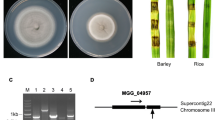
Abstract
Deubiquitination is an essential regulatory step in the Ub-dependent pathway. Deubiquitinating enzymes (DUBs) mediate the removal of ubiquitin moieties from substrate proteins, which are involved in many regulatory mechanisms. As a component of the DUB module (Ubp8/Sgf11/Sus1/Sgf73) in the SAGA (Spt-Ada-Gcn5-acetyltransferase) complex, Ubp8 plays a crucial role in both Saccharomyces cerevisiae and humans. In S. cerevisiae, Ubp8-mediated deubiquitination regulates transcriptional activation processes. To investigate the contributions of Ubp8 to physiological and pathological development of filamentous fungi, we generated the deletion mutant of ortholog MoUBP8 (MGG-03527) in Magnaporthe oryzae (syn. Pyricularia oryzae). The ΔMoubp8 strain showed reduced sporulation, pathogenicity, and resistance to distinct stresses. Even though the conidia of the ΔMoubp8 mutant were delayed in appressorium formation, the normal and abnormal (none-septum or one-septum) conidia could finally form appressoria. Reduced melanin in the ΔMoubp8 mutant is highly responsible for the attenuated pathogenicity since the appressoria of the ΔMoubp8 mutant was much more fragile than those of the wild type, due to the defective turgidity. The weakened ability to detoxify or scavenge host-derived reactive oxygen species (ROS) further restricted the invasion of the pathogen. We also showed that carbon derepression, on the one hand, rendered the ΔMoubp8 strain highly sensitive to allyl alcohol, on the other hand, it enhances the resistance of the MoUBP8 defective strain to deoxyglucose. Overall, we suggest that MoUbp8 is not only required for sporulation, melanin formation, appressoria development, and pathogenicity but also involved in carbon catabolite repression of M. oryzae.









Similar content being viewed by others
References
Adnan M, Zheng W, Islam W, Arif M, Abubakar YS, Wang Z, Lu G (2017) Carbon catabolite repression in filamentous fungi. Int J Mol Sci 19(1):48. https://doi.org/10.3390/ijms19010048
Akutsu M, Dikic I, Bremm A (2016) Ubiquitin chain diversity at a glance. J Cell Sci 129(5):875–880. https://doi.org/10.1242/jcs.183954
Alam MA, Kamlangdee N, Kelly JM (2017) The CreB deubiquitinating enzyme does not directly target the CreA repressor protein in Aspergillus nidulans. Curr Genet 63(4):647–667. https://doi.org/10.1007/s00294-016-0666-3
Amerik A, Swaminathan S, Krantz BA, Wilkinson KD, Hochstrasser M (1997) In vivo disassembly of free polyubiquitin chains by yeast Ubp14 modulates rates of protein degradation by the proteasome. EMBO J 16(16):4826–4838. https://doi.org/10.1093/emboj/16.16.4826
Boase NA, Kelly JM (2004) A role for creD, a carbon catabolite repression gene from Aspergillus nidulans, in ubiquitination. Mol Microbiol 53(3):929–940. https://doi.org/10.1111/j.1365-2958.2004.04172.x
Canzonetta C, Vernarecci S, Iuliani M, Marracino C, Belloni C, Ballario P, Filetici P (2015) SAGA DUB-Ubp8 deubiquitylates centromeric histone variant Cse4. G3 (Bethesda) 6(2):287–298. https://doi.org/10.1534/g3.115.024877
Clague MJ, Barsukov I, Coulson JM, Liu H, Rigden DJ, Urbe S (2013) Deubiquitylases from genes to organism. Physiol Rev 93(3):1289–1315. https://doi.org/10.1152/physrev.00002.2013
Dambacher CM, Worden EJ, Herzik MA, Martin A, Lander GC (2016) Atomic structure of the 26S proteasome lid reveals the mechanism of deubiquitinase inhibition. Elife 5:e13027. https://doi.org/10.7554/eLife.13027
de Assis LJ, Ulas M, Ries LNA, El Ramli NAM, Sarikaya-Bayram Ö, Braus GH, Bayram Ö, Goldman GH (2018) Regulation of Aspergillus nidulans CreA-mediated catabolite repression by the F-box proteins Fbx23 and Fbx47. MBio 9(3):e00840–e00818. https://doi.org/10.1128/mBio.00840-18
de Poot SAH, Tian G, Finley D (2017) Meddling with fate: the proteasomal deubiquitinating enzymes. J Mol Biol 429(22):3525–3545. https://doi.org/10.1016/j.jmb.2017.09.015
Du L, Li N, Chen L, Xu Y, Li Y, Zhang Y, Li C, Li Y (2014) The ubiquitin receptor DA1 regulates seed and organ size by modulating the stability of the ubiquitin-specific protease UBP15/SOD2 in Arabidopsis. Plant Cell 26(2):665–677. https://doi.org/10.1105/tpc.114.122663
Dudley AM, Janse DM, Tanay A, Shamir R, Church GM (2005) A global view of pleiotropy and phenotypically derived gene function in yeast. Mol Syst Biol 1:2005.0001. https://doi.org/10.1038/msb4100004
Eisenman HC, Casadevall A (2012) Synthesis and assembly of fungal melanin. Appl Microbiol Biotechnol 93(3):931–940. https://doi.org/10.1007/s00253-011-3777-2
Emre NC, Berger SL (2004) Histone H2B ubiquitylation and deubiquitylation in genomic regulation. Cold Spring Harb Symp Quant Biol 69:289–299. https://doi.org/10.1101/sqb.2004.69.289
Farman ML (2002) Meiotic deletion at the BUF1 locus of the fungus Magnaporthe grisea is controlled by interaction with the homologous chromosome. Genetics 160(1):137–148
Foot N, Henshall T, Kumar S (2017) Ubiquitination and the regulation of membrane proteins. Physiol Rev 97(1):253–281. https://doi.org/10.1152/physrev.00012.2016
Fu T, Kim JO, Han JH, Gumilang A, Lee YH, Kim KS (2018) A small GTPase RHO2 plays an important role in pre-infection development in the rice blast pathogen Magnaporthe oryzae. Plant Pathol J 34(6):470–479. https://doi.org/10.5423/ppj.Oa.04.2018.0069
Glickman MH, Ciechanover A (2002) The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev 82(2):373–428. https://doi.org/10.1152/physrev.00027.2001
Guo M, Gao F, Zhu X, Nie X, Pan Y, Gao Z (2015) MoGrr1, a novel F-box protein, is involved in conidiogenesis and cell wall integrity and is critical for the full virulence of Magnaporthe oryzae. Appl Microbiol Biotechnol 99(19):8075–8088. https://doi.org/10.1007/s00253-015-6820-x
Guterman A, Glickman MH (2004) Deubiquitinating enzymes are IN/(trinsic to proteasome function). Curr Protein Pept Sci 5(3):201–211
Henry KW, Wyce A, Lo WS, Duggan LJ, Emre NC, Kao CF, Pillus L, Shilatifard A, Osley MA, Berger SL (2003) Transcriptional activation via sequential histone H2B ubiquitylation and deubiquitylation, mediated by SAGA-associated Ubp8. Genes Dev 17(21):2648–2663. https://doi.org/10.1101/gad.1144003
Hershko A, Ciechanover A (1998) The ubiquitin system. Annu Rev Biochem 67:425–479. https://doi.org/10.1146/annurev.biochem.67.1.425
Ho HC, MacGurn JA, Emr SD (2017) Deubiquitinating enzymes Ubp2 and Ubp15 regulate endocytosis by limiting ubiquitination and degradation of ARTs. Mol Biol Cell 28(9):1271–1283. https://doi.org/10.1091/mbc.E17-01-0008
Howard RJ, Valent B (1996) Breaking and entering: host penetration by the fungal rice blast pathogen Magnaporthe grisea. Annu Rev Microbiol 50:491–512. https://doi.org/10.1146/annurev.micro.50.1.491
Howard RJ, Ferrari MA, Roach DH, Money NP (1991) Penetration of hard substrates by a fungus employing enormous turgor pressures. Proc Natl Acad Sci U S A 88(24):11281–11284. https://doi.org/10.1073/pnas.88.24.11281
Hsu HE, Liu TN, Yeh CS, Chang TH, Lo YC, Kao CF (2015) Feedback control of Snf1 protein and its phosphorylation is necessary for adaptation to environmental stress. J Biol Chem 290(27):16786–16796. https://doi.org/10.1074/jbc.M115.639443
Hu M, Li P, Li M, Li W, Yao T, Wu JW, Gu W, Cohen RE, Shi Y (2002) Crystal structure of a UBP-family deubiquitinating enzyme in isolation and in complex with ubiquitin aldehyde. Cell 111(7):1041–1054
Ingvarsdottir K, Krogan NJ, Emre NC, Wyce A, Thompson NJ, Emili A, Hughes TR, Greenblatt JF, Berger SL (2005) H2B ubiquitin protease Ubp8 and Sgf11 constitute a discrete functional module within the Saccharomyces cerevisiae SAGA complex. Mol Cell Biol 25(3):1162–1172. https://doi.org/10.1128/mcb.25.3.1162-1172.2005
Klaubauf S, Ribot C, Melayah D, Lagorce A, Lebrun MH, de Vries RP (2013) The pentose catabolic pathway of the rice-blast fungus Magnaporthe oryzae involves a novel pentose reductase restricted to few fungal species. FEBS Lett 587(9):1346–1352. https://doi.org/10.1016/j.febslet.2013.03.003
Komander D, Clague MJ, Urbe S (2009) Breaking the chains: structure and function of the deubiquitinases. Nat Rev Mol Cell Biol 10(8):550–563. https://doi.org/10.1038/nrm2731
Kornitzer D, Ciechanover A (2000) Modes of regulation of ubiquitin-mediated protein degradation. J Cell Physiol 182(1):1–11. https://doi.org/10.1002/(sici)1097-4652(200001)182:1<1::aid-jcp1>3.0.co;2-v
Kosinsky RL, Wegwitz F, Hellbach N, Dobbelstein M, Mansouri A, Vogel T, Begus-Nahrmann Y, Johnsen SA (2015) Usp22 deficiency impairs intestinal epithelial lineage specification in vivo. Oncotarget 6(35):37906–37918. https://doi.org/10.18632/oncotarget.5412
Lamb C, Dixon RA (1997) The oxidative burst in plant disease resistance. Annu Rev Plant Physiol Plant Mol Biol 48:251–275. https://doi.org/10.1146/annurev.arplant.48.1.251
Leo M, Fanelli G, Di Vito S, Traversetti B, La Greca M, Palladino RA, Montanari A, Francisci S, Filetici P (2018) Ubiquitin protease Ubp8 is necessary for S. cerevisiae respiration. Biochim Biophys Acta, Mol Cell Res 1865(10):1491–1500. https://doi.org/10.1016/j.bbamcr.2018.07.025
Li Y, Wang Y (2013) Ras protein/cAMP-dependent protein kinase signaling is negatively regulated by a deubiquitinating enzyme, Ubp3, in yeast. J Biol Chem 288(16):11358–11365. https://doi.org/10.1074/jbc.M112.449751
Li W, Zhong S, Li G, Li Q, Mao B, Deng Y, Zhang H, Zeng L, Song F, He Z (2011) Rice RING protein OsBBI1 with E3 ligase activity confers broad-spectrum resistance against Magnaporthe oryzae by modifying the cell wall defence. Cell Res 21(5):835–848. https://doi.org/10.1038/cr.2011.4
Li Y, Zhang X, Hu S, Liu H, Xu JR (2017) PKA activity is essential for relieving the suppression of hyphal growth and appressorium formation by MoSfl1 in Magnaporthe oryzae. PLoS Genet 13(8):e1006954. https://doi.org/10.1371/journal.pgen.1006954
Liu Y, Wang F, Zhang H, He H, Ma L, Deng XW (2008) Functional characterization of the Arabidopsis ubiquitin-specific protease gene family reveals specific role and redundancy of individual members in development. Plant J 55(5):844–856. https://doi.org/10.1111/j.1365-313X.2008.03557.x
Lockington RA, Kelly JM (2001) Carbon catabolite repression in Aspergillus nidulans involves deubiquitination. Mol Microbiol 40(6):1311–1321. https://doi.org/10.1046/j.1365-2958.2001.02474.x
Marroquin-Guzman M, Hartline D, Wright JD, Elowsky C, Bourret TJ, Wilson RA (2017) The Magnaporthe oryzae nitrooxidative stress response suppresses rice innate immunity during blast disease. Nat Microbiol 2:17054. https://doi.org/10.1038/nmicrobiol.2017.54
Matar KAO, Chen X, Chen D, Anjago WM, Norvienyeku J, Lin Y, Chen M, Wang Z, Ebbole DJ, Lu GD (2017) WD40-repeat protein MoCreC is essential for carbon repression and is involved in conidiation, growth and pathogenicity of Magnaporthe oryzae. Curr Genet 63(4):685–696. https://doi.org/10.1007/s00294-016-0668-1
McCormick MA, Mason AG, Guyenet SJ, Dang W, Garza RM, Ting MK, Moller RM, Berger SL, Kaeberlein M, Pillus L, La Spada AR, Kennedy BK (2014) The SAGA histone deubiquitinase module controls yeast replicative lifespan via Sir2 interaction. Cell Rep 8(2):477–486. https://doi.org/10.1016/j.celrep.2014.06.037
Mellersh DG, Foulds IV, Higgins VJ, Heath MC (2002) H2O2 plays different roles in determining penetration failure in three diverse plant-fungal interactions. Plant J 29(3):257–268. https://doi.org/10.1046/j.0960-7412.2001.01215.x
Morgan MT, Haj-Yahya M, Ringel AE, Bandi P, Brik A, Wolberger C (2016) Structural basis for histone H2B deubiquitination by the SAGA DUB module. Science 351(6274):725–728. https://doi.org/10.1126/science.aac5681
Nijman SM, Luna-Vargas MP, Velds A, Brummelkamp TR, Dirac AM, Sixma TK, Bernards R (2005) A genomic and functional inventory of deubiquitinating enzymes. Cell 123(5):773–786. https://doi.org/10.1016/j.cell.2005.11.007
Oh Y, Franck WL, Han SO, Shows A, Gokce E, Muddiman DC, Dean RA (2012) Polyubiquitin is required for growth, development and pathogenicity in the rice blast fungus Magnaporthe oryzae. PLoS One 7(8):e42868. https://doi.org/10.1371/journal.pone.0042868
Piao J, Tashiro A, Nishikawa M, Aoki Y, Moriyoshi E, Hattori A, Kakeya H (2015) Expression, purification and enzymatic characterization of a recombinant human ubiquitin-specific protease 47. J Biochem 158(6):477–484. https://doi.org/10.1093/jb/mvv063
Portnoy T, Margeot A, Linke R, Atanasova L, Fekete E, Sandor E, Hartl L, Karaffa L, Druzhinina IS, Seiboth B, Le Crom S, Kubicek CP (2011) The CRE1 carbon catabolite repressor of the fungus Trichoderma reesei: a master regulator of carbon assimilation. BMC Genomics 12:269. https://doi.org/10.1186/1471-2164-12-269
Reyes-Turcu FE, Ventii KH, Wilkinson KD (2009) Regulation and cellular roles of ubiquitin-specific deubiquitinating enzymes. Annu Rev Biochem 78:363–397. https://doi.org/10.1146/annurev.biochem.78.082307.091526
Ribeiro LFC, Chelius C, Boppidi KR, Naik NS, Hossain S, Ramsey JJJ, Kumar J, Ribeiro LF, Ostermeier M, Tran B, Ah Goo Y, de Assis LJ, Ulas M, Bayram Ö, Goldman GH, Lincoln S, Srivastava R, Harris SD, Marten MR (2019) Comprehensive analysis of Aspergillus nidulans PKA phosphorylome identifies a novel mode of CreA regulation. MBio 10(2):e02825–e02818. https://doi.org/10.1128/mBio.02825-18
Ries LN, Beattie SR, Espeso EA, Cramer RA, Goldman GH (2016) Diverse regulation of the CreA carbon catabolite repressor in Aspergillus nidulans. Genetics 203(1):335–352. https://doi.org/10.1534/genetics.116.187872
Romao J, Hamer JE (1992) Genetic organization of a repeated DNA sequence family in the rice blast fungus. Proc Natl Acad Sci U S A 89(12):5316–5320. https://doi.org/10.1073/pnas.89.12.5316
Shukla A, Stanojevic N, Duan Z, Sen P, Bhaumik SR (2006) Ubp8p, a histone deubiquitinase whose association with SAGA is mediated by Sgf11p, differentially regulates lysine 4 methylation of histone H3 in vivo. Mol Cel Biol 26(9):3339–3352. https://doi.org/10.1128/mcb.26.9.3339-3352.2006
Smith FC, Davies SP, Wilson WA, Carling D, Hardie DG (1999) The SNF1 kinase complex from Saccharomyces cerevisiae phosphorylates the transcriptional repressor protein Mig1p in vitro at four sites within or near regulatory domain 1. FEBS Lett 453(1–2):219–223. https://doi.org/10.1016/s0014-5793(99)00725-5
Spedale G, Timmers HT, Pijnappel WW (2012) ATAC-king the complexity of SAGA during evolution. Genes Dev 26(6):527–541. https://doi.org/10.1101/gad.184705.111
Wang L, Dent SY (2014) Functions of SAGA in development and disease. Epigenomics 6(3):329–339. https://doi.org/10.2217/epi.14.22
Wang J, Yin Z, Tang W, Cai X, Gao C, Zhang H, Zheng X, Wang P, Zhang Z (2017) The thioredoxin MoTrx2 protein mediates reactive oxygen species (ROS) balance and controls pathogenicity as a target of the transcription factor MoAP1 in Magnaporthe oryzae. Mol Plant Pathol 18(9):1199–1209. https://doi.org/10.1111/mpp.12484
Wang Z, Zhang H, Liu C, Xing J, Chen XL (2018) A deubiquitinating enzyme Ubp14 is required for development, stress response, nutrient utilization, and pathogenesis of Magnaporthe oryzae. Front Microbiol 9:769. https://doi.org/10.3389/fmicb.2018.00769
Wilkinson KD (1997) Regulation of ubiquitin-dependent processes by deubiquitinating enzymes. FASEB J 11(14):1245–1256
Zhang XY, Varthi M, Sykes SM, Phillips C, Warzecha C, Zhu W, Wyce A, Thorne AW, Berger SL, McMahon SB (2008) The putative cancer stem cell marker USP22 is a subunit of the human SAGA complex required for activated transcription and cell-cycle progression. Mol Cell 29(1):102–111. https://doi.org/10.1016/j.molcel.2007.12.015
Zhang J, Yun Y, Lou Y, Abubakar YS, Guo P, Wang S, Li C, Feng Y, Adnan M, Zhou J, Lu GD, Zheng W (2019a) FgAP-2 complex is essential for pathogenicity and polarised growth and regulates the apical localisation of membrane lipid flippases in Fusarium graminearum. Cell Microbiol 21(8):e13041. https://doi.org/10.1111/cmi.13041
Zhang L, Zhang D, Chen Y, Ye W, Lin Q, Lu G, Ebbole DJ, Olsson S, Wang Z (2019b) Magnaporthe oryzae CK2 accumulates in nuclei, nucleoli, at septal pores and forms a large ring structure in appressoria, and is involved in eice blast pathogenesis. Front Cell Infect Microbiol 9:113. https://doi.org/10.3389/fcimb.2019.00113
Acknowledgments
We really appreciate Dr. Justice Norvienyeku (Fujian Agriculture and Forestry University) for his valuable suggestions and language improvement for this paper.
Funding
This work was supported by NSFC (National Natural Science Foundation of China) (grants number 31571943) and Agricultural Guiding (Key) Project of Fujian Province, China (2019 N0003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 1297 kb)
Rights and permissions
About this article
Cite this article
Yang, J., Chen, D., Matar, K.A.O. et al. The deubiquitinating enzyme MoUbp8 is required for infection-related development, pathogenicity, and carbon catabolite repression in Magnaporthe oryzae. Appl Microbiol Biotechnol 104, 5081–5094 (2020). https://doi.org/10.1007/s00253-020-10572-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10572-5