss. Dev Comp Immunol 140:104610. https://doi.org/10.1016/j.dci.2022.104610
Zhang LM, Zhang J, Zhang Y, Wang L, Fei C, Yi ZW, Dong L (2018) Interleukin-18 binding protein attenuates lipopolysaccharide-induced acute lung injury in mice via suppression NF-κB and activation Nrf2 pathway. Biochem Biophys Res Commun 505:837–842. https://doi.org/10.1016/j.bbrc.2018.09.193
Funding
This work was supported by the Guangdong Provincial Department of Education (2021ZDZX2034) and the Guangdong Basic and Applied Basic Research Foundation (2022A1515010028).
Author information
Authors and Affiliations
Contributions
L.G. and X.C. performed experiments, analyzed figures, and wrote the manuscript. N.T. provided materials. H.Z. and W.L. performed partial experiments. J.Z. and Y.X. conceived the project, supervised experiments, analyzed data, and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(PDF 417 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, L., Chen, X., Zeng, H. et al. Production of recombinant human long-acting IL-18 binding protein: inhibitory effect on ulcerative colitis in mice. Appl Microbiol Biotechnol 107, 7135–7150 (2023). https://doi.org/10.1007/s00253-023-12806-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12806-8