Abstract
In recent years, imaging has become increasingly important to confirm diagnosis, monitor disease activity, and predict disease course and outcome in children with juvenile idiopathic arthritis (JIA). Over the past few decades, great efforts have been made to improve the quality of diagnostic imaging and to reach a consensus on which methods and scoring systems to use. However, there are still some critical issues, and the diagnosis, course, and management of JIA are closely related to clinical assessment. This review discusses the main indications for conventional radiography (XR), musculoskeletal ultrasound (US), and magnetic resonance imaging (MRI), while trying to maintain a clinical perspective. The diagnostic-therapeutic timing at which one or the other method should be used, depending on the disease/patient phenotype, will be assessed, considering the main advantages and disadvantages of each imaging modality according to the currently available literature. Some brief clinical case scenarios on the most frequently and severely involved joints in JIA are also presented.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Juvenile idiopathic arthritis (JIA) is the most common chronic rheumatic disease of childhood [1]. It has been defined according to the American College of Rheumatology (ACR) criteria, which require an age less than 16 years at onset and persistence of arthritis for a period of at least 6 weeks, following the exclusion of alternative potential causes of arthritis [2]. The current International League of Associations for Rheumatology (ILAR) classification for JIA is based on the extent of disease in the first six months and recognizes seven disease categories, summarized in Table 1.
Imaging plays a pivotal role in identifying the presence, severity, and extent of musculoskeletal involvement. It helps in monitoring disease complications, ruling out alternative diagnoses, and evaluating the response to treatment [3, 4].
Nevertheless, interpretation of skeletal imaging in children can be challenging due to the developing nature of the growing skeleton. It requires substantial experience and effort to effectively differentiate between healthy subjects and those with disease-related changes. Establishing reference standards and robust scoring systems tailored to the developmental stage of children remains an ongoing endeavor [5].
Although the European League Against Rheumatism (EULAR)/Pediatric Rheumatology European Society (PRES) imaging task force has recently highlighted the potential role of imaging in the diagnosis, progression, and monitoring of treatment efficacy in various aspects of JIA, the clinician’s primary focus remains the interweaving between an excellent clinical examination and the judicious use of laboratory and imaging techniques [6].
In this review, we aim to provide clarity on which patients and joint imaging can be used and at what time they should be used. We will outline the main indications for various imaging techniques depending on the joint and purpose, always keeping the clinical aspect in mind. We will also highlight the advantages and disadvantages associated with these imaging techniques.
State of play on imaging in juvenile idiopathic arthritis
Considerable progress has been made in recent decades to improve the quality of diagnostic imaging and to reach a consensus on the methods and scoring systems to be used.
The latest OMERACT (Outcome Measures in Rheumatology Clinical Trials) Filter, uploaded in version 2.1, provided the tools to assess the quality of an imaging technique by evaluating the outcomes against the three “pillars” of truth, discrimination, and feasibility, as well as their ability to discriminate between different degrees of disease [7].
Over the past years, several international collaborative research groups on JIA imaging have emerged. In 2018, Nusman et al. [8] provided an overview of ongoing international initiatives, their focus, and imaging-related outcomes in JIA. Some study groups concentrate on all available imaging modalities (e.g., the ACR’s Pediatric Rheumatology Working Group or the EULAR-PRES Task Force) and encompass all joints. Other groups specialize in magnetic resonance imaging (MRI) (such as the OMERACT group, subdivided into three subgroups for small joints, large joints, and the temporomandibular joint [TMJ]) or in ultrasound (US) (e.g., the Childhood Arthritis and Rheumatology Research AllianceUS Group) [8].
Experts’ opinion: critical points
The potential use of imaging studies in modern pediatric rheumatology practice, alongside its main challenges in JIA, remains controversial in both clinical practice and research. This ambiguity can be attributed to several factors, such as the absence of standardized imaging protocols specifically designed for pediatric populations and the difficulty associated with evaluating tissues that are still maturing and developing. While highlighting the higher sensitivity of musculoskeletal US and MRI in detecting inflammation in asymptomatic joints and their potential aid in early diagnosis, it is important to note that a significant challenge remains: the need for consensus on fundamental lesions and the standardization of imaging protocols [4, 9,10,11].
In addition, MRI should be optimized as a robust biomarker and outcome measure. Nevertheless, its application for treatment monitoring is still limited to clinical trials, as estimates of bone erosion and cartilage loss in children remain inherently imprecise. MRI is known to be constrained by several limitations, including cost, accessibility, and patient acceptance [12, 13].
Regarding the prognostic value of musculoskeletal US in predicting disease flare-up and planning therapeutic strategy, it remains unclear whether subclinical synovitis carries the risk of silent progression of joint damage and should influence the clinician’s decision to discontinue treatment [14].
Finally, clinical questions about the timing of imaging examinations, the selection of appropriate imaging modalities, and how many joints should be evaluated remain partially unresolved.
When should imaging be used in juvenile idiopathic arthritis?
Diagnosis
In adult patients, MRI and musculoskeletal US have been integrated into diagnostic algorithms to assess the extent of joint involvement, as outlined in the 2010 revised diagnostic criteria for rheumatoid arthritis, increasing diagnostic accuracy, particularly in early disease stages [15]. In addition, Duer-Jensen et al. [16] regarded MRI bone marrow edema and the combined synovitis and erosion pattern as valuable indicators for diagnosing rheumatoid arthritis.
Contrastingly, a specific imaging signature for JIA has not yet been described. Up to now, no study has specifically addressed the role of MRI in the diagnosis of JIA, although several papers have reported that MRI can assist physical examination in the early differentiation of childhood arthritis.
Even though the diagnosis of JIA remains one of exclusion, based solely on clinical criteria, imaging is increasingly being used to help to confirm the diagnosis.
Conventional radiography (XR) can reveal early-stage manifestations of the disease, including soft tissue swelling and periostitis. However, these findings are not exclusive to arthritis, and their sensitivity is very limited, especially in detecting early-stage disease [17].
Musculoskeletal US is of particular importance as it can detect subclinical synovitis and improve the classification of patients into different JIA subtypes [18].
MRI provides distinct advantages over clinical evaluation, particularly when assessing specific joints, due to its capability to image the TMJ and the axial skeleton, for which MRI serves as a reference method for detecting early changes [19,20,21,22,23].
Initial confirmation of the clinical diagnosis and differential diagnoses
In cases where the history or clinical findings are inconclusive, XR, musculoskeletal US, or MRI may be used to improve the certainty or uncertainty of a JIA diagnosis beyond clinical features and to narrow the differential diagnosis [6].
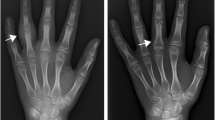
In the early stages of disease, XR has its place in excluding other bone pathology, such as trauma, tumors, avascular necrosis, bone dysplasia, osteomyelitis, and other bone marrow or hematological disorders affecting joints, such as leukemia or hemophilia [24, 25]. XR is crucial for the early detection of disease-related damage, detecting subclinical changes that may have occurred prior to diagnosis, such as soft tissue thickening, joint effusion, periarticular changes, and periarticular or diffuse osteoporosis (Fig. 1) [19].
Anteroposterior radiograph of both hands in a 13.5-year-old boy with enthesitis-related (HLA B27 positive) juvenile idiopathic arthritis: general mild osteoporosis of the right hand (long arrow), soft tissue edema around the carpal bones on the right (short arrow), narrowed carpometacarpal joint spaces on the right (long thin arrow), discrepancy of the size of the carpal bones due to accelerated growth on the right (short thin arrow), suspicious erosions at the base of the second to fourth metacarpals and at os trapezoideum and os capitatum (arrowheads). Normal structure and morphology of the bones of the left hand
Musculoskeletal US is valuable in identifying extra-articular causes of tissue swelling mimicking joint effusion [26] and in determining the site of inflammation by differentiating between synovial, tendinous, and entheses involvement [27,28,29].
The role of MRI remains undisputed in ruling out other joint or synovial pathologies that may mimic JIA (pigmented villonodular synovitis, hemangioma, synovial chondromatosis, lipoma arborescens, or chronic recurrent multifocal osteomyelitis (Fig. 2) [30, 31].
Magnetic resonance images in a 15-year-old boy with chronic recurrent multifocal osteomyelitis. a, b Coronal T2 turbo inversion recovery magnitude (TIRM) (a) and T1 (b) images of the hips show bone edema in the metaphysis and epiphysis of the left femur and at the base of the right greater trochanter (long arrows). There is a small reactive effusion in the left hip joint (short arrow in a). c Coronal T2 TIRM image shows bone edema in the distal metaphysis of the left femur (arrow)
Assessment of disease activity/remission and treatment efficacy
Many studies on rheumatoid arthritis have shown that residual synovitis is associated with a significant risk of disease relapse and progression of structural damage. MRI is increasingly being incorporated as an endpoint in numerous clinical trials aimed at evaluating the effectiveness of novel antirheumatic drugs for adult patients [32,33,34,35].
In JIA, multiple studies have been performed that have aimed to link imaging results with the ability to evaluate disease activity and treatment efficacy.
Literature articles from 2011 to 2013 have shown that the presence of US-detected synovial abnormalities, including power Doppler signals, does not predict subsequent synovitis flare in JIA patients in clinical remission. Interestingly, this contrasts with the findings in adults. Paradoxically, patients with persistent inactive disease had a higher frequency of power Doppler signals than those who experienced a disease flare [14, 36,37,38].
Subsequently, with improved US technology, it was demonstrated that musculoskeletal US abnormalities were shown to increase the risk of disease relapse in clinically inactive JIA patients by almost fourfold. This highlights musculoskeletal US as a valuable tool for stratifying the risk of disease relapse in patients with JIA in clinical remission [39, 40].
A recent study by Mazzoni et al. [12] found that subclinical synovitis and bone marrow edema detected on MRI in 65.5% and 46.7% of patients in their cohort, respectively, were the best predictors of disease relapse and joint deterioration, despite clinical remission. These findings have important implications for disease management [12].
Due to the lack of standardized measurements and techniques, imaging is not currently part of the composite measures of disease activity, which focus on clinician- and patient-reported assessments and inflammatory markers. Nevertheless, imaging is often used in clinical practice to guide decisions, especially for joints that are difficult to assess clinically (TMJ, sacroiliac joint (SIJ), cervical spine) [20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42].
Which imaging modalities should be used in juvenile idiopathic arthritis from the clinical perspective?
Indications for conventional radiology
XR remains the most readily available imaging modality for detecting and monitoring structural damage and growth abnormalities, but given the current emphasis on early intervention, the detection of pre-erosive joint changes has become a priority.
However, in cases of clinical uncertainty, XR plays a crucial role, excluding other differential diagnostic options such as traumatic or orthopedic diseases (fractures, osteochondral lesions), tumors, and infectious causes (osteomyelitis) [4, 6].
In 2018, the Task Force of the French Societies of Rheumatology, Radiology, and Paediatric Rheumatology, focusing on XR, attempted for the first time to provide pragmatic guidelines for daily practice specific for each non-systemic JIA subtype and for situations of particular interest [43], summarized in Table 2.
Interestingly, Weiss et al. [44] recently provided the first consensus-derived radiographic definition of sacroiliitis in skeletally immature adolescents as a criterion for classifying axial disease in juvenile spondyloarthritis when MRI is not available. Nevertheless, the use of XR in the diagnosis of sacroiliitis is discouraged.
In recent decades, new radiological scoring systems have been developed and adult radiological scores have been adapted for use in JIA. Their application in non-controlled JIA clinical trials has demonstrated that standardized assessment of radiological progression is feasible. This has led to the suggestion that semiquantitative measurement of radiological damage should also be considered when evaluating treatment efficacy in JIA [45,46,47].
In advanced stages of disease, XR allows visualization of late complications (erosions, ankylosis, subluxation or joint malalignment, enlarged epiphysis, premature growth plate fusion leading to limb length inequality, spinal deformities, muscle atrophy) (Fig. 3) [48,49,50,51].
Anteroposterior radiographs of both hands in a 19-year-old young woman with a long history of aggressive seronegative polyarticular juvenile idiopathic arthritis, onset at the age of 7 years and with poor adherence to treatment. Images show mild bilateral periarticular osteoporosis (long arrows), growth arrest lines in the radial metaphysis and bone-in-bone appearance of carpal bones (short arrows), joint space narrowing of carpo-metacarpal joints and intercarpal bones, more pronounced on the right (arrowheads), and mild soft tissue edema around the right ulna (thin arrow)
In addition, XR has a historical role in assessing bone maturity and detecting bone age delay or progression, which in JIA may also help to distinguish where disease control is suboptimal or whether other factors are influencing growth retardation [52].
In summary, XR is a useful method for differential diagnosis in doubtful cases, evaluation of structural and morphological changes before diagnosis (indication based on clinical examination), and evaluation and monitoring of joint destruction and growth disorders.
Indications for musculoskeletal ultrasound
Musculoskeletal US represents an easily accessible, clinically relevant routine examination in children with JIA.
Before US can be established as a valuable imaging modality, two significant challenges must be overcome: understanding age-related normal findings and standardizing the US protocol for different joints.
Numerous papers, including the 2018 OMERACT US in Paediatrics Working Group, have described the standardization of US examination for different joints, including physiological intra-articular vascularization, patient and joint position, and transducer placement for each examination approach [53,54,55,56,57,58].
Although these reports provide information to help differentiate between normal and pathological findings of joints in children, they currently serve as baseline information. Assessment of changes in US characteristics within an individual subject over time could potentially be more informative than a simple comparison with a cutoff value [56]. Another issue is the minimum and optimal set of joints that should be scanned for routine musculoskeletal US surveillance. Scanning all accessible joints is not feasible in routine practice, and different studies have tested different numbers of joints.
The reduced model with ten joints by Collado et al. [59] showed higher responsiveness to changes than the evaluation of a larger number of joints. Overall, these results suggest that an US assessment which focuses on a reduced number of joints and includes the sites that are most commonly affected in JIA may satisfactorily provide information about the overall burden of disease activity [59].
Joints most suitable for musculoskeletal US examination are the ankle, knee, hip, wrist, and small joints of the hands and feet (Figs. 4 and 5) [18]. Apart from the anatomical consideration and the challenge of clinical assessment of these joints, Magni-Manzoni et al. [60] have demonstrated a higher incidence of subclinical synovitis in the wrists, proximal interphalangeal (PIP) joints, subtalar joints, and ankles.
Power Doppler ultrasound of left talonavicular joint in sagittal projection in a 19-year-old young woman with a long history of aggressive seronegative polyarticular juvenile idiopathic arthritis, onset at the age of 7 years and with poor adherence to treatment (same patient as in Fig. 3) shows thickened and chronically altered synovium without a joint effusion or significant hyperemia (arrow)
Ankle ultrasound of a 2-year-old girl with juvenile idiopathic arthritis (HLA B27 negative, anti-nuclear antibodies positive). a, b Sagittal projection of right (a) and left (b) ankle with effusion in the right side (arrow); (c) Sagittal power Doppler study of right ankle with effusion (long arrow), and thickened and hyperemic synovium (short arrow)—signs of synovitis
Although the role of US in assessing the axial skeleton remains limited, a recent study by Falsetti et al. [61] suggested the potential role of power Doppler ultrasound with spectral wave analysis as a screening method in children with suspected juvenile spondyloarthritis. They found higher power Doppler US scores at the SIJs in patients with a confirmed diagnosis of juvenile spondyloarthritis based on MRI diagnosis [61].
The most important clinical contribution of US is the identification and differentiation of synovitis, tenosynovitis, bursitis, and enthesitis.
In several studies, US has been shown to be superior to physical examination in the diagnosis of synovitis [26, 28, 37, 60, 62,63,64,65,66,67,68]. Nevertheless, it remains questionable whether physical examination or US can be more accurate in detecting joint inflammation. Indeed, confidence and competence in musculoskeletal examinations may be low, particularly in pediatrics. A systematic literature review on the assessment of synovitis in JIA by Collado et al. [69] on the assessment of synovitis in JIA highlighted key issues, such as small sample size, lack of MRI comparison, technical difficulties, and lack of a control score.
Enthesitis is the main feature of the JIA subgroup of enthesitis-related arthritis. Clinical recognition of enthesitis in children is challenging due to the particular distribution of fat, which can obscure anatomical landmarks, and the often-inadequate cooperation of very young children.
A number of studies confirm a higher sensitivity of musculoskeletal US compared to clinical examination in detecting enthesitis [70,71,72,73]. On the other hand, a recent systematic review indicated that the existing evidence suggests that there is no standardized US definition of enthesitis in children, and that discriminant validity has not been demonstrated [74].
The additional role of US is to monitor the response to treatment and the disease course. In this context, the importance of power Doppler US in addition to conventional US and the use of a standardized US scoring system seems crucial.
US-guided procedures are the next important application of US in JIA. They allow precise localization of inflammation and accurate needle placement in clinically difficult-to-assess or hard-to-reach sites, such as wrists, TMJs, hip, small joints of hands and feet, ankles, and tendons. This maximizes treatment efficacy while minimizing local side effects, such as subcutaneous atrophy or localized skin hypopigmentation [75,76,77,78,79]. Due to limited and conflicting data, certain critical aspects require further investigation, particularly for specific sites such as the TMJs [80].
In summary, musculoskeletal US is useful in daily practice to assess the presence and degree of inflammation in areas that are more difficult to assess clinically, such as the wrist, ankle, and foot joints. It can be used to guide intra-articular injections. Ideally, it should be performed by an experienced radiologist or rheumatologist.
Indications for magnetic resonance imaging
MRI is the most promising imaging technique for assessing the presence and extent of inflammation (synovial hypertrophy, joint effusion, soft tissue swelling), bone marrow changes, and cartilage status (Figs. 6 and 7). Furthermore, MRI can serve as a diagnostic tool for certain intra-articular pathologies that mimic JIA [81, 82].
Magnetic resonance imaging of the right ankle in a 2-year-old girl with juvenile idiopathic arthritis (HLA B27 negative, anti-nuclear antibodies positive, same patient as in Fig. 5). a Sagittal proton-density-weighted fat suppressed image shows marked synovial proliferation and effusion of the anterior and posterior recesses of the ankle joint (long arrows) and around the extensor tendons (short arrow)—signs of synovitis and tenosynovitis. b Sagittal postcontrast T1-weighted fat-suppressed sequence shows marked synovial proliferation and enhancement of the anterior recess of the ankle joint (long arrow) and around the tarsal bones (short arrow)—signs of active synovitis. c Axial postcontrast T1-weighted fat-suppressed sequence shows marked synovial proliferation and enhancement of the anterior and posterior recesses of the ankle joint (long arrows) and around the flexor tendons (short arrow)—signs of active synovitis and tenosynovitis
Magnetic resonance imaging of the right wrist in a 14-year-old boy with juvenile idiopathic arthritis with enthesitis (HLA B27 positive). Coronal (a) and axial (b) proton-density-weighted fat-suppressed images show marked synovial proliferation (long arrows), reactive edema of the carpal bones (short arrows), and a small joint effusion (arrowhead in b)—signs of synovitis
As with all imaging modalities, the main criticisms of using MRI include the challenge of differentiating between pathological and physiological changes in bone marrow depending on the age and sex of the patient [83,84,85,86,87,88,89,90,91]. There is also a need for standardization, quantification, and validation of scoring systems to rigorously and consistently assess joint changes, in both cross-sectional and longitudinal studies. It is worth noting that efforts are currently underway to achieve this standardization [92].
The importance of MRI for monitoring inflammation and response to treatment has been confirmed. In 2015, the EULAR-PRES task force [6] and the European Society of Musculoskeletal Radiology (ESSR) Arthritis Subcommittee [93] published the indications for performing MRI for diagnosis, monitoring, and prediction, as well as MRI protocols for the most commonly affected joints in JIA; these have recently been updated by the ESSR and the European Society of Paediatric Radiology musculoskeletal imaging taskforce [19].
Emerging issues in MRI surveillance of JIA patients relate to the presence of subclinical synovitis as a predictor of disease flare-up (Fig. 8) [12]. Bone marrow edema is a questionable predictor of an unfavorable outcome, as it can be found in more than 50% of healthy children, as shown by the Norwegian group [87].
Coronal magnetic resonance imaging of the hips in a 15-year-old girl with psoriatic arthritis (HLA B27 negative, ANA positive). a T2 turbo inversion recovery image shows an effusion in the left hip joint (arrow); bone and cartilage are normal in appearance. b Postcontrast T1-weighted fat-suppressed image shows synovial enhancement on the left (arrow)—a sign of active synovitis
MRI has demonstrated greater sensitivity than US and CR in detecting bone erosions, even in the early stages of the disease [94,95,96]. In contrast, there are conflicting data regarding the detection of cartilage erosions, probably due to the lack of cartilage-specific sequences in MRI protocols for JIA [36].
Specifically, standardized MRI protocols and semi-quantitative classification systems have been developed to assess inflammation and osteochondral changes in the large and small joints of JIA patients. These are currently undergoing validation, including assessing their correlation with clinical disease activity [97,98,99,100,101,102]. The ability to identify a “target joint” that reflects the global burden of disease activity may be an optimal target [46, 97].
MRI is highly valuable for difficult-to-access joints like the TMJ and the axial skeleton, which are commonly affected in JIA. Early detection is essential to prevent functional issues, including mandibular condyle growth inhibition and micrognathia (Fig. 9) [98, 102,103,104]. Recently, a consensus MRI protocol for the examination of the TMJ has been developed by Inarejos Clemente et al. [20], describing the degree of normal and pathological findings using the currently available MRI scoring systems developed for JIA.
Postcontrast T1-weighted fat-suppressed magnetic resonance images of the temporomandibular joints in a 14-year-old girl with oligoarticular juvenile idiopathic arthritis (ANA positive). a Axial image shows synovial thickening and enhancement on the right (arrow)—a sign of active synovitis. b Sagittal image shows a flattened mandibular head (long arrow) and synovial thickening and enhancement on the right (short arrow)—signs of chronic bone changes and active synovitis
Contrast-enhanced MRI is the preferred method for identifying cervical spine involvement, a significant prognostic factor for JIA progression [21, 22]. It has demonstrated higher sensitivity than clinical examination, although cervical arthritis is often clinically silent [23].
MRI is valuable for monitoring disease progression, response to treatment, and evaluation of late changes and complications, including atlantoaxial instability, dens deformity, joint ankylosis, and spinal cord compression [105].
MRI is also the method of choice for the assessment of the SIJ.
Nevertheless, the ASAS criteria, commonly used in adults to evaluate both active inflammatory and structural lesions, may present challenges applied to children [42].
The OMERACT expert working group together with OMERACT-JAMRIS-SIJ is developing and evaluating a preliminary pediatric consensus scoring system of SIJMRI. This system assesses inflammation and structural changes in the SIJ of children, including erosion, sclerosis, fat lesion, and ankylosis considering growing bone and active bone marrow [10].
In addition, pelvic MRI in juvenile ankylosing spondylitis is also valuable for assessing enthesopathy at the tendon and fascial attachment sites and for coxofemoral joint involvement, which is often associated with sacroiliitis [13].
Whole-body MRI is a promising tool for detecting and monitoring inflammation involving the peripheral joints, the axial joints, and the entheses in rheumatological diseases such as spondyloarthropathies [106,107,108]. On the other hand, there are no clear guidelines for the standardized detection, interpretation, and quantification of JIA on whole-body MRI. Moreover, MRI is still not widely available in clinical practice due to limitations of cost, access, and relatively long acquisition time, requiring sedation or general anesthesia in young children.
In an effort to reduce the use of gadolinium-based contrast agents, Barendregt et al. [109] conducted interesting research on the potential use of diffusion-weighted imaging as an alternative for the assessment of synovial inflammation.
In summary, MRI is an excellent method for monitoring disease activity in response to treatment, especially in difficult-to-access joints such as the axial skeleton (spine and SIJs) and TMJs. It is also very sensitive in detecting subclinical arthritis, the importance of which needs to be further assessed. The importance of bone marrow edema as a potential bad outcome predictor also needs to be further investigated.
Advantages and disadvantages
The advantages and disadvantages of imaging modalities in JIA are systematically presented in Table 3 (XR), Table 4 (US), and Table 5 (MRI).
Conclusion
This article provides a concise overview of the clinical utility of different imaging modalities in patients with JIA. Imaging plays an important role in confirming the diagnosis of JIA, assessing joint and bone involvement, and in tracking disease activity and treatment response. In addition, imaging can potentially predict poor prognosis by detecting subclinical inflammation or structural damage, even in cases of clinically inactive disease. Nonetheless, it is crucial to use these imaging modalities judiciously, either to confirm or complement findings from physical examination of the musculoskeletal system.
Data availability
The images in the manuscript are available from the authors upon reasonable request. No other datasets were generated for this manuscript.
References
Thierry S, Fautrel B, Lemelle I, Guillemin F (2014) Prevalence and incidence of juvenile idiopathic arthritis: a systematic review. Joint Bone Spine 81:112–117
Brewer EJ Jr, Bass J, Baum J et al (1977) Current proposed revision of JRA Criteria. JRA Criteria Subcommittee of the Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Section of The Arthritis Foundation. Arthritis Rheum 20:195–199
Carol HA, Chauvin NA, Weiss PF (2023) Imaging in pediatric spondyloarthritis. Curr Opin Rheumatol 35:226–234
Malattia C, Tzaribachev N, van den Berg JM, Magni-Manzoni S (2018) Juvenile idiopathic arthritis - the role of imaging from a rheumatologist’s perspective. Pediatr Radiol 48:785–791
Navallas M, Tolend M, Otobo TM et al (2023) Developing standards for MRI evaluation of joints in children with juvenile idiopathic arthritis utilizing the temporomandibular joint as a model. Jpn J Radiol (in press). https://doi.org/10.1007/s11604-023-01479-y
Colebatch-Bourn AN, Edwards CJ, Collado P et al (2015) EULAR-PReS points to consider for the use of imaging in the diagnosis and management of juvenile idiopathic arthritis in clinical practice. Ann Rheum Dis 74:1946–1957
Beaton DE, Maxwell LJ, Shea BJ et al (2019) Instrument selection using the OMERACT Filter 2.1: the OMERACT methodology. J Rheumatol 46:1028–1035
Nusman CM, de Horatio LT, Hemke R et al (2018) Imaging in juvenile idiopathic arthritis - international initiatives and ongoing work. Pediatr Radiol 48:828–834
Swami VG, Jaremko JL, Rumsey DG et al (2019) Diagnostic accuracy of MRI-based sacroiliitis scoring systems: a systematic review. AJR Am J Roentgenol 212:1112–1125
Otobo TM, Conaghan PG, Maksymowych WP et al (2019) Preliminary definitions for sacroiliac joint pathologies in the OMERACT juvenile idiopathic arthritis magnetic resonance imaging score (OMERACT JAMRIS-SIJ). J Rheumatol 46:1192–1197
Tanturri de Horatio L, Shelmerdine SC, d’Angelo P et al (2023) A novel magnetic resonance imaging scoring system for active and chronic changes in children and adolescents with juvenile idiopathic arthritis of the hip. Pediatr Radiol 53:426–437
Mazzoni M, Pistorio A, Magnaguagno F et al (2023) Predictive value of magnetic resonance imaging in patients with juvenile idiopathic arthritis in clinical remission. Arthritis Care Res 75:198–205
Porter-Young FM, Offiah AC, Broadley P et al (2018) Inter- and intra-observer reliability of contrast-enhanced magnetic resonance imaging parameters in children with suspected juvenile idiopathic arthritis of the hip. Pediatr Radiol 48:1891–1900
Magni-Manzoni S, Scirè CA, Ravelli A et al (2013) Ultrasound-detected synovial abnormalities are frequent in clinically inactive juvenile idiopathic arthritis, but do not predict a flare of synovitis. Ann Rheum Dis 72:223–228
Aletaha D, Neogi T, Silman AJ et al (2010) 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 62:2569–2581
Duer-Jensen A, Hørslev-Petersen K, Hetland ML et al (2011) Bone edema on magnetic resonance imaging is an independent predictor of rheumatoid arthritis development in patients with early undifferentiated arthritis. Arthrit Rheum 63:2192–2202
Ording Muller LS, Humphries P, Rosendahl K (2015) The joints in juvenile idiopathic arthritis. Insights Imaging 6:275–284
Magni-Manzoni S (2016) Ultrasound in juvenile idiopathic arthritis. Pediatric Rheumatol Online J 14:33
Hemke R, Herregods N, Jaremko JL et al (2020) Imaging assessment of children presenting with suspected or known juvenile idiopathic arthritis: ESSR-ESPR points to consider. Eur Radiol 30:5237–5249
Inarejos Clemente EJ, Tolend M, Navallas M et al (2023) MRI of the temporomandibular joint in children with juvenile idiopathic arthritis: protocol and findings. Pediatr Radiol 53:1498–1512
Hospach T, Maier J, Müller-Abt P et al (2014) Cervical spine involvement in patients with juvenile idiopathic arthritis - MRI follow-up study. Pediatr Rheumatol Online J 12:9
Oren B, Oren H, Osma E, Cevik N (1996) Juvenile rheumatoid arthritis: cervical spine involvement and MRI in early diagnosis. Turk J Pediatr 38:189–194
Hofer MF, Cimaz R (2013) Is cervical spine involvement in juvenile polyarthritis under-recognized? Rheumatology 52:221–222
Tafaghodi F, Aghighi Y, RokniYazdi H et al (2009) Predictive plain X-ray findings in distinguishing early stage acute lymphoblastic leukemia from juvenile idiopathic arthritis. Clin Rheumatol 28:1253–1258
Chlosta EM, Kuhns LR, Holt JF (1975) The “patellar ratio” in hemophilia and juvenile rheumatoid arthritis. Radiology 116:137–138
Filippou G, Cantarini L, Bertoldi I et al (2011) Ultrasonography vs. clinical examination in children with suspected arthritis. Does it make sense to use poliarticular ultrasonographic screening? Clin Exp Rheumatol 29:345–350
Ramos PC, Calvo C, Diaz-Delgado R (2011) The role of musculoskeletal ultrasound in children with pain in the upper limbs. Clin Exp Rheumatol 29:410
Pascoli L, Wright S, McAllister C, Rooney M (2010) Prospective evaluation of clinical and ultrasound findings in ankle disease in juvenile idiopathic arthritis: importance of ankle ultrasound. J Rheumatol 37:2409–2414
Brunner E, Ting T, Vega-Fernandez P (2020) Musculoskeletal ultrasound in children: current state and future directions. Eur J Rheumatol 7:S28–S37
Kirkhus E, Flatø B, Riise O et al (2011) Differences in MRI findings between subgroups of recent-onset childhood arthritis. Pediatr Radiol 41:432–440
Uhl M, Krauss M, Kern S et al (2001) The knee joint in early juvenile idiopathic arthritis. An ROC study for evaluating the diagnostic accuracy of contrast-enhanced MR imaging. Acta Radiol 42:6–9
Brown AK, Conaghan PG, Karim Z et al (2008) An explanation for the apparent dissociation between clinical remission and continued structural deterioration in rheumatoid arthritis. Arthritis Rheum 58:2958–2967
Nguyen H, Ruyssen-Witrand A, Gandjbakhch F et al (2014) Prevalence of ultrasound-detected residual synovitis and risk of relapse and structural progression in rheumatoid arthritis patients in clinical remission: a systematic review and meta-analysis. Rheumatology 53:2110–2118
Baker JF, Conaghan PG, Emery P et al (2016) Validity of early MRI structural damage end points and potential impact on clinical trial design in rheumatoid arthritis. Ann Rheum Dis 75:1114–1119
Østergaard M, Jacobsson LT, Schaufelberger C et al (2015) MRI assessment of early response to certolizumab pegol in rheumatoid arthritis: a randomised, double-blind, placebo-controlled phase IIIb study applying MRI at weeks 0, 1, 2, 4, 8 and 16. Ann Rheum Dis 74:1156–1163
Malattia C, Consolaro A, Pederzoli S et al (2013) MRI versus conventional measures of disease activity and structural damage in evaluating treatment efficacy in juvenile idiopathic arthritis. Ann Rheum Dis 72:363–368
Rebollo-Polo M, Koujok K, Weisser C et al (2011) Ultrasound findings on patients with juvenile idiopathic arthritis in clinical remission. Arthritis Care Res 63:1013–1019
Brown A, Hirsch R, Laor T et al (2012) Do patients with juvenile idiopathic arthritis in clinical remission have evidence of persistent inflammation on 3T magnetic resonance imaging? Arthritis Care Res 64:1846–1854
De Lucia O, Ravagnani V, Pregnolato F et al (2018) Baseline ultrasound examination as possible predictor of relapse in patients affected by juvenile idiopathic arthritis (JIA). Ann Rheum Dis 77:1426–1431
Miotto E, Silva VB, Mitraud SAV et al (2017) Patients with juvenile idiopathic arthritis in clinical remission with positive power Doppler signal in joint ultrasonography have an increased rate of clinical flare: a prospective study. Pediatr Rheumatol Online J 15:80
Adib N, Silman A, Thomson W (2005) Outcome following onset of juvenile idiopathic inflammatory arthritis: II. predictors of outcome in juvenile arthritis. Rheumatology 44:1002–1007
Herregods N, Dehoorne J, Van den Bosch F et al (2017) ASAS definition for sacroiliitis on MRI in SpA: applicable to children? Pediatr Rheumatol Online J 15:24
Marteau P, Adamsbaum C, Rossi-Semerano L et al (2018) Conventional radiography in juvenile idiopathic arthritis: joint recommendations from the French societies for rheumatology, radiology and paediatric rheumatology. Eur Radiol 28:3963–3976
Weiss PF, Brandon TG, Lambert RG et al (2023) Consensus-driven definition for unequivocal sacroiliitis on radiographs in juvenile spondyloarthritis. J Rheumatol 50:1173–1177
Bertamino M, Rossi F, Pistorio A et al (2010) Development and initial validation of a radiographic scoring system for the hip in juvenile idiopathic arthritis. J Rheumatol 37:432–439
van Rossum MA, Boers M, Zwinderman AH et al (2005) Development of a standardized method of assessment of radiographs and radiographic change in juvenile idiopathic arthritis: introduction of the Dijkstra composite score. Arthritis Rheum 52:2865–2872
Ravelli A (2008) The time has come to include assessment of radiographic progression in juvenile idiopathic arthritis clinical trials. J Rheumatol 35:553–557
Johnson K (2006) Imaging of juvenile idiopathic arthritis. Pediatr Radiol 36:743–758
Oen K, Reed M, Malleson PN et al (2003) Radiologic outcome and its relationship to functional disability in juvenile rheumatoid arthritis. J Rheumatol 30:832–840
Damasio MB, Malattia C, Martini A, Tomà P (2010) Synovial and inflammatory diseases in childhood: role of new imaging modalities in the assessment of patients with juvenile idiopathic arthritis. Pediatr Radiol 40:985–998
Ravelli A, Ioselian M, Norambuena X et al (2007) Adapted versions of the Sharp/van der Heijde score are reliable and valid for assessment of radiographic progression in juvenile idiopathic arthritis. Arthritis Rheum 56:3087–3095
Greulich WW, Pyle SI (1959) Radiographic atlas of skeletal development of the hand and wrist. Stanford University Press
Roth J, Jousse-Joulin S, Magni-Manzoni S et al (2015) Definitions for the sonographic features of joints in healthy children. Arthritis Care Res 67:136–142
Collado P, Windschall D, Vojinovic J et al (2018) Amendment of the OMERACT ultrasound definitions of joints’ features in healthy children when using the DOPPLER technique. Pediatr Rheumatol Online J 16:23
Windschall D, Collado P, Vojinovic J et al (2020) Age-related vascularization and ossification of joints in children: an international pilot study to test multiobserver ultrasound reliability. Arthritis Care Res 72:498–506
Lanni S (2021) The recent evolution of ultrasound in juvenile idiopathic arthritis. Clin Exp Rheumatol 39: 1413–1421
Collado P, Vojinovic J, Nieto JC et al (2016) Toward standardized musculoskeletal ultrasound in pediatric rheumatology: normal age-related ultrasound findings. Arthritis Care Res 68:348–356
Lanni S, Bovis F, Ravelli A et al (2016) Delineating the application of ultrasound in detecting synovial abnormalities of the subtalar joint in juvenile idiopathic arthritis. Arthritis Care Res 68:1346–1353
Collado P, Naredo E, Calvo C et al (2013) Reduced joint assessment vs comprehensive assessment for ultrasound detection of synovitis in juvenile idiopathic arthritis. Rheumatology 52:1477–1484
Magni-Manzoni S, Epis O, Ravelli A et al (2009) Comparison of clinical versus ultrasound-determined synovitis in juvenile idiopathic arthritis. Arthritis Rheum 61:1497–1504
Falsetti P, Conticini E, Gaggiano C et al (2022) Doppler and spectral ultrasound of sacroiliac joints in pediatric patients with suspected juvenile spondyloarthritis. Diagnostics 12:992
Rooney ME, McAllister C, Burns JF (2009) Ankle disease in juvenile idiopathic arthritis: ultrasound findings in clinically swollen ankles. J Rheumatol 36:1725–1729
Algergawy S, Haliem T, Al-Shaer O (2011) Clinical, laboratory, and ultrasound assessment of the knee in juvenile rheumatoid arthritis. Clin Med Insights Arthritis Musculoskelet Disord 4:21–27
Janow GL, Panghaal V, Trinh A et al (2011) Detection of active disease in juvenile idiopathic arthritis: sensitivity and specificity of the physical examination vs ultrasound. J Rheumatol 38:2671–2674
Collado P, Gamir ML, López-Robledillo JC et al (2014) Detection of synovitis by ultrasonography in clinically inactive juvenile idiopathic arthritis on and off medication. Clin Exp Rheumatol 32:597–603
Breton S, Jousse-Joulin S, Cangemi C et al (2011) Comparison of clinical and ultrasonographic evaluations for peripheral synovitis in juvenile idiopathic arthritis. Semin Arthritis Rheum 41:272–278
Bugni Miotto e Silva V, de Freitas Tavares da Silva C, de Aguiar Vilela Mitraud S et al (2014) Do patients with juvenile idiopathic arthritis in remission exhibit active synovitis on joint ultrasound? Rheumatol Int 34:937–945
Haslam KE, McCann LJ, Wyatt S, Wakefield RJ (2010) The detection of subclinical synovitis by ultrasound in oligoarticular juvenile idiopathic arthritis: a pilot study. Rheumatology 49:123–127
Collado P, Jousse-Joulin S, Alcalde M et al (2012) Is ultrasound a validated imaging tool for the diagnosis and management of synovitis in juvenile idiopathic arthritis? A systematic literature review. Arthritis Care Res 64:1011–1019
Jousse-Joulin S, Breton S, Cangemi C et al (2011) Ultrasonography for detecting enthesitis in juvenile idiopathic arthritis. Arthritis Care Res 63:849–855
Weiss PF, Chauvin NA, Klink AJ et al (2014) Detection of enthesitis in children with enthesitis-related arthritis: dolorimetry compared to ultrasonography. Arthritis Rheumatol 66:218–227
Shenoy S, Aggarwal A (2016) Sonologic enthesitis in children with enthesitis-related arthritis. Clin Exp Rheumatol 34:143–147
Rossi-Semerano L, Ravagnani V, Collado P et al (2023) Validity of ultrasonography in detecting enthesitis in children: a systematic literature review. Joint Bone Spine 90:105538
Sande NK, Lilleby V, Aga AB et al (2023) Associations between power Doppler ultrasound findings and B-mode synovitis and clinical arthritis in juvenile idiopathic arthritis using a standardised scanning approach and scoring system. RMD Open 9:e002937
Young CM, Shiels WE, Coley BD et al (2012) Ultrasound-guided corticosteroid injection therapy for juvenile idiopathic arthritis: 12-year care experience. Pediatr Radiol 42:1481–1489
Tynjälä P, Honkanen V, Lahdenne P (2004) Intra-articular steroids in radiologically confirmed tarsal and hip synovitis of juvenile idiopathic arthritis. Clin Exp Rheumatol 22:643–648
Laurell L, Court-Payen M, Nielsen S et al (2011). Ultrasonography and color Doppler in juvenile idiopathic arthritis: diagnosis and follow-up of ultrasound-guided steroid injection in the ankle region. A descriptive interventional study. Pediatr Rheumatol Online J 9:4
Habibi S, Ellis J, Strike H, Ramanan AV (2012) Safety and efficacy of US-guided CS injection into temporomandibular joints in children with active JIA. Rheumatology 51:874–877
Parra DA, Chan M, Krishnamurthy G et al (2010) Use and accuracy of US guidance for image-guided injections of the temporomandibular joints in children with arthritis. Pediatr Radiol 40:1498–1504
Fritz J, Pereira PL, Lewin JS (2010) Temporomandibular joint injections: interventional MR imaging demonstrates anatomical landmark approach to be inaccurate when compared to direct visualization of the injectant. Pediatr Radiol 40:1964–1967
Johnson K, Wittkop B, Haigh F et al (2002) The early magnetic resonance imaging features of the knee in juvenile idiopathic arthritis. Clin Radiol 57:466–471
Kirkhus E, Flatø B, Riise O et al (2011) Differences in MRI findings between subgroups of recent-onset childhood arthritis. Pediatr Radiol 41:432–440
Laor T, Jaramillo D (2009) MR imaging insights into skeletal maturation: what is normal? Radiology 250:28–38
Vallejo JM, Jaramillo D (2001) Normal MR imaging anatomy of the ankle and foot in the pediatric population. Magn Reson Imaging Clin N Am 9:435–446
Moore SG, Dawson KL (1990) Red and yellow marrow in the femur: age-related changes in appearance at MR imaging. Radiology 175:219–223
Shabshin N, Schweitzer ME, Morrison WB et al (2006) High-signal T2 changes of the bone marrow of the foot and ankle in children: red marrow or traumatic changes? Pediatr Radiol 36:670–676
Müller LS, Avenarius D, Damasio B et al (2011) The paediatric wrist revisited: redefining MR findings in healthy children. Ann Rheum Dis 70:605–610
Chauvin NA, Xiao R, Brandon TG et al (2019) MRI of the sacroiliac joint in healthy children. AJR Am J Roentgenol 212:1303–1309
Vleeming A, Schuenke MD, Masi AT et al (2012) The sacroiliac joint: an overview of its anatomy, function and potential clinical implications. J Anat 221:537–567
Ording Muller LS, Boavida P, Avenarius D et al (2013) MRI of the wrist in juvenile idiopathic arthritis: erosions or normal variants? A prospective case-control study. Pediatr Radiol 43:785–795
Navallas M, Ares J, Beltrán B et al (2013) Sacroiliitis associated with axial spondyloarthropathy: new concepts and latest trends. Radiographics 33:933–956
Malattia C, Tolend M, Mazzoni M et al (2020) Current status of MR imaging of juvenile idiopathic arthritis. Best Pract Res Clin Rheumatol 34:101629
Sudoł-Szopińska I, Jurik AG, Eshed I et al (2015) Recommendations of the ESSR arthritis subcommittee for the use of magnetic resonance imaging in musculoskeletal rheumatic diseases. Semin Musculoskelet Radiol 19:396–411
Boavida P, Hargunani R, Owens CM, Rosendahl K (2012) Magnetic resonance imaging and radiographic assessment of carpal depressions in children with juvenile idiopathic arthritis: normal variants or erosions? J Rheumatol 39:645–650
Eich GF, Hallé F, Hodler J et al (1994) Juvenile chronic arthritis: imaging of the knees and hips before and after intraarticular steroid injection. Pediatr Radiol 24:558–563
Malattia C, Damasio MB, Magnaguagno F et al (2008) Magnetic resonance imaging, ultrasonography, and conventional radiography in the assessment of bone erosions in juvenile idiopathic arthritis. Arthritis Rheum 59:1764–1772
Nusman CM, Rosendahl K, Maas M (2016) MRI protocol for the assessment of juvenile idiopathic arthritis of the wrist: recommendations from the OMERACT MRI in JIA Working Group and Health-e-Child. J Rheumatol 43:1257–1258
Miller E, Inarejos Clemente EJ, Tzaribachev N et al (2018) Imaging of temporomandibular joint abnormalities in juvenile idiopathic arthritis with a focus on developing a magnetic resonance imaging protocol. Pediatr Radiol 48:792–800
Malattia C, Damasio MB, Pistorio A et al (2011) Development and preliminary validation of a paediatric-targeted MRI scoring system for the assessment of disease activity and damage in juvenile idiopathic arthritis. Ann Rheum Dis 70:440–446
Vaid YN, Dunnavant FD, Royal SA et al (2014) Imaging of the temporomandibular joint in juvenile idiopathic arthritis. Arthritis Care Res 66:47–54
Kellenberger CJ, Abramowicz S, Arvidsson LZ et al (2018) Recommendations for a standard magnetic resonance imaging protocol of temporomandibular joints in juvenile idiopathic arthritis. J Oral Maxillofac Surg 76:2463–2465
Tolend MA, Twilt M, Cron RQ et al (2018) Toward establishing a standardized magnetic resonance imaging scoring system for temporomandibular joints in juvenile idiopathic arthritis. Arthritis Care Res 70:758–767
Piancino MG, Cannavale R, Dalmasso P et al (2018) Cranial structure and condylar asymmetry of patients with juvenile idiopathic arthritis: a risky growth pattern. Clin Rheumatol 37:2667–2673
Müller L, Kellenberger CJ, Cannizzaro E et al (2009) Early diagnosis of temporomandibular joint involvement in juvenile idiopathic arthritis: a pilot study comparing clinical examination and ultrasound to magnetic resonance imaging. Rheumatology 48:680–685
Ključevšek D, Emeršič N, Toplak N, Avčin T (2017) Clinical and MRI outcome of cervical spine lesions in children with juvenile idiopathic arthritis treated with anti-TNFα drugs early in disease course. Pediatr Rheumatol Online J 15:38
Panwar J, Patel H, Tolend M et al (2021) Toward developing a semiquantitative whole body-MRI scoring for juvenile idiopathic arthritis: critical appraisal of the state of the art, challenges, and opportunities. Acad Radiol 28:271–286
Aggarwal A, Misra DP (2015) Enthesitis-related arthritis. Clin Rheumatol 34:1839–1846
Aquino MR, Tse SM, Gupta S et al (2015) Whole-body MRI of juvenile spondyloarthritis: protocols and pictorial review of characteristic patterns. Pediatr Radiol 45:754–762
Barendregt AM, van Gulik EC, Lavini C et al (2017) Diffusion-weighted imaging for assessment of synovial inflammation in juvenile idiopathic arthritis: a promising imaging biomarker as an alternative to gadolinium-based contrast agents. Eur Radiol 27:4889–4899
Funding
This work was partially supported by the Slovenian Research Agency [grant number J3-3061] and University Medical Centre Ljubljana [grant numbers 20210069 and 20220090].
Author information
Authors and Affiliations
Contributions
M. T.: conceptualization, data curation, analysis, and drafting of the initial manuscript; M. Z. A.: analysis, review and editing; A. G.: interpreting and describing the imaging findings, review and editing; D. K.: conceptualization, review and editing; T. A.: supervision, conceptualization, funding acquisition, review and editing. All authors critically reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Consent for publication
The images in the manuscript do not contain any identifiable information, and according to the Hospital Ethics Committee guidelines, there was no need to obtain individual patient consent to publish.
Conflicts of interest
None
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tarsia, M., Zajc Avramovič, M., Gazikalović, A. et al. A clinical perspective on imaging in juvenile idiopathic arthritis. Pediatr Radiol 54, 490–504 (2024). https://doi.org/10.1007/s00247-023-05815-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-023-05815-2