Abstract
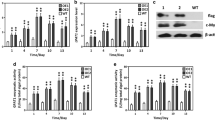
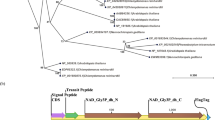
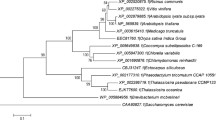
Commercial production of biofuel from oleaginous microalgae is often impeded by their slow growth rate than other fast-growing algal species. A promising strategy is to genetically engineer the fast-growing algae to accumulate lipids by expressing key lipogenic genes from oleaginous microalgae. However, lacking of strong expression cassette to transform most of the algal species and potential metabolic target to engineer lipid metabolism has hindered its biotechnological applications. In this study, we engineered the oxidative pentose phosphate pathway (PPP) of green microalga Chlorella pyrenoidosa for lipid enhancement by expressing a glucose-6-phosphate dehydrogenase (G6PD) from oleaginous diatom Phaeodactylum tricornutum. Molecular characterization of transformed lines revealed that heterologous PtG6PD was transcribed and expressed successfully. Interestingly, subcellular localization analyses revealed that PtG6PD was targeted to chloroplasts of C. pyrenoidosa. PtG6PD expression remarkably elevated NADPH content and consequently enhanced the lipid content without affecting growth rate. Collectively, this report represents a promising candidate to engineer lipid biosynthesis in heterologous hosts with notable commercial significance, and it highlights the potential role of plastidial PPP in supplying lipogenic NADPH in microalgae.






Similar content being viewed by others
References
Ajjawi I, Verruto J, Aqui M, Soriaga LB, Coppersmith J, Kwok K, Peach L, Orchard E, Kalb R, Xu W, Carlson TJ, Francis K, Konigsfeld K, Bartalis J, Schultz A, Lambert W, Schwartz AS, Brown R, Moellering ER (2017) Lipid production in Nannochloropsis gaditana is doubled by decreasing expression of a single transcriptional regulator. Nat Biotechnol 35(7):647–652. https://doi.org/10.1038/nbt.3865
Al-Dwairi A, Pabona JM, Simmen RC, Simmen FA (2012) Cytosolic malic enzyme 1 (ME1) mediates high fat diet-induced adiposity, endocrine profile, and gastrointestinal tract proliferation-associated biomarkers in male mice. PLoS One 7(10):e46716. https://doi.org/10.1371/journal.pone.0046716
Allen MM, Stanier RY (1968) Growth and division of some unicellular blue-green algae. Microbiology 51(2):199–202. https://doi.org/10.1099/00221287-51-2-199
Armbrust EV, Berges JA, Bowler C, Green BR, Martinez D, Putnam NH, Zhou S, Allen AE, Apt KE, Bechner M, Brzezinski MA, Chaal BK, Chiovitti A, Davis AK, Demarest MS, Detter JC, Glavina T, Goodstein D, Hadi MZ, Hellsten U, Hildebrand M, Jenkins BD, Jurka J, Kapitonov VV, Kroger N, Lau WW, Lane TW, Larimer FW, Lippmeier JC, Lucas S, Medina M, Montsant A, Obornik M, Parker MS, Palenik B, Pazour GJ, Richardson PM, Rynearson TA, Saito MA, Schwartz DC, Thamatrakoln K, Valentin K, Vardi A, Wilkerson FP, Rokhsar DS (2004) The genome of the diatom Thalassiosira pseudonana: ecology, evolution, and metabolism. Science 306(5693):79–86. https://doi.org/10.1126/science.1101156
Balamurugan S, Wang X, Wang HL, An CJ, Li H, Li DW, Yang WD, Liu JS, Li HY (2017) Occurrence of plastidial triacylglycerol synthesis and the potential regulatory role of AGPAT in the model diatom Phaeodactylum tricornutum. Biotechnol Biofuels 10:97. https://doi.org/10.1186/s13068-017-0786-0
Belotti G, de Caprariis B, De Filippis P, Scarsella M, Verdone N (2014) Effect of Chlorella vulgaris growing conditions on bio-oil production via fast pyrolysis. Biomass Bioenergy 61:187–195. https://doi.org/10.1016/j.biombioe.2013.12.011
Beopoulos A, Mrozova Z, Thevenieau F, Le Dall M-T, Hapala I, Papanikolaou S, Chardot T, Nicaud J-M (2008) Control of lipid accumulation in the yeast Yarrowia lipolytica. Appl Environ Microbiol 74(24):7779–7789. https://doi.org/10.1128/aem.01412-08
Blank LM, Lehmbeck F, Sauer U (2005) Metabolic-flux and network analysis in fourteen hemiascomycetous yeasts. FEMS Yeast Res 5(6–7):545–558. https://doi.org/10.1016/j.femsyr.2004.09.008
Chen J-W, Liu W-J, Hu D-X, Wang X, Balamurugan S, Alimujiang A, Yang W-D, Liu J-S, Li H-Y (2016) Identification of a malonyl CoA-acyl carrier protein transacylase and its regulatory role in fatty acid biosynthesis in oleaginous microalga Nannochloropsis oceanica. Biotechnol Appl Biochem 64(5):620–626. https://doi.org/10.1002/bab.1531
Chiaiese P, Palomba F, Tatino F, Lanzillo C, Pinto G, Pollio A, Filippone E (2011) Engineered tobacco and microalgae secreting the fungal laccase PDXA1b reduce phenol content in olive oil mill wastewater. Enzym Microb Technol 49(6–7):540–546. https://doi.org/10.1016/j.enzmictec.2011.06.002
Chou K-C (2010) Cell-PLoc 2.0: an improved package of web-servers for predicting subcellular localization of proteins in various organisms. Nat Sci 02(10):1090–1103
Eastmond PJ, Rawsthorne S (2000) Coordinate changes in carbon partitioning and plastidial metabolism during the development of oilseed rape embryo. Plant Physiol 122(3):767–774. https://doi.org/10.1104/pp.122.3.767
Esposito S, Guerriero G, Rigano V, Carfagna S, Rigano C (2006) Glucose-6P dehydrogenase in Chlorella sorokiniana (211/8k): an enzyme with unusual characteristics. Planta 223(4):796–804. https://doi.org/10.1007/s00425-005-0110-2
Fan J, Andre C, Xu C (2011) A chloroplast pathway for the de novo biosynthesis of triacylglycerol in Chlamydomonas reinhardtii. FEBS Lett 585(12):1985–1991. https://doi.org/10.1016/j.febslet.2011.05.018
Ferrara M, Guerriero G, Cardi M, Esposito S (2013) Purification and biochemical characterisation of a glucose-6-phosphate dehydrogenase from the psychrophilic green alga Koliella antarctica. Extremophiles 17(1):53–62. https://doi.org/10.1007/s00792-012-0492-6
Ge F, Huang W, Chen Z, Zhang C, Xiong Q, Bowler C, Yang J, Xu J, Hu H (2014) Methylcrotonyl-CoA carboxylase regulates triacylglycerol accumulation in the model diatom Phaeodactylum tricornutum. Plant Cell 26(4):1681–1697. https://doi.org/10.1105/tpc.114.124982
Görs M, Schumann R, Hepperle D, Karsten U (2009) Quality analysis of commercial Chlorella products used as dietary supplement in human nutrition. J Appl Phycol 22(3):265–276. https://doi.org/10.1007/s10811-009-9455-4
Gruber A, Weber T, Bartulos CR, Vugrinec S, Kroth PG (2009) Intracellular distribution of the reductive and oxidative pentose phosphate pathways in two diatoms. J Basic Microbiol 49(1):58–72. https://doi.org/10.1002/jobm.200800339
Guccione A, Biondi N, Sampietro G, Rodolfi L, Bassi N, Tredici MR (2014) Chlorella for protein and biofuels: from strain selection to outdoor cultivation in a Green Wall Panel photobioreactor. Biotechnol Biofuels 7:84. https://doi.org/10.1186/1754-6834-7-84
Guo SL, Zhao XQ, Tang Y, Wan C, Alam MA, Ho SH, Bai FW, Chang JS (2013) Establishment of an efficient genetic transformation system in Scenedesmus obliquus. J Biotechnol 163(1):61–68. https://doi.org/10.1016/j.jbiotec.2012.10.020
Hockin NL, Mock T, Mulholland F, Kopriva S, Malin G (2012) The response of diatom central carbon metabolism to nitrogen starvation is different from that of green algae and higher plants. Plant Physiol 158(1):299–312. https://doi.org/10.1104/pp.111.184333
Hoelscher C, Meyer T, von Schaewen A (2014) Dual-targeting of Arabidopsis 6-phosphogluconolactonase 3 (PGL3) to chloroplasts and peroxisomes involves interaction with Trx m2 in the cytosol. Mol Plant 7(1):252–255. https://doi.org/10.1093/mp/sst126
Hsieh HJ, Su CH, Chien LJ (2012) Accumulation of lipid production in Chlorella minutissima by triacylglycerol biosynthesis-related genes cloned from Saccharomyces cerevisiae and Yarrowia lipolytica. J Microbiol Biotechnol 50(3):526–534. https://doi.org/10.1007/s12275-012-2041-5
Jesus Ramos M, Maria Fernandez C, Casas A, Rodriguez L, Perez A (2009) Influence of fatty acid composition of raw materials on biodiesel properties. Bioresour Technol 100(1):261–268. https://doi.org/10.1016/j.biortech.2008.06.039
Junker BH, Lonien J, Heady LE, Rogers A, Schwender J (2007) Parallel determination of enzyme activities and in vivo fluxes in Brassica napus embryos grown on organic or inorganic nitrogen source. Phytochemistry 68(16–18):2232–2242. https://doi.org/10.1016/j.phytochem.2007.03.032
Kabashima T, Kawaguchi T, Wadzinski BE, Uyeda K (2003) Xylulose 5-phosphate mediates glucose-induced lipogenesis by xylulose 5-phosphate-activated protein phosphatase in rat liver. Proc Natl Acad Sci U S A 100(9):5107–5112. https://doi.org/10.1073/pnas.0730817100
Kang NK, Kim EK, Kim YU, Lee B, Jeong W-J, Jeong B-R, Chang YK (2017) Increased lipid production by heterologous expression of AtWRI1 transcription factor in Nannochloropsis salina. Biotechnol Biofuels 10:231. https://doi.org/10.1186/s13068-017-0919-5
Kletzien RF, Harris PK, Foellmi LA (1994) Glucose-6-phosphate dehydrogenase: a “housekeeping” enzyme subject to tissue-specific regulation by hormones, nutrients, and oxidant stress. FASEB J 8(2):174–181
Kroth PG, Chiovitti A, Gruber A, Martin-Jezequel V, Mock T, Parker MS, Stanley MS, Kaplan A, Caron L, Weber T, Maheswari U, Armbrust EV, Bowler C, Kroymann J (2008) A model for carbohydrate metabolism in the diatom Phaeodactylum tricornutum deduced from comparative whole genome analysis. PLoS One 3(1):e1426. https://doi.org/10.1371/journal.pone.0001426
Lee KM, DaSilva NA (2005) Evaluation of the Saccharomyces cerevisiae ADH2 promoter for protein synthesis. Yeast 22(6):431–440. https://doi.org/10.1002/yea.1221
Legan SK, Rebrin I, Mockett RJ, Radyuk SN, Klichko VI, Sohal RS, Orr WC (2008) Overexpression of glucose-6-phosphate dehydrogenase extends the life span of Drosophila melanogaster. J Biol Chem 283(47):32492–32499
Li Y, Han D, Hu G, Dauvillee D, Sommerfeld M, Ball S, Hua Q (2010) Chlamydomonas starchless mutant defective in ADP-glucose pyrophosphorylase hyper-accumulates triacylglycerol. Metab Eng 12(4):387–391. https://doi.org/10.1016/j.ymben.2010.02.002
Loiudice FH, Silva DP, Zanchin NI, Oliveira CC, Pessoa A Jr (2001) Overexpression of glucose-6-phosphate dehydrogenase in genetically modified Saccharomyces cerevisiae. Appl Biochem Biotechnol 91-93:161–169
Lu Y, Wang X, Balamurugan S, Yang W-D, Liu J-S, Dong H-P, Li H-Y (2017) Identification of a putative seipin ortholog involved in lipid accumulation in marine microalga Phaeodactylum tricornutum. J Appl Phycol 29(6):2821–2829. https://doi.org/10.1007/s10811-017-1173-8
Meher LC, Sagar DV, Naik SN (2006) Technical aspects of biodiesel production by transesterification - a review. Renew Sust Energ Rev 10(3):248–268. https://doi.org/10.1016/j.rser.2004.09.002
Mejias R, Villadiego J, Pintado CO, Vime PJ, Gao L, Toledo-Aral JJ, Echevarria M, Lopez-Barneo J (2006) Neuroprotection by transgenic expression of glucose-6-phosphate dehydrogenase in dopaminergic nigrostriatal neurons of mice. J Neurosci 26(17):4500–4508. https://doi.org/10.1523/jneurosci.0122-06.2006
Mus F, Toussaint J-P, Cooksey KE, Fields MW, Gerlach R, Peyton BM, Carlson RP (2013) Physiological and molecular analysis of carbon source supplementation and pH stress-induced lipid accumulation in the marine diatom Phaeodactylum tricornutum. Appl Microbiol Biotechnol 97(8):3625–3642. https://doi.org/10.1007/s00253-013-4747-7
Nicol CJ, Zielenski J, Tsui L-C, Wells PG (2000) An embryoprotective role for glucose-6-phosphate dehydrogenase in developmental oxidative stress and chemical teratogenesis. FASEB J 14(1):111–127
Niu YF, Wang X, Hu DX, Balamurugan S, Li DW, Yang WD, Liu JS, Li HY (2016) Molecular characterization of a glycerol-3-phosphate acyltransferase reveals key features essential for triacylglycerol production in Phaeodactylum tricornutum. Biotechnol Biofuels 9:60. https://doi.org/10.1186/s13068-016-0478-1
Park J, Rho HK, Kim KH, Choe SS, Lee YS, Kim JB (2005) Overexpression of glucose-6-phosphate dehydrogenase is associated with lipid dysregulation and insulin resistance in obesity. Mol Cell Biol 25(12):5146–5157. https://doi.org/10.1128/MCB.25.12.5146-5157.2005
Ratledge C (2014) The role of malic enzyme as the provider of NADPH in oleaginous microorganisms: a reappraisal and unsolved problems. Biotechnol Lett 36(8):1557–1568. https://doi.org/10.1007/s10529-014-1532-3
Reumann S, Babujee L, Ma C, Wienkoop S, Siemsen T, Antonicelli GE, Rasche N, Lueder F, Weckwerth W, Jahn O (2007) Proteome analysis of Arabidopsis leaf peroxisomes reveals novel targeting peptides, metabolic pathways, and defense mechanisms. Plant Cell 19(10):3170–3193. https://doi.org/10.1105/tpc.107.050989
Riethmuller-Haage I, Bastiaans L, Harbinson J, Kempenaar C, Kropff MJ (2006) Influence of the acetolactate synthase inhibitor metsulfuron-methyl on the operation, regulation and organisation of photosynthesis in Solanum nigrum. Photosynth Res 88(3):331–341. https://doi.org/10.1007/s11120-006-9062-z
Santoro N, Caprio S, Pierpont B, Van Name M, Savoye M, Parks EJ (2015) Hepatic de novo lipogenesis in obese youth is modulated by a common variant in the GCKR gene. J Clin Endocrinol Metab 100(8):E1125–E1132. https://doi.org/10.1210/jc.2015-1587
Schnarrenberger C, Oeser A, Tolbert NE (1973) Two isoenzymes each of glucose-6-phosphate dehydrogenase and 6-phosphogluconate dehydrogenase in spinach leaves. Arch Biochem Biophys 154(1):438–448. https://doi.org/10.1016/0003-9861(73)90077-5
Schwender J, Shachar-Hill Y, Ohlrogge JB (2006) Mitochondrial metabolism in developing embryos of Brassica napus. J Biol Chem 281(45):34040–34047. https://doi.org/10.1074/jbc.M606266200
Szabo NJ, Matulka RA, Chan T (2013) Safety evaluation of whole Algalin protein (WAP) from Chlorella protothecoides. Food Chem Toxicol 59:34–45. https://doi.org/10.1016/j.fct.2013.05.035
Tai M, Stephanopoulos G (2013) Engineering the push and pull of lipid biosynthesis in oleaginous yeast Yarrowia lipolytica for biofuel production. Metab Eng 15:1–9. https://doi.org/10.1016/j.ymben.2012.08.007
Talebi AF, Tohidfar M, Tabatabaei M, Bagheri A, Mohsenpor M, Mohtashami SK (2013) Genetic manipulation, a feasible tool to enhance unique characteristic of Chlorella vulgaris as a feedstock for biodiesel production. Mol Biol Rep 40(7):4421–4428. https://doi.org/10.1007/s11033-013-2532-4
Tan KWM, Lee YK (2017) Expression of the heterologous Dunaliella tertiolecta fatty acyl-ACP thioesterase leads to increased lipid production in Chlamydomonas reinhardtii. J Biotechnol 247:60–67. https://doi.org/10.1016/j.jbiotec.2017.03.004
Valenzuela J, Mazurie A, Carlson RP, Gerlach R, Cooksey KE, Peyton BM, Fields MW (2012) Potential role of multiple carbon fixation pathways during lipid accumulation in Phaeodactylum tricornutum. Biotechnol Biofuels 5:40. https://doi.org/10.1186/1754-6834-5-40
Wang X, Liu Y-H, Hu D-X, Balamurugan S, Lu Y, Yang W-D, Liu J-S, Li H-Y (2015) Identification of a putative patatin-like phospholipase domain-containing protein 3 (PNPLA3) ortholog involved in lipid metabolism in microalga Phaeodactylum tricornutum. Algal Res 12:274–279. https://doi.org/10.1016/j.algal.2015.09.005
Wasylenko TM, Ahn WS, Stephanopoulos G (2015) The oxidative pentose phosphate pathway is the primary source of NADPH for lipid overproduction from glucose in Yarrowia lipolytica. Metab Eng 30:27–39. https://doi.org/10.1016/j.ymben.2015.02.007
Wendt UK, Wenderoth I, Tegeler A, von Schaewen A (2000) Molecular characterization of a novel glucose-6-phosphate dehydrogenase from potato (Solanum tuberosum L.). Plant J 23(6):723–733. https://doi.org/10.1046/j.1365-313x.2000.00840.x
Xie W-H, Zhu C-C, Zhang N-S, Li D-W, Yang W-D, Liu J-S, Sathishkumar R, Li H-Y (2014) Construction of novel chloroplast expression vector and development of an efficient transformation system for the diatom Phaeodactylum tricornutum. Mar Biotechnol 16(5):538–546. https://doi.org/10.1007/s10126-014-9570-3
Xue J, Niu Y-F, Huang T, Yang W-D, Liu J-S, Li H-Y (2015) Genetic improvement of the microalga Phaeodactylum tricornutum for boosting neutral lipid accumulation. Metab Eng 27:1–9. https://doi.org/10.1016/j.ymben.2014.10.002
Xue J, Wang L, Zhang L, Balamurugan S, Li D-W, Zeng H, Yang W-D, Liu J-S, Li H-Y (2016) The pivotal role of malic enzyme in enhancing oil accumulation in green microalga Chlorella pyrenoidosa. Microb Cell Factories 15(1):120. https://doi.org/10.1186/s12934-016-0519-2
Xue J, Balamurugan S, Li D-W, Liu Y-H, Zeng H, Wang L, Yang W-D, Liu J-S, Li H-Y (2017) Glucose-6-phosphate dehydrogenase as a target for highly efficient fatty acid biosynthesis in microalgae by enhancing NADPH supply. Metab Eng 41:212–221. https://doi.org/10.1016/j.ymben.2017.04.008
Yang ZK, Niu YF, Ma YH, Xue J, Zhang MH, Yang WD, Liu JS, Lu SH, Guan Y, Li HY (2013) Molecular and cellular mechanisms of neutral lipid accumulation in diatom following nitrogen deprivation. Biotechnol Biofuels 6(1):67. https://doi.org/10.1186/1754-6834-6-67
Zaslavskaia LA, Lippmeier JC, Kroth PG, Grossman AR, Apt KE (2000) Transformation of the diatom Phaeodactylum tricornutum (Bacillariophyceae) with a variety of selectable marker and reporter genes. J Phycol 36(2):379–386. https://doi.org/10.1046/j.1529-8817.2000.99164.x
Zhang Y, Adams IP, Ratledge C (2007) Malic enzyme: the controlling activity for lipid production? Overexpression of malic enzyme in Mucor circinelloides leads to a 2.5-fold increase in lipid accumulation. Microbiology 153(7):2013–2025. https://doi.org/10.1099/mic.0.2006/002683-0
Zhang X, Li M, Agrawal A, San K-Y (2011) Efficient free fatty acid production in Escherichia coli using plant acyl-ACP thioesterases. Metab Eng 13(6):713–722. https://doi.org/10.1016/j.ymben.2011.09.007
Funding
This work was supported by the Guangdong Natural Science Foundation (2014A030308010) and the Natural Science Foundation of China (41576132).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Xue, J., Chen, TT., Zheng, JW. et al. The role of diatom glucose-6-phosphate dehydrogenase on lipogenic NADPH supply in green microalgae through plastidial oxidative pentose phosphate pathway. Appl Microbiol Biotechnol 102, 10803–10815 (2018). https://doi.org/10.1007/s00253-018-9415-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9415-5