Abstract
Microbial communities provide essential information about host ecology and could be helpful as a tool to improve species conservation efforts. However, microbes can also infect and compromise the host development process and viability. Caretta caretta is the most widespread marine turtle species in the Mediterranean basin and is the only species of sea turtle nesting along the Italian coasts. Little is known about the microbiota composition of the nest of sea turtles and its correlation with hatching failures. In this study, the microbial composition of two nests of C. caretta featuring different rates of hatching success from a nesting beach in Lampedusa (Italy) was analyzed and compared. The bacterial community was determined using culture-dependent methods and next-generation sequencing based on 16S rRNA gene metabarcoding analysis. Our results showed five dominant bacterial phyla (Proteobacteria, Bacteroidetes, Actinobacteria, Verrucomicrobia, and Firmicutes) and indicated different bacterial families (Pseudomonadaceae and Brucellaceae) as likely causes of hatching failures. Besides, our findings demonstrated the nests’ active role in modulating the sand’s bacterial communities. This study suggests microbiological analysis could be a valuable tool in monitoring nests to take preventive actions and reduce hatching failures.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The sea turtle Caretta caretta is the most widespread marine turtle species in the Mediterranean basin and is the only species of sea turtle nesting along the Italian coasts. In the last years, the coastal zones of Italy (i.e., the south zones and the islands) have registered an increase in nesting cases. The assessment of new nesting sites in the Pelagian Islands (i.e., Linosa and Lampedusa) highlighted how C. caretta’s nesting has occurred over the last 40 years [1, 2].
According to the International Union for Conservation of Nature (IUCN), C. caretta is considered a species of least concern [3]. Many factors, particularly anthropic, threaten the conservation of this species during the juvenile and adult ages at sea and the spawning period and embryonic development in coastal nesting areas. The sea and beach pollution, overbuilding, and degradation of the coastal areas, tourism, climate change, presence of predators, rains, floods, and microbial infections are compromising factors for both hatching and survival rates of the newborn [1,2,3,4,5,6,7,8,9,10]). To date, there is an increasing interest in microbial community studies to provide information on the ecology of the host and improve conservation efforts, rescue, and rehabilitation practices. Culture-dependent methods led to the isolation of pathogens from adults [11,12,13] and nests [9, 14] of C. caretta turtles of the Mediterranean Sea. Various bacterial and fungal agents have been described as the major causes of the failure of hatching phenomena of sea turtle eggs, as they can penetrate the outer layer of the eggshell, exploiting embryonic tissues as a source of nutrients [15,16,17]. Among bacteria, the most representatives belong to the genera Pseudomonas, Vibrio, Escherichia, Klebsiella, Enterobacter, Aeromonas, and Salmonella, while Fusarium is mostly considered the fungus responsible for infection of sea turtle eggs and nests [9, 14, 18]. In recent years, studies on microbial communities by next-generation sequencing (NGS) of 16S rRNA gene have been applied to the study of the cloacal, oral, intestinal, and skin microbiota of C. caretta sea turtles in the Mediterranean Sea [19,20,21,22,23]. Regarding nest microbiota, only a study has been carried out by analyzing metagenomic DNA [24], and it was suggested that the maternal and environmental influence alongside a protective role of eggshells shape the egg microbiota of C. caretta sea turtles, as found in other turtles [25, 26]. In the summer of 2020, two C. caretta’s nests located at the beach of Guitgia (Lampedusa, Italy) featured different hatching success rates (0% for nest 1 and 58.90% for nest 2). Guitgia beach, extending for about 4.500 m2, is located in the southeastern part of Lampedusa Island near the downtown and the port (Fig. 1). Although C. caretta’s oviposition has been ascertained since 2018 [27], it is the beach most affected by potential disturbing factors related to mass tourism and flooding phenomena due to rainwater that pours from the surrounding roads onto the beach during heavy rain seasons. The aim of this study was to investigate and compare the microbial composition of the two nests by classical microbiological methods and by 16S rRNA gene metabarcoding to get insights into the failure reasons of C. caretta hatchings.
Experimental Procedures
Site Description and Sampling
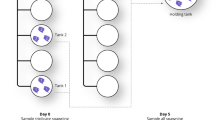
Samples were collected from two nests of the sea turtle C. caretta on the beach of Guitgia (35°29′55.2″N, 12°35′57.3″E) at Lampedusa Island (Sicily, Italy) during the summer of 2020 under the authorization from n. 0006828 04/04/2018 for the years 2018–2020 of the Ministero dell’Ambiente e della tutela del Territorio e del Mare. The two nests were 20 m apart. Eighty of 80 eggs were found unhatched in nest 1; 30/73 unhatched eggs were found (58.9% success rate of hatching) in nest 2 (Table 1).
About 72 h after the last hatchling emerged from nest 2, the nests were dug and examined. Four unhatched eggs from nest 1 and four unhatched eggs and fragments of eggshells of hatched eggs from nest 2 were collected together with four sand samples at a depth of 50 cm from both nests. Four samples of external sand were also collected as a reference. All samples were placed in separate sterile bags and kept at 4 °C until stored at − 20 °C in the laboratory. Before analysis, the egg surface was washed with 15 ml of distilled water to remove loosely attached biofilm. The shell of unhatched eggs was opened aseptically with a sterile scalpel, and the inner membrane was separated using sterilized pliers and transferred into sterile tubes. The details of the analyzed samples are summarized in Table 1.
Bacteriological and Mycological Analyses
Swabs from the eggshells and the internal content of unhatched eggs were taken to isolate bacterial and fungal pathogens. Approximately 1 g of sand samples was incubated in a liquid medium at 37 °C overnight, and 0.1 ml was streaked on agar plates. Three solid culture media were used: blood agar to isolate hemolytic fastidious and non-fastidious bacterial strains, MacConkey agar to select Gram-negative bacteria, thiosulfate-citrate-bile-sucrose agar to isolate Vibrio, tryptone soya agar to isolate environmental strains and Sabouraud dextrose agar to allow the growth of the fungus Fusarium. For bacterial and fungal growth, plates were incubated at 30 °C for 24–48 h and at room temperature for 7 days, respectively. Bacterial colonies were distinguished based on their morphology and subsequently streaked onto fresh plates. As the growth of suspected fungi was visible, a portion of hyphae was collected with a sterile scalpel and used for further analysis and molecular identification.
Molecular Identification of Bacteria and Fungi
The identification of bacterial and fungal isolates was carried out by a molecular approach. DNA was extracted and purified by using the phenol-chloroform extraction reported in [28] from bacterial and fungal suspensions grown for 24 h (bacteria) and 48–72 h (fungi) in agitation (180 rpm) at 30 °C in 10 ml of liquid medium Luria-Bertani (10 g tryptone, 5 g yeast extract, and 10 g NaCl, pH 7.0 ± 0.2, autoclaved for 15 min at 121 °C). Bacterial DNA was used as a template in a PCR reaction to amplify the 16S rRNA gene (1500 bp), using the primers F1 (GAGTTTGATCCTGGCTCAG) and R12 (ACGGCTACCTTGTTACGACT) [29]. Fungal DNA was used to amplify a 600-bp internal fragment of the ITS gene using the primer pair ITS-1 (TCCGTAGGTGAACCTGCGG) and ITS-4 (TCCTCCGCTTATTGATATGC) [30]. The presence of the 16S and ITS amplicons was verified by electrophoresis on 1% w/v agarose gel. The amplicons were purified using the QIAquick PCR Purification Kit (Qiagen, West Sussex, UK) according to the manufacturer’s instructions and quantified at the NanoDrop 2000c spectrophotometer (Thermo Fisher Scientific, MA, USA). The amplicons were sequenced by the Sanger method at BMR Genomics s.r.l. (Padova, Italy). The sequence outputs were analyzed using the alignment tool (BLAST). The sequence dataset was deposited in the GenBank database (OM857961-OM857964; OM860305-OM860307; OM860310, OM860311; OM860313-OM860315; OM860317; OM860320; OM860321).
16S rRNA Gene Metabarcoding
Samples listed in Table 1 were subjected to metagenomic DNA extraction following the protocol reported in [20] with minor modifications. Sand samples and inner membranes (1 g) were incubated in 1 ml of sterile water for 1 h at room temperature (500 rpm). Small fragments of the shell of hatched and unhatched eggs (of the same size as a 50 ml tube stopper) were homogenized by vortexing in 3 ml of sterile water using sterile glass beads and stirred for 1 h at room temperature. Following this incubation step, samples were processed following the protocol. Metagenomic DNA was verified by electrophoresis on 1% w/v agarose gel. The purity and quantity of DNA were assessed using a NanoDrop 2000c spectrophotometer (Thermo Fisher Scientific, MA, USA). An equal amount of the four extracted metagenomic DNAs from each type of samples was pooled. Thus, a total of 8 metagenomic DNA pools were obtained. A 464-bp fragment corresponding to the V3–V4 region of 16S rRNA gene was amplified using the primers Pro341-F (CCTACGGGNBGCASCAG) and Pro805R (GACTACNVGGGTATCTAATCC) [31]. Amplification products were sequenced in one 300-bp paired-end run on an Illumina MiSeq platform at BMR Genomics s.r.l. (Padova, Italy). The raw 16S rDNA data were processed by using the QIIME2 environment [32] as paired-end sequences. In the denoising approach, overlapping paired-end reads were processed with the plug-in DADA2 [33]. Unique Amplicon Sequence Variants (ASVs) were assigned and aligned to the Greengenes reference database at 99% sequence similarity (https://greengenes.secondgenome.com/). QIIME2 was used to generate rarefaction curves, Good’s coverage index, and alpha diversity metrics (Fig. S1 and Table S1, Supplementary Material). Rarefaction analysis was carried out by plotting the number of the observed ASVs against the total number of filtered reads for each sample. The number of ASVs and the percentage of the relative abundances of different taxonomic levels were determined. Based on the rarefaction curve, the alpha diversity metrics were calculated on a rarefied frequency-feature table with a minimum number of 13,965 sequences per sample. The sequence dataset was deposited in the GenBank database (BioProjectID: PRJNA804141).
Results and Discussion
Presence of Fusarium spp.
The search for the presence of Fusarium in all our samples showed that, independently from the nest, two genera of Fusarium, i.e., F. solani and F. falciforme, were present. According to several reports [9, 15,16,17], fungi of the genus Fusarium were recognized as primary causes of death and reduced hatching success rates in sea turtles. Although the shell represents a protective barrier from the outer environment, it does not completely inhibit the passage of fungi, which produce lipolytic and proteolytic enzymes and penetrate the shell layers, causing the reduction of respiratory gas exchange, decreasing the availability of calcium for developing embryos, and using developing tissues as nutrient sources [34]. Specifically, F. solani was isolated from the shell of unhatched eggs of nest 1 and the shell of hatched eggs of nest 2, while F. falciforme was present in the internal content of unhatched eggs of nest 1 and the shell of unhatched eggs of nest 2. Although Fusarium was not found inside the eggs of nest 2, its effect on egg development and hatchings could occur without direct contact with the eggs. In fact, some species of Fusarium are known to produce volatile mycotoxins or other metabolites that affect adjacent eggs influencing embryo development [34, 35]. Our results strongly suggest that Fusarium spp. did not represent the only cause of the mortality of eggs in nest 1 since the two genera of fungi were found in both nests. Therefore, a difference in bacterial abundance of the two nests might have a more predominant role in hatching success.
Bacteriological Analysis
Interestingly, the search for bacterial pathogens showed different bacteria present inside the nests (Table 2).
The most striking feature was the presence of Pseudomonas in all the samples analyzed from nest 1 (sand, eggshells, and inner membranes). Differently, Pseudomonas was never isolated from sand, eggshells, and inner membranes from nest 2 and from the sand collected outside of both the nests. It was isolated only from fragments of the hatched eggs of nest 2, suggesting colonization after the hatching event. Even if these analyses are not quantitatively supported, we could surmise that Pseudomonas could have affected egg development. Indeed, Pseudomonas has been frequently associated with the failure of hatchings in several turtle species [14, 36, 37]. As described below, 16S rRNA gene metabarcoding confirmed that the Pseudomonadaceae family was abundant in nest 1, even if it was also found in the sand outside the nests. Sand from the two nests and sand outside contained Bacillus sp., which is commonly found in the soil. The sand of nest 2 also contained Lysinibacillus sp. Some species of Lysinibacillus produce secondary metabolites that act as very potent antimicrobial compounds [38]; thus, its presence in nest 2 could have had a role in protecting the development and hatching of the eggs. The second striking difference was the detection of Brucella only in nest 1. Brucella is an opportunistic pathogen present in various environments and is becoming a growing cause of serious infections [39]. It has been isolated from the eggs of sea turtle nests in other reports [14, 40, 41]. Its role in the failure of hatchings can be surmised, since 16S rRNA gene metabarcoding revealed its presence in all samples, except in the sand external to the nest; thus, we hypothesize that it derives from the mother. To our surprise, we have not isolated Salmonella, Aeromonas, Citrobacter, and Vibrio, which have been frequently isolated from the egg interior of unhatched C. caretta sea turtle eggs [8, 9, 14].
Microbial Composition and Biodiversity by 16S rRNA Gene Metabarcoding
High-quality reads (125,568) from the 320,073 raw reads were obtained. After filtration, denoising, and merging, 1688 ASVs (amplicon sequence variants) were identified using the suite QIIME2 (Table S1, Supplementary material). The rarefaction curves (Fig. S1, Supplementary Material) based on the comparison of ASVs abundances and the number of sequences showed that the analyses performed were representative of the communities under investigation, as confirmed by Good’s coverage index (an average of 1 for all the samples) (Table S1, Supplementary material). ASVs were classified at different taxa levels: phylum, class, order, and family (Fig. S2, Supplementary Material). The most abundant phyla in both nests were Proteobacteria, Bacteroidetes, Verrucomicrobia, Actinobacteria, and Firmicutes. Bacteria belonging to other phyla (such as Fusobacteria, Acidobacteria, Chloroflexi) were minor components and were not present in all samples. To the best of our knowledge, only one study has been carried out on the microbiota of sea turtle eggs of the species C. caretta which is mainly dominated by Proteobacteria, Actinobacteria, Firmicutes, Bacteroidetes, and Verrucomicrobia [24], as we found in our study with differences in the percentages of the phyla. The heat map of the bacterial families (Fig. 2) showed that the external sand was apart from all the samples of both nests, suggesting an active role of the sea turtle eggs in shaping the microbial community of the nesting site [17]. The external sand differed from the sample sands inside the two nests and showed a greater diversity, as suggested by the different ASVs found in each type of sample (Table S1, Supplementary material). Indeed, the external sand (813) contained at least 2.5-fold more ASVs than other sand samples (306 and 197 in sand inside nest 1 and nest 2, respectively). Furthermore, the Shannon-Wiener diversity index (H′) was 4.38 ± 2.15 for sand inside the nests and 7.93 for the external sand (Table S1, Supplementary material). This aspect could be ascribed to the environmental conditions (incubation, temperature, and humidity) of the nesting chambers that could favor the colonization of specific bacterial strains and discourage others. To our surprise, the heatmap showed that all the other samples clustered into two main groups, one corresponding to the samples of nest 1, and the other one to nest 2, again strongly suggesting an active effect of the eggs on the surrounding microbial communities of the nest.
Bacterial Composition of Sand Samples
In the sand outside the nests, a greater diversity of the low abundant bacterial taxa than in the sand collected inside the two nests was registered. The minor bacterial components are indicated as other in Fig. 3.
Proteobacteria is the predominant phylum in the microbiota of all the sand samples (49.34–57.35%), in accordance with recent reports [24, 42]. Sand outside the nests and that one inside nest 1 contained Pseudomonadaceae and Phyllobacteriaceae families that were completely absent in the sand inside nest 2; sand inside both nests was more similar to each other, as foreseen by heatmap analysis; both nests contained Flavobacteriaceae, Verrucomicrobiaceae, Brucellaceae, and Xanthomonadaceae. This finding confirms that Pseudomonas, found by microbiological assay from nest 1 and sand outside, could be responsible for the failure of hatchings. We can infer that it was present in the beach sand, and it was able to colonize only samples of nest 1 because of environmental humidity and to penetrate the eggs of nest 1; in fact, it was abundant also in the shells and the inner membrane of the eggs of nest 1 (Fig. 4). As reported above, Pseudomonas has been frequently associated with the failure of hatchings in several turtle species [14, 36, 37]. The role of Phyllobacteriaceae is difficult to predict since a few reports describe its presence upon lipopolysaccharide instillation [43] and its increase in patients affected by esophageal squamous cell carcinoma [44]. Differently from the sand outside the nests, both the sands inside the nests were enriched with Bacteroidetes (29.3 and 17.89%, respectively) indicating a maternal transmission during the egg passage through the oviduct or cloaca, as already suggested [45]. Indeed, Bacteroidetes are usually less abundant in the sand and on the eggshells than inside the eggs and the cloacal samples [15, 20, 46]. Interestingly, a large abundance of Actinobacteria was registered in the sand of nest 2 (11.04%) and outside of the nests (11.84%) with respect to the sand of nest 1 containing only 2.20%. Actinobacteria produce a plethora of biologically active secondary metabolites with antibacterial and antifungal activity; thus, we could hypothesize that some elements of the nest inhibited their growth with the resulting reduction of protection of the eggs from microbial attacks [15].
Each sand sample featured specific phyla; Fusobacteria was present only in the sand inside nest 1, Verrucomicrobia was highly represented in the sand of nest 2, while the sand outside the nests contained Acidobacteria, Chloroflexi, Gemmatimonadetes, and Nitrospirae (2.87–8.76%) that were almost absent inside both nests. Usually, Fusobacteria represent a dominant phylum in the microbiota of vertebrates that feed on carrion, i.e., alligators and vultures [20, 47], and we could hypothesize that its abundance inside nest 1 was due to a premature egg death that created the proper environmental conditions for its colonization.
Bacterial Content of Eggshells
In this study, two types of eggshells were analyzed: those deriving from the unhatched eggs, found in both the nests and fragments of eggshells found inside nest 2 after digging. These latter could derive from healthy sea turtles after the hatching event. Proteobacteria are the predominant phylum (71.19–95.81%) in turtle eggshells (Fig. 4).
As sands contained less abundance of Proteobacteria, we could surmise that bacteria belonging to this phylum have found proper growth conditions in the nests or that a maternal influence occurred. Studies of the microbiota of turtle nests have generally reported similar, but not identical, results to ours. For example, Proteobacteria, even if represented the predominant phylum, were more abundant in our study than in other reports on C. caretta [24] and Eretmochelys imbricata nests [15]. Among Proteobacteria, the Brucellaceae family entirely colonized the shells of the eggs of nest 2, while Pseudomonadaceae the nest 1. Differently, the shells of the hatched eggs showed a homogeneous bacterial distribution with a reduction of the Brucellaceae and Pseudomonadaceae families. These results strongly suggest that different bacteria affected egg development in the two nests. Although we cannot rule out that the bacterial colonization of the fragments of eggshells occurred after the hatchings in which the shells could have lost their active and protective role, together with the loss of the antimicrobial molecules inside the yolk, we suppose that Pseudomonas colonization derived from the sand while Brucella could be transmitted by the mother.
Verrucomicrobiaceae and Flavobacteriaceae were present in the eggshells of the hatched eggs of nest 2 and the unhatched eggs of nest 1. The Verrucomicrobiaceae family encodes a wide variety of glycoside hydrolases, sulfatases, peptidases, carbohydrate lyases, and esterases that could be used to metabolize released nutrients [46]. Flavobacteriaceae are commonly associated with urban environments, sewage-polluted waters, and stormwater since many members of this family need environments containing complex carbon compounds [48].
Bacterial Content of Inner Membranes of the Unhatched Eggs
At the phylum level, the most evident differences were the large abundance of Bacteroidetes and Firmicutes in the inner membrane of eggs of nest 1; indeed, Proteobacteria, Bacteroidetes, Firmicutes, and Actinobacteria were found in nest 1 while Proteobacteria, Bacteroidetes, Actinobacteria, and Verrucomicrobia in nest 2 (Fig. 5). Firmicutes and Bacteroidetes are found in the maternal oviduct and are less represented in the sand and seawater [21, 45]. In addition, Bacteroidetes and Firmicutes are the most represented phyla in animal and human gut microbiota [19, 20, 49]. Therefore, our hypothesis is that possible municipal wastewater could have contaminated nest 1, favoring the proliferation of Firmicutes and Bacteroidetes and disfavoring the normal proliferation of Actinobacteria and Verrucomicrobia, bacterial phyla typical of the sandy environment [50]. Thus, their presence in the inner membranes of the unhatched eggs could derive from the contaminated sand.
At the family level, analysis of the inner membranes of the unhatched eggs confirmed different colonization of the eggs, Pseudomonadaceae and Flavobacteriaceae (28% and 26 %) in nest 1 and Brucellaceae in nest 2 (71%). Bacteroidetes, including the Flavobacteriaceae family, and some families belonging to Firmicutes, such as Peptostreptococcaceae, Lachnospiraceae, and Clostridiaceae, are considered fecal markers, especially in humid growth conditions [51, 52]. Flavobacteriaceae are commonly associated with urban environments, sewage-polluted waters, and stormwater since many members of this family need environments containing complex carbon compounds [48]. Brucellaceae has been isolated in sea turtles with conjunctivitis infection [53] and in the nest of Chelonia mydas [41] and Caretta caretta turtles [14]. Members of the genus Brucella have recently been identified in several species of cetaceans and pinnipeds, and a Brucella species was isolated from the aborted fetal tissue of bottlenose dolphins along the California coast [54]. Thus, we surmise that 59% of the unsuccessful hatching of the eggs of nest 2 could be associated with this bacterial genus.
Conclusions
The analyses of the microbial community in C. caretta nests could be used a useful tool to preserve the conservation of the species and increase hatching success. The present study is based on the analysis of two nests of C. caretta on the beach of Guitgia of Lampedusa, with a different success rate of hatching: 0% for nest 1 and 59% for nest 2. Combining the results of this report with our previous work conducted on nests from other sites, our findings suggest a crucial role for the sand and show that each nest has its microbial profile. Indeed, the sand inside the nests displayed a lower bacterial diversity than the sand outside, and different bacterial phyla could be responsible for the hatching failure of both nests. Although we cannot rule out the involvement of Fusarium, in this study, we considered Pseudomonas and Brucella the key players governing the hatching fate, with Pseudomonas deriving from the sand of the beach and Brucella from the mother. Limitations of the microbiological culture and 16S rRNA metabarcoding have to be taken into consideration pointing out that both methods should be carried out to investigate microbial community; however, it is noteworthy that bacteria detectable by DNA-based methods could not be viable and metabolically active organisms, even if DNA is detectable. Thus, periodic and preventive monitoring of microbial content of sand and nests could be helpful for nest management and protection activities. As an example, the identification of known pathogens in the sand could lead to moving the nest to another site, eliminating some threats to the conservation of the species.
Data Availability
The datasets analyzed during the current study are available in the GenBank database with the accession numbers OM857961-OM857964; OM860305-OM860307; OM860310, OM860311; OM860313-OM860315; OM860317; OM860320; OM860321 and BioProject ID: PRJNA804141.
References
Prazzi E, Nicolini G, Piovano S, Giacoma C (2013) La spiaggia dei Conigli a Lampedusa: un modello di gestione per la conservazione di Caretta caretta. In: Di Tizio L, Brugnola L, Cameli A, Di Francesco N (Eds.), Atti Il Congresso SHI Abruzzo Molise “Testuggini e Tartarughe” (Chieti, 27–29 settembre 2013). Ianieri Edizoni, Pescara, pp. 127–133.
Prazzi E, Giacoma C (2018) Nidificazione di Caretta caretta (Reptilia Cheloniidae) sull’isola di Lampedusa nel 2016, con ritrovamento di due nuovi siti di deposizione. Naturalista sicil, S IV, XLII 1:111–124
Casale P (2015) Caretta caretta (Mediterranean subpopulation). IUCN Red List Threat Species. 8235: e. T83644804A83646294. Available: https://doi.org/10.2305/IUCN.UK.2015-4.RLTS.T83644804A83646294.en
Alessandro L, Antonello S (2010) An overview of loggerhead sea turtle (Caretta caretta) bycatch and technical mitigation measures in the Mediterranean Sea. Rev Fish Biol Fish 20:141–161. https://doi.org/10.1007/s11160-009-9126-1
Blasi MF, Mattei M (2017) Seasonal encounter rate, life stages and main threats to the loggerhead sea turtle (Caretta caretta) in the Aeolian Archipelago (southern Thyrrenian Sea). Aquat Conserv Mar Freshwat Ecosyst 27(3):617–630. https://doi.org/10.1002/aqc.2723
Matiddi M, Hochsheid S, Camedda A, Baini M, Cocumelli C, Serena F et al (2017) Loggerhead sea turtles (Caretta caretta): a target species for monitoring litter ingested by marine organisms in the Mediterranean Sea. Environ Pollut 230:199–209. https://doi.org/10.1016/j.envpol.2017.06.054
Caracappa S, Persichetti MF, Piazza A, Caracappa G, Gentile A, Marineo S et al (2018) Incidental catch of loggerhead sea turtles (Caretta caretta) along the Sicilian coasts by longline fishery. PeerJ. 7(6):e5392. https://doi.org/10.7717/peerj.5392
Alduina R, Gambino D, Presentato A, Gentile A, Sucato A, Savoca D, et al. (2020) Is Caretta Caretta a carrier of antibiotic resistance in the Mediterranean Sea? Antibiotics (Basel) 10;9(3):116. https://doi.org/10.3390/antibiotics9030116.
Gambino D, Persichetti MF, Gentile A, Arculeo M, Visconti G, Currò V et al (2020) First data on microflora of loggerhead sea turtle (Caretta caretta) nests from the coastlines of Sicily. Biol Open 9(1):bio045252. https://doi.org/10.1242/bio.045252
Blasi MF, Hochscheid S, Bardelli R, Bruno C, Melodia C, Salzeri P et al (2022) First report on two loggerhead turtle (Caretta caretta) nests in the Aeolian Archipelago (Southern Italy). Acta Herpetologica 17(1):37-43.doi
Fichi G, Cardeti G, Cersini A, Mancusi C, Guarducci M, Di Guardo G et al (2016) Bacterial and viral pathogens detected in sea turtles stranded along the coast of Tuscany, Italy. Vet Microbiol 185:56–61. https://doi.org/10.1016/j.vetmic.2016.02.003
Blasi MF, Migliore L, Mattei D, Rotini A, Thaller MC, Alduina R (2020) Antibiotic resistance of gram-negative bacteria from wild captured loggerhead sea turtles. Antibiotics (Basel) 6;9(4):162. https://doi.org/10.3390/antibiotics9040162.
Trotta A, Marinaro M, Sposato A, Galgano M, Ciccarelli S, Paci S et al (2021) Antimicrobial resistance in loggerhead sea turtles (Caretta caretta): a comparison between clinical and commensal bacterial isolateS. Animals (Basel) 11(8):2435. https://doi.org/10.3390/ani11082435
Craven KS, Awong-Taylor J, Griffiths L, Bass C, Muscarella M (2007) Identification of bacterial isolates from unhatched loggerhead (Caretta caretta) sea turtle eggs in Georgia, USA. Mar Turt Newsl 115:9–11
Sarmiento-Ramírez JM, van der Voort M, Raaijmakers JM, Diéguez-Uribeondo J (2014) Unravelling the microbiome of eggs of the endangered sea turtle Eretmochelys imbricata identifies bacteria with activity against the emerging pathogen Fusarium falciforme. PLoS One 9(4):e95206. https://doi.org/10.1371/journal.pone.0095206
Cafarchia C, Paradies R, Figueredo LA, Iatta R, Desantis S, Di Bello AVF et al (2020) Fusarium spp. in loggerhead sea turtles (Caretta caretta): from colonization to infection. Vet Pathol 57(1):139–146. https://doi.org/10.1177/0300985819880347
Hoh DZ, Lin Y, Liu W, Sidique S, Tsai IJ (2020) Nest microbiota and pathogen abundance in sea turtle hatcheries. Fungal Ecol 47. https://doi.org/10.1016/j.funeco.2020.100964
Gleason FH, Allerstorfer M, Lilje O (2020) Newly emerging diseases of marine turtles, especially sea turtle egg fusariosis (SEFT), caused by species in the Fusarium solani complex (FSSC). Mycology. 11(3):184–194. https://doi.org/10.1080/21501203.2019.1710303
Abdelrhman K, Bacci G, Mancusi C, Mengoni A, Serena F, Ugolini A (2016) A first insight into the gut microbiota of the sea turtle Caretta caretta. Front.Microbiol. https://doi.org/10.3389/fmicb.2016.01060
Arizza V, Vecchioni L, Caracappa S, Sciurba G, Berlinghieri F, Gentile A, et al (2019) New insights into the gut microbiome in loggerhead sea turtles Caretta caretta stranded on the Mediterranean coast. PLoS One 14;14(8):e0220329. https://doi.org/10.1371/journal.pone.0220329.
Biagi E, D'Amico F, Soverini M, Angelini V, Barone M, Turroni S et al (2019) Faecal bacterial communities from Mediterranean loggerhead sea turtles (Caretta caretta). Environ Microbiol Rep 11(3):361–371. https://doi.org/10.1111/1758-2229.12683
Filek K, Trotta A, Gračan R, Di Bello A, Corrente M, Bosak S (2021) Characterization of oral and cloacal microbial communities of wild and rehabilitated loggerhead sea turtles (Caretta caretta). Anim Microbiome 3;3(1):59. https://doi.org/10.1186/s42523-021-00120-5
Blasi MF, Rotini R, Bacci T, Targusi M, Bonanno Ferraro G, Vecchioni L, et al (2021) On Caretta caretta’s shell: first spatial analysis of micro- and macro-epibionts on the Mediterranean loggerhead sea turtle carapace, Mar Biol Res 17:7-8, 762-774. https://doi.org/10.1080/17451000.2021.2016840
Vecchioni L, Pace A, Sucato A, Berlinghieri F, Cambera I, Visconti G et al (2022) Unveiling the egg microbiota of the loggerhead sea turtle Caretta caretta in nesting beaches of the Mediterranean Sea. PLoS One 17(5):e0268345. https://doi.org/10.1371/journal.pone.0268345
Rafferty AR, Santidrián Tomillo P, Spotila JR, Paladino FV, Reina RD (2011) Embryonic death is linked to maternal identity in the leatherback turtle (Dermochelys coriacea). PLoS One 6(6):e21038. https://doi.org/10.1371/journal.pone.0021038
Bezy VS, Valverde RA, Plante CJ (2015) Olive ridley sea turtle hatching success as a function of the microbial abundance in nest sand at Ostional. Plos One, Costa Rica. https://doi.org/10.1371/journal.pone.0118579
Prazzi E (2022) Nesting activity of Loggerhead sea turtle Caretta caretta (Linnaeus, 1758) (Reptilia Cheloniidae) from 2018 to 2021 on Lampedusa Island (Pelagie Islands, Sicily Channel). Biodiversity Journal 13(1):141–144
Presentato A, Lampis S, Vantini A, Manea F, Daprà F,Zuccoli S, et al. (2020) On the Ability of perfluorohexane sulfonate (PFHxS) bioaccumulation by two Pseudomonas sp. strains isolated from PFAS-contaminated environmental matrices. Microorganisms 8(1):92 https://doi.org/10.3390/microorganisms8010092.
Coy MR, Hoffmann M, Kingdom Gibbard HN, Kuhns EH, Pelz-Stelinski KS, Stelinski LL (2014) Nested-quantitative PCR approach with improved sensitivity for the detection of low titer levels of Candidatus Liberibacter asiaticus in the Asian citrus psyllid, Diaphorina citri Kuwayama. J Microbiol Methods 102:15–22. https://doi.org/10.1016/j.mimet.2014.04.007
White T, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications (ed. Innis M., Gelfand D., Shinsky J. and White T.) pp. 315-322. Academic Press.
Takahashi K, Nishida A, Fujimoto T, Fujii M, Shioya M, Imaeda H et al (2016) Reduced abundance of butyrate-producing bacteria species in the fecal microbial community in Crohn’s disease. Digestion. 93:59–65. https://doi.org/10.1159/000441768
Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet CC et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Callahan B, McMurdie P, Rosen M, Wan AW, Johnson AJA et al (2016) DADA2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13:581–583. https://doi.org/10.1038/nmeth.3869
Phillott AD, Parmenter CJ (2014) Fungal colonization of green sea turtle (Chelonia mydas) nests is unlikely to affect hatchling condition. Herpetol Conserv Biol 9(2):297–301
Elshafie A, Al-Bahry SN, AlKindi AY, Ba-Omar T, Mahmoud I (2007) Mycoflora and aflatoxins in soil, eggshells, and failed eggs of chelonia mydas at Ras Al-Jinz, Oman. Chelonian Conserv Biol 6(2):267–270. https://doi.org/10.2744/1071-8443(2007)6[267:MAAISE]2.0.CO;2
Al-Bahry S, Mahmoud I, Elshafie A, Al-Harthy A, Al-Ghafri S, Al-Amri I et al (2009) Bacterial flora and antibiotic resistance from eggs of green turtles Chelonia mydas: an indication of polluted effluents. Mar Pollut Bull 58(5):720–725. https://doi.org/10.1016/j.marpolbul.2008.12.018
Keene E, Soule T, Paladino FV (2014) Microbial isolations from Olive Ridley (Lepidochelys olivacea) and East Pacific Green (Chelonia mydas agassizii) sea turtle nests in Pacific Costa Rica, and testing of cloacal fluid antimicrobial properties. Chelonian Conserv Biol 13(1):49–55. https://doi.org/10.2744/CCB-1051.1
Naureen Z, Rehman NU, Hussain H, Hussain J, Gilani SA, Al Housni SK et al (2017) Exploring the potentials of Lysinibacillus sphaericus ZA9 for plant growth promotion and biocontrol activities against phytopathogenic fungi. Front Microbiol 8:1477. https://doi.org/10.3389/fmicb.2017.01477
Ryan MP, Pembroke JT (2020) The genus Ochrobactrum as major opportunistic pathogens. Microorganisms. 8(11):1797. https://doi.org/10.3390/microorganisms8111797
Awong-Taylor J, Craven KS, Griffiths L, Bass C, Muscarella M (2008) Comparison of biochemical and molecular methods for the identification of bacterial isolates associated with failed loggerhead sea turtle eggs. J Appl Microbiol 104(5):1244–1251. https://doi.org/10.1111/j.1365-2672.2007.03650.x
Candan O, Candan ED (2020) Bacterial diversity of the green turtle (Chelonia mydas) nest environment. Sci Total Environ 720. https://doi.org/10.1016/j.scitotenv.2020.1377
Scheelings TF, Moore RJ, Van TTH, Klaassen M, Reina RD (2020) Microbial symbiosis and coevolution of an entire clade of ancient vertebrates: the gut microbiota of sea turtles and its relationship to their phylogenetic history. Anim Microbiome 2:17. https://doi.org/10.1186/s42523-020-00034-8
Sze MA, Tsuruta M, Yang SW, Oh Y, Man SF, Hogg JC et al (2014) Changes in the bacterial microbiota in gut, blood, and lungs following acute LPS instillation into mice lungs. PLoS One 9(10):e111228. https://doi.org/10.1371/journal.pone.0111228
Xu L, Qi Y, Jiang Y, Ji Y, Zhao Q, Wu J et al (2022) Crosstalk between the gut microbiome and clinical response in locally advanced thoracic esophageal squamous cell carcinoma during neoadjuvant camrelizumab and chemotherapy. Ann Transl Med 10(6):325. https://doi.org/10.21037/atm-22-1165
Sucato A, Vecchioni L, Savoca D, Presentato A, Arculeo M, Alduina R (2021) A comparative analysis of aquatic and polyethylene-associated antibiotic-resistant microbiota in the mediterranean sea. Biology. 10:1–13. https://doi.org/10.3390/biology10030200
Martinez-Garcia M, Brazel DM, Swan BK, Arnosti C, Chain PS, Reitenga KG et al (2012) Capturing single cell genomes of active polysaccharide degraders: an unexpected contribution of Verrucomicrobia. PLoS One 7(4):e35314. https://doi.org/10.1371/journal.pone.0035314
Mendoza MLZ, Roggenbuck M, Vargas KM, Hansen LH, Brunak S, Gilbert MTP et al (2018) Protective role of the vulture facial skin and gut microbiomes aid adaptation to scavenging. Acta Vet Scand 60. https://doi.org/10.1186/s13028-018-0415-3
McClary-Gutierrez JS, Driscoll Z, Nenn C, Newton RJ (2021) Human fecal contamination corresponds to changes in the freshwater bacterial communities of a large river basin. Microbiol Spectr 9(2):e0120021. https://doi.org/10.1128/Spectrum.01200-21
Galluzzo P, Capri FC, Vecchioni L, Realmuto S, Scalisi L et al (2021) Comparison of the intestinal microbiome of Italian patients with multiple sclerosis and their household relatives. Life (Basel) 11(7):620. https://doi.org/10.3390/life11070620
Freitas S, Hatosy S, Fuhrman JA, Huse SM, Welch DB, Sogin ML et al (2012) Global distribution and diversity of marine Verrucomicrobia. ISME J 6(8):1499–1505. https://doi.org/10.1038/ismej.2012.3
Heaney CD, Sams E, Dufour AP, Brenner KP, Haugland RA, Chern E et al (2012) Fecal indicators in sand, sand contact, and risk of enteric illness among beachgoers. Epidemiology. 23(1):95–106. https://doi.org/10.1097/EDE.0b013e31823b504c
Teixeira P, Dias D, Costa S, Brown B, Silva S, Valerio E (2020) Bacteroides spp. And traditional fecal indicator bacteria in water quality assessment-an integrated approach for hydric resources management in urban centers. J Environ Manag 271. https://doi.org/10.1016/j.jenvman.2020.110989
Di Ianni F, Dodi PL, Cabassi CS, Pelizzone I, Sala A, Cavirani S et al (2015) Conjunctival flora of clinically normal and diseased turtles and tortoises. BMC Vet Res 11:91. https://doi.org/10.1186/s12917-015-0405-x
House C, Aguirre AA, House JA (2002) Emergence of infectious diseases in marine mammals. In: Conservation Medicine: Ecological Health in Practice, Aguirre AA, Ostfield R, Tabor G, House C, Pearl M (editors), et al. New York:Oxford University Press, pp 104–117.
Acknowledgements
The study is part of the research project named “Monitoring and protection of Caretta caretta nests on Lampedusa island” authorized by the Ministry of the Environment and carried out by Legambiente Sicilia as the managing body of the Nature Reserve “Isola di Lampedusa.”
Funding
Open access funding provided by Università degli Studi di Palermo within the CRUI-CARE Agreement. This research was partly funded by Legambiente Sicilia and FFR160878 and Ministero della Salute (project n. IZS SI 07/20).
Author information
Authors and Affiliations
Contributions
Fanny Claire Capri carried out the metagenomic analysis and molecular analysis of isolated bacteria and fungi, analyzed the data, and wrote the draft of the manuscript; Elena Prazzi and Giulia Casamento monitored the nests and carried out the collection of the samples; Delia Gambino and Giovanni Cassata performed bacteriological and fungal isolation; Rosa Alduina conceived the study, interpreted the data, and revised the article. All the authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Supplementary Information

Figure S1.
Rarefaction curves on sequencing data obtained from different compartments. The number of observed characteristics (representative of the ASVs) found in each sample is reported as a function of the sequencing effort. The asymptotic trend of the curves indicates that the number of readings generated is representative of the entire community. (PNG 988 kb)

Figure S2.
Relative abundance (%) of phyla (A), class (B), order (C), and 25 most abundant families (D) detected in the sand, fragments of eggshells of hatched and unhatched eggs, and inner membrane of sea turtles’ eggs of two nests. (PNG 2313 kb)
Table S1.
Bioinformatic analyses of obtained reads: filtering, denoising, and merging results. (PDF 153 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Capri, F.C., Prazzi, E., Casamento, G. et al. Correlation Between Microbial Community and Hatching Failure in Loggerhead Sea Turtle Caretta caretta. Microb Ecol 86, 1923–1933 (2023). https://doi.org/10.1007/s00248-023-02197-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-023-02197-8