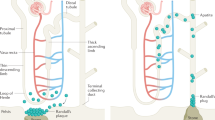
Abstract
This concise review summarizes our current understanding and the recent developments in genetics and related renal tubular disorders that have been linked with, or have been shown to be causal in, renal stone disease. The aim is to provide a readily accessible quick and easy update for urologists, nephrologists and endocrine or metabolic physicians whose practice involves the diagnosis and management of nephrolithiasis. An important message is to always consider a seemingly rare, and usually genetic, cause of kidney stones, since some of these are emerging as more common than originally thought, especially in adult clinical practice in which a family history of stones is a common finding.
Similar content being viewed by others
References
Scales CD Jr, Smith AC, Hanley JM, Saigal CS (2012) Prevalence of kidney stones in the United States. Eur Urol 62(1):160–165. doi:10.1016/j.eururo.2012.03.052
Gee HY, Jun I, Braun DA, Lawson JA, Halbritter J, Shril S, Nelson CP, Tan W, Stein D, Wassner AJ, Ferguson MA, Gucev Z, Sayer JA, Milosevic D, Baum M, Tasic V, Lee MG, Hildebrandt F (2016) Mutations in SLC26A1 cause nephrolithiasis. Am J Hum Genet 98(6):1228–1234. doi:10.1016/j.ajhg.2016.03.026
Halbritter J, Baum M, Hynes AM, Rice SJ, Thwaites DT, Gucev ZS, Fisher B, Spaneas L, Porath JD, Braun DA, Wassner AJ, Nelson CP, Tasic V, Sayer JA, Hildebrandt F (2015) Fourteen monogenic genes account for 15% of nephrolithiasis/nephrocalcinosis. J Am Soc Nephrol 26(3):543–551. doi:10.1681/ASN.2014040388
Claes DJ, Jackson E (2012) Cystinuria: mechanisms and management. Pediatr Nephrol 27(11):2031–2038. doi:10.1007/s00467-011-2092-6
Saravakos P, Kokkinou V, Giannatos E (2014) Cystinuria: current diagnosis and management. Urology 83(4):693–699. doi:10.1016/j.urology.2013.10.013
Dello Strologo L, Rizzoni G (2006) Cystinuria. Acta Paediatr Suppl 95(452):31–33. doi:10.1080/08035320600649473
Cassidy MJ, McCulloch T, Fairbanks LD, Simmonds HA (2004) Diagnosis of adenine phosphoribosyltransferase deficiency as the underlying cause of renal failure in a renal transplant recipient. Nephrol Dial Transplant 19(3):736–738
Edvardsson V, Palsson R, Olafsson I, Hjaltadottir G, Laxdal T (2001) Clinical features and genotype of adenine phosphoribosyltransferase deficiency in iceland. Am J Kidney Dis 38(3):473–480
Mohebbi NWCA (2015) Inherited forms of distal renal tubular acidosis. Der Nephrologe. doi:10.1007/s11560-014-0946-1
Stehberger PA, Schulz N, Finberg KE, Karet FE, Giebisch G, Lifton RP, Geibel JP, Wagner CA (2003) Localization and regulation of the ATP6V0A4 (a4) vacuolar H+-ATPase subunit defective in an inherited form of distal renal tubular acidosis. J Am Soc Nephrol 14(12):3027–3038
Dhayat NA, Schaller A, Albano G, Poindexter J, Griffith C, Pasch A, Gallati S, Vogt B, Moe OW, Fuster DG (2016) The vacuolar H+-ATPase B1 subunit polymorphism p. E161K associates with impaired urinary acidification in recurrent stone formers. J Am Soc Nephrol 27(5):1544–1554. doi:10.1681/ASN.2015040367
Mohebbi N, Vargas-Poussou R, Hegemann SC, Schuknecht B, Kistler AD, Wuthrich RP, Wagner CA (2013) Homozygous and compound heterozygous mutations in the ATP6V1B1 gene in patients with renal tubular acidosis and sensorineural hearing loss. Clin Genet 83(3):274–278. doi:10.1111/j.1399-0004.2012.01891.x
Hennings JC, Picard N, Huebner AK, Stauber T, Maier H, Brown D, Jentsch TJ, Vargas-Poussou R, Eladari D, Hubner CA (2012) A mouse model for distal renal tubular acidosis reveals a previously unrecognized role of the V-ATPase a4 subunit in the proximal tubule. EMBO Mol Med 4(10):1057–1071. doi:10.1002/emmm.201201527
Karet FE, Finberg KE, Nelson RD, Nayir A, Mocan H, Sanjad SA, Rodriguez-Soriano J, Santos F, Cremers CW, Di Pietro A, Hoffbrand BI, Winiarski J, Bakkaloglu A, Ozen S, Dusunsel R, Goodyer P, Hulton SA, Wu DK, Skvorak AB, Morton CC, Cunningham MJ, Jha V, Lifton RP (1999) Mutations in the gene encoding B1 subunit of H+-ATPase cause renal tubular acidosis with sensorineural deafness. Nat Genet 21(1):84–90. doi:10.1038/5022
Wagner CA, Devuyst O, Bourgeois S, Mohebbi N (2009) Regulated acid-base transport in the collecting duct. Pflugers Arch 458(1):137–156. doi:10.1007/s00424-009-0657-z
Khositseth S, Sirikanaerat A, Khoprasert S, Opastirakul S, Kingwatanakul P, Thongnoppakhun W, Yenchitsomanus PT (2008) Hematological abnormalities in patients with distal renal tubular acidosis and hemoglobinopathies. Am J Hematol 83(6):465–471. doi:10.1002/ajh.21151
Batlle D, Haque SK (2012) Genetic causes and mechanisms of distal renal tubular acidosis. Nephrol Dial Transplant 27(10):3691–3704. doi:10.1093/ndt/gfs442
Hu PY, Roth DE, Skaggs LA, Venta PJ, Tashian RE, Guibaud P, Sly WS (1992) A splice junction mutation in intron 2 of the carbonic anhydrase II gene of osteopetrosis patients from Arabic countries. Hum Mutat 1(4):288–292. doi:10.1002/humu.1380010404
Ismail EA, Abul Saad S, Sabry MA (1997) Nephrocalcinosis and urolithiasis in carbonic anhydrase II deficiency syndrome. Eur J Pediatr 156(12):957–962
Haque SK, Ariceta G, Batlle D (2012) Proximal renal tubular acidosis: a not so rare disorder of multiple etiologies. Nephrol Dial Transplant 27(12):4273–4287. doi:10.1093/ndt/gfs493
Biber J, Murer H, Mohebbi N, Wagner CA (2014) Renal handling of phosphate and sulfate. Compr Physiol 4(2):771–792. doi:10.1002/cphy.c120031
Bergwitz C, Roslin NM, Tieder M, Loredo-Osti JC, Bastepe M, Abu-Zahra H, Frappier D, Burkett K, Carpenter TO, Anderson D, Garabedian M, Sermet I, Fujiwara TM, Morgan K, Tenenhouse HS, Juppner H (2006) SLC34A3 mutations in patients with hereditary hypophosphatemic rickets with hypercalciuria predict a key role for the sodium-phosphate cotransporter NaPi-IIc in maintaining phosphate homeostasis. Am J Hum Genet 78(2):179–192. doi:10.1086/499409
Lorenz-Depiereux B, Benet-Pages A, Eckstein G, Tenenbaum-Rakover Y, Wagenstaller J, Tiosano D, Gershoni-Baruch R, Albers N, Lichtner P, Schnabel D, Hochberg Z, Strom TM (2006) Hereditary hypophosphatemic rickets with hypercalciuria is caused by mutations in the sodium-phosphate cotransporter gene SLC34A3. Am J Hum Genet 78(2):193–201. doi:10.1086/499410
Dasgupta D, Wee MJ, Reyes M, Li Y, Simm PJ, Sharma A, Schlingmann KP, Janner M, Biggin A, Lazier J, Gessner M, Chrysis D, Tuchman S, Baluarte HJ, Levine MA, Tiosano D, Insogna K, Hanley DA, Carpenter TO, Ichikawa S, Hoppe B, Konrad M, Savendahl L, Munns CF, Lee H, Juppner H, Bergwitz C (2014) Mutations in SLC34A3/NPT2c are associated with kidney stones and nephrocalcinosis. J Am Soc Nephrol 25(10):2366–2375. doi:10.1681/ASN.2013101085
Arcidiacono T, Mingione A, Macrina L, Pivari F, Soldati L, Vezzoli G (2014) Idiopathic calcium nephrolithiasis: a review of pathogenic mechanisms in the light of genetic studies. Am J Nephrol 40(6):499–506. doi:10.1159/000369833
Prie D, Huart V, Bakouh N, Planelles G, Dellis O, Gerard B, Hulin P, Benque-Blanchet F, Silve C, Grandchamp B, Friedlander G (2002) Nephrolithiasis and osteoporosis associated with hypophosphatemia caused by mutations in the type 2a sodium-phosphate cotransporter. N Engl J Med 347(13):983–991. doi:10.1056/NEJMoa020028
Wagner CA, Rubio-Aliaga I, Biber J, Hernando N (2014) Genetic diseases of renal phosphate handling. Nephrol Dial Transplant 29(Suppl 4):iv45–54. doi:10.1093/ndt/gfu217
Magen D, Berger L, Coady MJ, Ilivitzki A, Militianu D, Tieder M, Selig S, Lapointe JY, Zelikovic I, Skorecki K (2010) A loss-of-function mutation in NaPi-IIa and renal Fanconi’s syndrome. N Engl J Med 362(12):1102–1109. doi:10.1056/NEJMoa0905647
Virkki LV, Forster IC, Hernando N, Biber J, Murer H (2003) Functional characterization of two naturally occurring mutations in the human sodium-phosphate cotransporter type IIa. J Bone Miner Res 18(12):2135–2141. doi:10.1359/jbmr.2003.18.12.2135
Lapointe JY, Tessier J, Paquette Y, Wallendorff B, Coady MJ, Pichette V, Bonnardeaux A (2006) NPT2a gene variation in calcium nephrolithiasis with renal phosphate leak. Kidney Int 69(12):2261–2267. doi:10.1038/sj.ki.5000437
Dinour D, Davidovits M, Ganon L, Ruminska J, Forster IC, Hernando N, Eyal E, Holtzman EJ, Wagner CA (2016) Loss of function of NaPiIIa causes nephrocalcinosis and possibly kidney insufficiency. Pediatr Nephrol. doi:10.1007/s00467-016-3443-0
Schlingmann KP, Kaufmann M, Weber S, Irwin A, Goos C, John U, Misselwitz J, Klaus G, Kuwertz-Broking E, Fehrenbach H, Wingen AM, Guran T, Hoenderop JG, Bindels RJ, Prosser DE, Jones G, Konrad M (2011) Mutations in CYP24A1 and idiopathic infantile hypercalcemia. N Engl J Med 365(5):410–421. doi:10.1056/NEJMoa1103864
Colussi G, Ganon L, Penco S, De Ferrari ME, Ravera F, Querques M, Primignani P, Holtzman EJ, Dinour D (2014) Chronic hypercalcaemia from inactivating mutations of vitamin D 24-hydroxylase (CYP24A1): implications for mineral metabolism changes in chronic renal failure. Nephrol Dial Transplant 29(3):636–643. doi:10.1093/ndt/gft460
Ferraro PM, Minucci A, Primiano A, De Paolis E, Gervasoni J, Persichilli S, Naticchia A, Capoluongo E, Gambaro G (2016) A novel CYP24A1 genotype associated to a clinical picture of hypercalcemia, nephrolithiasis and low bone mass. Urolithiasis. doi:10.1007/s00240-016-0923-4
Gigante M, Santangelo L, Diella S, Caridi G, Argentiero L, D’’Alessandro MM, Martino M, Stea ED, Ardissino G, Carbone V, Pepe S, Scrutinio D, Maringhini S, Ghiggeri GM, Grandaliano G, Giordano M, Gesualdo L (2016) Mutational spectrum of CYP24A1 gene in a cohort of italian patients with idiopathic infantile hypercalcemia. Nephron 133(3):193–204. doi:10.1159/000446663
Nesterova G, Malicdan MC, Yasuda K, Sakaki T, Vilboux T, Ciccone C, Horst R, Huang Y, Golas G, Introne W, Huizing M, Adams D, Boerkoel CF, Collins MT, Gahl WA (2013) 1,25-(OH)2D-24 Hydroxylase (CYP24A1) deficiency as a cause of nephrolithiasis. Clin J Am Soc Nephrol 8(4):649–657. doi:10.2215/CJN.05360512
Vargas-Poussou R, Mansour-Hendili L, Baron S, Bertocchio JP, Travers C, Simian C, Treard C, Baudouin V, Beltran S, Broux F, Camard O, Cloarec S, Cormier C, Debussche X, Dubosclard E, Eid C, Haymann JP, Kiando SR, Kuhn JM, Lefort G, Linglart A, Lucas-Pouliquen B, Macher MA, Maruani G, Ouzounian S, Polak M, Requeda E, Robier D, Silve C, Souberbielle JC, Tack I, Vezzosi D, Jeunemaitre X, Houillier P (2016) Familial Hypocalciuric hypercalcemia types 1 and 3 and primary hyperparathyroidism: similarities and differences. J Clin Endocrinol Metab 101(5):2185–2195. doi:10.1210/jc.2015-3442
Stratta P, Merlotti G, Musetti C, Quaglia M, Pagani A, Izzo C, Radin E, Airoldi A, Baorda F, Palladino T, Leone MP, Guarnieri V (2014) Calcium-sensing-related gene mutations in hypercalcaemic hypocalciuric patients as differential diagnosis from primary hyperparathyroidism: detection of two novel inactivating mutations in an Italian population. Nephrol Dial Transplant 29(10):1902–1909. doi:10.1093/ndt/gfu065
Pearce SH, Williamson C, Kifor O, Bai M, Coulthard MG, Davies M, Lewis-Barned N, McCredie D, Powell H, Kendall-Taylor P, Brown EM, Thakker RV (1996) A familial syndrome of hypocalcemia with hypercalciuria due to mutations in the calcium-sensing receptor. N Engl J Med 335(15):1115–1122. doi:10.1056/NEJM199610103351505
Oddsson A, Sulem P, Helgason H, Edvardsson VO, Thorleifsson G, Sveinbjornsson G, Haraldsdottir E, Eyjolfsson GI, Sigurdardottir O, Olafsson I, Masson G, Holm H, Gudbjartsson DF, Thorsteinsdottir U, Indridason OS, Palsson R, Stefansson K (2015) Common and rare variants associated with kidney stones and biochemical traits. Nat Commun 6:7975. doi:10.1038/ncomms8975
Vezzoli G, Terranegra A, Aloia A, Arcidiacono T, Milanesi L, Mosca E, Mingione A, Spotti D, Cusi D, Hou J, Hendy GN, Soldati L, Paloschi V, Dogliotti E, Brasacchio C, Dell’Antonio G, Montorsi F, Bertini R, Bellinzoni P, Guazzoni G, Borghi L, Guerra A, Allegri F, Ticinesi A, Meschi T, Nouvenne A, Lupo A, Fabris A, Gambaro G, Strazzullo P, Rendina D, De Filippo G, Brandi ML, Croppi E, Cianferotti L, Trinchieri A, Caudarella R, Cupisti A, Anglani F, Del Prete D (2013) Decreased transcriptional activity of calcium-sensing receptor gene promoter 1 is associated with calcium nephrolithiasis. J Clin Endocrinol Metab 98(9):3839–3847. doi:10.1210/jc.2013-1834
Vezzoli G, Terranegra A, Rainone F, Arcidiacono T, Cozzolino M, Aloia A, Dogliotti E, Cusi D, Soldati L (2011) Calcium-sensing receptor and calcium kidney stones. J Transl Med 9:201. doi:10.1186/1479-5876-9-201
Windpessl M, Ritelli M, Wallner M, Colombi M (2016) A novel homozygous SLC2A9 mutation associated with renal-induced hypouricemia. Am J Nephrol 43(4):245–250. doi:10.1159/000445845
Mou LJ, Jiang LP, Hu Y (2015) A novel homozygous GLUT9 mutation cause recurrent exercise-induced acute renal failure and posterior reversible encephalopathy syndrome. J Nephrol 28(3):387–392. doi:10.1007/s40620-014-0073-0
Nishida H, Kaida H, Ishibashi M, Baba K, Kouno K, Okuda S (2005) Evaluation of exercise-induced acute renal failure in renal hypouricemia using Tc-99m DTPA renography. Ann Nucl Med 19(4):325–329
Bhasin B, Stiburkova B, De Castro-Pretelt M, Beck N, Bodurtha JN, Atta MG (2014) Hereditary renal hypouricemia: a new role for allopurinol? Am J Med 127(1):e3–e4. doi:10.1016/j.amjmed.2013.08.025
Lloyd SE, Pearce SH, Fisher SE, Steinmeyer K, Schwappach B, Scheinman SJ, Harding B, Bolino A, Devoto M, Goodyer P, Rigden SP, Wrong O, Jentsch TJ, Craig IW, Thakker RV (1996) A common molecular basis for three inherited kidney stone diseases. Nature 379(6564):445–449. doi:10.1038/379445a0
Devuyst O, Christie PT, Courtoy PJ, Beauwens R, Thakker RV (1999) Intra-renal and subcellular distribution of the human chloride channel, CLC-5, reveals a pathophysiological basis for Dent’s disease. Hum Mol Genet 8(2):247–257
Copelovitch L, Nash MA, Kaplan BS (2007) Hypothesis: dent disease is an underrecognized cause of focal glomerulosclerosis. Clin J Am Soc Nephrol 2(5):914–918. doi:10.2215/CJN.00900207
Wrong OM, Norden AG, Feest TG (1994) Dent’s disease; a familial proximal renal tubular syndrome with low-molecular-weight proteinuria, hypercalciuria, nephrocalcinosis, metabolic bone disease, progressive renal failure and a marked male predominance. QJM 87(8):473–493
De Leo MG, Staiano L, Vicinanza M, Luciani A, Carissimo A, Mutarelli M, Di Campli A, Polishchuk E, Di Tullio G, Morra V, Levtchenko E, Oltrabella F, Starborg T, Santoro M, di Bernardo D, Devuyst O, Lowe M, Medina DL, Ballabio A, De Matteis MA (2016) Autophagosome-lysosome fusion triggers a lysosomal response mediated by TLR9 and controlled by OCRL. Nat Cell Biol 18(8):839–850. doi:10.1038/ncb3386
Blanchard A, Vargas-Poussou R, Peyrard S, Mogenet A, Baudouin V, Boudailliez B, Charbit M, Deschesnes G, Ezzhair N, Loirat C, Macher MA, Niaudet P, Azizi M (2008) Effect of hydrochlorothiazide on urinary calcium excretion in dent disease: an uncontrolled trial. Am J Kidney Dis 52(6):1084–1095. doi:10.1053/j.ajkd.2008.08.021
Cebotaru V, Kaul S, Devuyst O, Cai H, Racusen L, Guggino WB, Guggino SE (2005) High citrate diet delays progression of renal insufficiency in the ClC-5 knockout mouse model of Dent’s disease. Kidney Int 68(2):642–652. doi:10.1111/j.1523-1755.2005.00442.x
Yu AS (2015) Claudins and the kidney. J Am Soc Nephrol 26(1):11–19. doi:10.1681/ASN.2014030284
Thorleifsson G, Holm H, Edvardsson V, Walters GB, Styrkarsdottir U, Gudbjartsson DF, Sulem P, Halldorsson BV, de Vegt F, d’Ancona FC, den Heijer M, Franzson L, Christiansen C, Alexandersen P, Rafnar T, Kristjansson K, Sigurdsson G, Kiemeney LA, Bodvarsson M, Indridason OS, Palsson R, Kong A, Thorsteinsdottir U, Stefansson K (2009) Sequence variants in the CLDN14 gene associate with kidney stones and bone mineral density. Nat Genet 41(8):926–930. doi:10.1038/ng.404
Toka HR, Genovese G, Mount DB, Pollak MR, Curhan GC (2013) Frequency of rare allelic variation in candidate genes among individuals with low and high urinary calcium excretion. PLoS One 8(8):e71885. doi:10.1371/journal.pone.0071885
Weber S, Schneider L, Peters M, Misselwitz J, Ronnefarth G, Boswald M, Bonzel KE, Seeman T, Sulakova T, Kuwertz-Broking E, Gregoric A, Palcoux JB, Tasic V, Manz F, Scharer K, Seyberth HW, Konrad M (2001) Novel paracellin-1 mutations in 25 families with familial hypomagnesemia with hypercalciuria and nephrocalcinosis. J Am Soc Nephrol 12(9):1872–1881
Yamaguti PM, Neves FA, Hotton D, Bardet C, de La Dure-Molla M, Castro LC, Scher MD, Barbosa ME, Ditsch C, Fricain JC, de La Faille R, Figueres ML, Vargas-Poussou R, Houiller P, Chaussain C, Babajko S, Berdal A, Acevedo AC (2016) Amelogenesis imperfecta in familial hypomagnesaemia and hypercalciuria with nephrocalcinosis caused by CLDN19 gene mutations. J Med Genet. doi:10.1136/jmedgenet-2016-103956
Praga M, Vara J, Gonzalez-Parra E, Andres A, Alamo C, Araque A, Ortiz A, Rodicio JL (1995) Familial hypomagnesemia with hypercalciuria and nephrocalcinosis. Kidney Int 47(5):1419–1425
Zimmermann B, Plank C, Konrad M, Stohr W, Gravou-Apostolatou C, Rascher W, Dotsch J (2006) Hydrochlorothiazide in CLDN16 mutation. Nephrol Dial Transplant 21(8):2127–2132. doi:10.1093/ndt/gfl144
Hoppe B (2012) An update on primary hyperoxaluria. Nat Rev Nephrol 8(8):467–475. doi:10.1038/nrneph.2012.113
Pelle A, Cuccurullo A, Mancini C, Sebastiano R, Stallone G, Negrisolo S, Benetti E, Peruzzi L, Petrarulo M, De Marchi M, Marangella M, Amoroso A, Giachino D, Mandrile G (2016) Updated genetic testing of Italian patients referred with a clinical diagnosis of primary hyperoxaluria. J Nephrol. doi:10.1007/s40620-016-0287-4
Hoyer-Kuhn H, Kohbrok S, Volland R, Franklin J, Hero B, Beck BB, Hoppe B (2014) Vitamin B6 in primary hyperoxaluria I: first prospective trial after 40 years of practice. Clin J Am Soc Nephrol 9(3):468–477. doi:10.2215/CJN.06820613
Cochat P, Rumsby G (2013) Primary hyperoxaluria. N Engl J Med 369(7):649–658. doi:10.1056/NEJMra1301564
Filler G, Hoppe B (2014) Combined liver-kidney transplantation for hyperoxaluria type II? Pediatr Transplant 18(3):237–239. doi:10.1111/petr.12243
Naderi G, Latif A, Tabassomi F, Esfahani ST (2014) Failure of isolated kidney transplantation in a pediatric patient with primary hyperoxaluria type 2. Pediatr Transplant 18(3):E69–E73. doi:10.1111/petr.12240
Hoppe B (2012) The enzyme 4-hydroxy-2-oxoglutarate aldolase is deficient in primary hyperoxaluria type III. Nephrol Dial Transplant 27(8):3024–3026. doi:10.1093/ndt/gfs308
Williams EL, Bockenhauer D, van’t Hoff WG, Johri N, Laing C, Sinha MD, Unwin R, Viljoen A, Rumsby G (2012) The enzyme 4-hydroxy-2-oxoglutarate aldolase is deficient in primary hyperoxaluria type 3. Nephrol Dial Transplant 27(8):3191–3195. doi:10.1093/ndt/gfs039
Zhao F, Bergstralh EJ, Mehta RA, Vaughan LE, Olson JB, Seide BM, Meek AM, Cogal AG, Lieske JC, Milliner DS (2016) Predictors of incident ESRD among patients with primary hyperoxaluria presenting prior to kidney failure. Clin J Am Soc Nephrol 11(1):119–126. doi:10.2215/CJN.02810315
Lu X, Sun D, Xu B, Pan J, Wei Y, Mao X, Yu D, Liu H, Gao B (2016) In silico screening and molecular dynamic study of nonsynonymous single nucleotide polymorphisms associated with kidney stones in the SLC26A6 gene. J Urol 196(1):118–123. doi:10.1016/j.juro.2016.01.093
Jiang Z, Asplin JR, Evan AP, Rajendran VM, Velazquez H, Nottoli TP, Binder HJ, Aronson PS (2006) Calcium oxalate urolithiasis in mice lacking anion transporter Slc26a6. Nat Genet 38(4):474–478. doi:10.1038/ng1762
Ferraro PM, D’Addessi A, Gambaro G (2013) When to suspect a genetic disorder in a patient with renal stones, and why. Nephrol Dial Transplant 28(4):811–820. doi:10.1093/ndt/gfs545
Acknowledgements
R.U. has a Chief Scientist role (secondment) at Astra Zeneca in Gothenburg, Sweden.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have nothing to disclose.
Rights and permissions
About this article
Cite this article
Mohebbi, N., Ferraro, P.M., Gambaro, G. et al. Tubular and genetic disorders associated with kidney stones. Urolithiasis 45, 127–137 (2017). https://doi.org/10.1007/s00240-016-0945-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00240-016-0945-y