Abstract
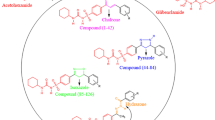
This study aims at identifying novel chemical scaffolds as inhibitors specific to the acetyltransferase domain of a bifunctional enzyme, Escherichia coli GlmU, involved in the cell wall biosynthesis of Gram-negative organisms. A two-pronged approach was used to screen a 50,000 small-molecule library. Using the first approach, the library was in silico screened by docking the library against acetyltransferase domain of E. coli GlmU studies. In the second approach, complete library was screened against Escherichia coli ATCC 25922 to identify the whole cell active compounds. Active compounds from both the screens were screened in a colorimetric absorbance-based assay to identify inhibitors of acetyltransferase domain of E. coli GlmU which resulted in the identification of 1 inhibitor out of 56 hits identified by in silico screening and 4 inhibitors out of 35 whole cell active compounds on Gram-negative bacteria with the most potent inhibitor showing IC50 of 1.40 ± 0.69 μM. Mode of inhibition studies revealed these inhibitors to be competitive with AcCoA and uncompetitive with GlcN-1-P. These selected inhibitors were also tested for their antibacterial and cytotoxic activities. Compounds 5175178 and 5215319 exhibited antibacterial activity that co-related with GlmU inhibition. These compounds, therefore, represent novel chemical scaffolds targeting acetyltransferase activity of E. coli GlmU.






Similar content being viewed by others
References
Aquino A, Chan J, Giolma K, Loh M (2010) The effect of a fullerene water suspension on the growth, cell viability, and membrane integrity of Escherichia coli B23. J Exp Microbiol Immunol 14:13–20
Bassetti M, Ginocchio F, Mikulska M (2011) New treatment options against Gram-negative organisms. Crit Care 15:215
Brown K, Pompeo F, Dixon S, Lecreulx DM, Cambillau C, Bourne Y (1999) Crystal structure of the bifunctional N-acetylglucosamine-1-phoaphate uridyltransferase from E. coli: a paradigm for the related pyrophosphorylase superfamily. EMBO J 18:4096–4107
Burton E, Gawande PV, Yakandawala N, Vetri KL, Zhanel GG, Romeo T, Friesen AD, Madhyastha S (2006) Antibiofilm activity of GlmU enzyme inhibitors against catheter-associated uropathogens. Antimicrob Agents Chemother 50:1835–1840
Buurman ET, Andrews B, Gao N, Hu J, Keating TA, Lahiri S, Otterbein LR, Patten AD, Strokes SS, Shapiro AB (2011) In vitro validation of acetyltransferase activity of GlmU as an antibacterial target in H. influenzae. J Biol Chem 286:40734–40742
Clinical and Laboratory Standards Institute (2009) Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically: approved standard, vol 2, 29th edn, 9th Ed., M07-A8. Clinical and Laboratory Standards Institute, Wayne, PA
Copeland RA (2004) Enzymes: a practical introduction to structure, mechanism, and data analysis, 2nd edn. Wiley, NY
Dorsey WC, Tchounwou PB, Sutton D (2004) Mitogenic and cytotoxic effects of pentachlorophenol to AML 12 mouse hepatocytes. Int J Environ Res Publ Health 1:100–105
El-Nakeeb MA, Abou-Shleib HM, Khalil AM, Omar HG, El-Halfawy OM (2011) Membrane permeability alteration of some bacterial clinical isolates by selected antihistaminics. Braz J Microbiol 42:992–1000
Fleischmann RD, Adams MD, White O, Clayton RA, Kirkness EF, Kerlavage AR, Bult CJ, Tomb JF, Dougherty BA, Merrick JM (1995) Whole genome random sequencing and assembly of Haemophilus influenzae Rd. Science 269:496–512
Friesner RA, Banks JL, Murphy RB, Halgren TA, Klicic JJ, Mainz DT, Repasky MP, Knoll EH, Shaw DE, Shelley M, Perry JK, Francis P, Shenkin PS (2004) Glide: a new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J Med Chem 47:1739–1749
Gehring AM, Lees WJ, Mindiola DJ, Walsh CT, Brown ED (1996) Acetyltransferase precedes uridyltransfer in the formation of UDP-N-acetylglucosamine in separable sites of bifunctional GlmU protein of E. coli. Biochemistry 35:579–585
Green OM, Mckenzie AR, Shapiro AB, Otterbein L, Ni H, Patten A, Stokes S, Albert R, Kawatkar S, Breed J (2012) Inhibitors of acetyltransferase domain of N-acetylglucosamine-1-phosphate-uridyltransferase/glucosamine-1-phosphate acetyltransferase (GlmU). Part 1: hit to lead evaluation of a novel arylsulfonamide series. Bioorg Med Chem Lett 22:1510–1519
Jagtap PKA, Soni V, Vithani N, Jhingan GD, Bais VS, Nandicoori VK, Prakash B (2012) Substrate-bound crystal structures reveal features unique to Mycobacterium tuberculosis N-acetyl-glucosamine 1-phosphate uridyltransferase and a catalytic mechanism for acetyl transfer. J Biol Chem 287:39524–39537
Kaya EG, Özbilge H, Albayrak S (2009) Against staphylococcus aureus by determination of the effect of using gentamicin microbroth kinetic system. ANKEM Derg 23:110–114
Lecreulx DM, Heijenoort JV (1993) Identification of the glmU gene encoding N-acetylglucosamine-1-phosphate uridyltransferase in E. coli. J Bacteriol 175:6150–6157
Lecreulx DM, Heijenoort JV (1994) Copurification of glucosamine-1-phosphate acetyltransferase and N-acetylglucosamine-1-phosphate uridyltransferase activities of Escherichia coli: characterization of the glmU gene product as a bifunctional enzyme catalyzing two subsequent steps in the pathway for UDP-N-acetylglucosamine synthesis. J Bacteriol 176:5788–5795
Mehra R, Sharma R, Khan IA, Nargotra A (2014) Identification and optimization of Escherichia coli GlmU inhibitors: an in silico approach with validation thereof. Eur J Med Chem 92:78–90
Milewski S, Gabriel I, Olchowy J (2006) Enzymes of UDP-GlcNAc biosynthesis in yeast. Yeast 23:1–14
Mochalkin I, Lightle S, Yaqizhu OJF, Spessard C, Chirgadze NY, Banotai C, Melnick M, Mcdowell L (2007) Characterization of substrate binding and catalysis in the potential antibacterial target N-acetylglucosamine-1-phosphate uridyltransferase (GlmU). Protein Sci 16:2657–2666
Olesen SH, Ingles DJ, Yang Y, Schönbrunn E (2014) Differential antibacterial properties of the acid and fosfomycin terreic MurA inhibitors. J Basic Microbiol 54:322–326
Olsen LR, Roderick SL (2001) Structure of the Escherichia coli GlmU pyrophosphorylase and acetyltransferase active sites. Biochemistry 40:1913–1921
Olsen LR, Vetting MW, Roderick SL (2007) Structure of the E. coli bifunctional GlmU acetyltransferase active site with substrates and products. Protein Sci 16:1230–1235
Pereira MP, Blanchard JE, Murphy C, Roderick SL, Brown ED (2009) High throughput screening identifies novel inhibitors of the acetyltransferase activity of E. coli GlmU. Antimicrob Agents Chemother 53:2306–2311
Pompeo F, Heijenoort JV, Lecreulx DM (1998) Probing the role of cysteine residues in glucosamine-1-phosphate acetyltransferase activity of the bifunctional GlmU protein from Escherichia coli: site-directed mutagenesis and characterization of the mutant enzymes. J Bacteriol 180:4799–4803
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Sarvas M (1971) Mutant of E. coli K-12 defective in D-glucosamine biosynthesis. J Bacteriol 105:467–471
Schrödinger Release (2015) LigPrep, version 3.3, Schrödinger, LLC, New York, NY, 2015 Schrödinger software. http://www.schrodinger.com
Siegel RE (2008) Emerging gram-negative antibiotic resistance: daunting challenges, declining senstivities, and dire consequences. Respir Care 53:471–479
Small-molecule drug discovery suite (2015)–1: Glide, version 6.6, Schrödinger, LLC, New York, NY, 2015
Strokes SS, Albert R, Buurman ET, Andrews B, Shapiro AB, Green OM, Mckenzie AR, Otterbein LR (2012) Inhibitors of the acetyltransferase domain of N-acetylglucosamine-1-phosphate uridylyltransferase/glucosamine-1-phosphate-acetyltransferase (GlmU). Part 2: optimization of physical properties leading to antibacterial aryl sulphonamides. Bioorg Med Chem Lett 22:7019–7023
Usha V, Hobrath JV, Gurcha SS, Reynolds RC, Besra GS (2012) Identification of novel active Mt-Guab2 inhibitor series against M. tuberculosis. PLoS One 7:E33886
Vertesy L, Kurz M, Erb AM, Toti L (2006) Z-phenylbenzofuran derivatives, a process for preparing them, and their use, U.S. Patent 7,148,254, December 12, 2006
Weinstein RA, Gaynes R, Edwards JR (2015) Overview of nosocomial infections caused by gram-negative bacilli. Clin Infect Dis 41:848–854
White RJ (1968) Control of amino sugar metabolism in Escherichia coli and isolation of mutants unable to degrade amino sugars. Biochem J 106:847–858
World Health Organization. (2014) Antimicrobial resistance: global report on surveillance. Geneva, World Health Organisation. Geneva, Switzerland: WHO:2014
Xu ZQ, Flavin MT, Flavin J (2014) Combating multidrug-resistant Gram-negative bacterial infections. Expert Opin Investig Drugs 23:163–182
Zhang JH, Chung TDY, Oldenburg KR (1999) A simple statistical parameter for use in evaluation and validation of high throughput screening assays. J Biomol Screen 4:67–73
Acknowledgments
We are grateful to Dr. Ed T. Buurman (AstraZeneca R&D Boston, USA) for providing us the expression vector pBA750 expressing 1.7 kb H. influenzae glmU and the inhibitor, 1-[2, 4-dimethoxy-5-(10H-phenoxazin-10-ylsulfonyl)phenylamino]-2-(−4-pyridyl)-1-ethanone, for standardizing the GlmU assay.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was funded by Council of Scientific and Industrial Research Indian Council, (CSIR) New Delhi, India (Grant no. BSC0205). Authors (RS, CR, VSR, SK) were provided Junior and Senior Research Fellowship by CSIR and UGC, India.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 100 kb)
Rights and permissions
About this article
Cite this article
Sharma, R., Rani, C., Mehra, R. et al. Identification and characterization of novel small molecule inhibitors of the acetyltransferase activity of Escherichia coli N-acetylglucosamine-1-phosphate-uridyltransferase/glucosamine-1-phosphate-acetyltransferase (GlmU). Appl Microbiol Biotechnol 100, 3071–3085 (2016). https://doi.org/10.1007/s00253-015-7123-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-7123-y