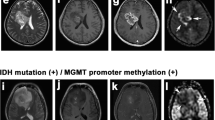
Abstract
Purpose
To determine if dynamic susceptibility contrast perfusion MR imaging (DSC-pMRI) can predict significant genomic alterations in glioblastoma (GB).
Methods
A total of 47 patients with treatment-naive GB (M/F: 23/24, mean age: 54 years, age range: 20–90 years) having DSC-pMRI with leakage correction and genomic analysis were reviewed. Mean relative cerebral blood volume (rCBV), maximum rCBV, relative percent signal recovery (rPSR), and relative peak height (rPH) were derived from T2* signal intensity-time curves by ROI analysis. Major genomic alterations of IDH1-132H, MGMT, p53, EGFR, ATRX, and PTEN status were correlated with DSC-pMRI-derived GB parameters. Statistical analysis was performed utilizing the independent-samples t-test, ROC (receiver operating characteristic) curve analysis, and multivariable stepwise regression model.
Results
rCBVmean and rCBVmax were significantly different in relation to the IDH1, MGMT, p53, and PTEN mutation status (all p < 0.05). The rPH of the p53 mutation-positive GBs (mean 5.8 ± 2.8) was significantly higher than those of the p53 mutation-negative GBs (mean 4.0 ± 1.5) (p = 0.022). Multivariable stepwise regression analysis revealed that the presence of IDH-1 mutation (B = − 2.81, p = 0.005) was associated with decreased rCBVmean; PTEN mutation (B = − 1.21, p = 0.003) and MGMT methylation (B = − 1.47, p = 0.038) were associated with decreased rCBVmax; and ATRX loss (B = − 1.05, p = 0.008) was associated with decreased rPH.
Conclusion
Significant associations were identified between DSC-pMRI-derived parameters and major genomic alterations, including IDH-1 mutation, MGMT methylation, ATRX loss, and PTEN mutation status in GB.






Similar content being viewed by others
References
Ostrom QT, Barnholtz-Sloan JS (2011) Current state of our knowledge on brain tumor epidemiology. Curr Neurol Neurosci Rep 11(3):329–335. https://doi.org/10.1007/s11910-011-0189-8
Jeon HJ, Kong DS, Park KB et al (2009) Clinical outcome of concomitant chemoradiotherapy followed by adjuvant temozolomide therapy for glioblastaomas: single-center experience. Clin Neurol Neurosurg 111(8):679–682. https://doi.org/10.1016/j.clineuro.2009.06.013
Peng S, Dhruv H, Armstrong B et al (2017) Integrated genomic analysis of survival outliers in glioblastoma. Neuro-Oncology 19(6):833–844. https://doi.org/10.1093/neuonc/now269
Karsy M, Guan J, Cohen AL, Jensen RL, Colman H (2017) New molecular considerations for glioma: IDH, ATRX, BRAF, TERT, H3 K27M. Curr Neurol Neurosci Rep 17(2):19. https://doi.org/10.1007/s11910-017-0722-5
Ozturk K, Soylu E, Tolunay S, Narter S, Hakyemez B (2019) Dynamic contrast-enhanced T1-weighted perfusion magnetic resonance imaging identifies glioblastoma immunohistochemical biomarkers via tumoral and peritumoral approach: a pilot study. World Neurosurg 128:e195–e208. https://doi.org/10.1016/j.wneu.2019.04.089
Chang PD, Malone HR, Bowden SG et al (2017) A multiparametric model for mapping cellularity in glioblastoma using radiographically localized biopsies. AJNR Am J Neuroradiol 38(5):890–898. https://doi.org/10.3174/ajnr.A5112
Rotkopf LT, Wiestler B, Preibisch C et al (2020) The wavelet power spectrum of perfusion weighted MRI correlates with tumor vascularity in biopsy-proven glioblastoma samples. PLoS One 15(1):e0228030. https://doi.org/10.1371/journal.pone.0228030
Barajas R Jr, Phillips J, Vandenberg S et al (2015) Pro-angiogenic cellular and genomic expression patterns within glioblastoma influences dynamic susceptibility weighted perfusion MRI. Clin Radiol 70(10):1087–1095
Cohen JF, Korevaar DA, Altman DG et al (2016) STARD 2015 guidelines for reporting diagnostic accuracy studies: explanation and elaboration. BMJ Open 6(11):e012799. https://doi.org/10.1136/bmjopen-2016-012799
Boxerman JL, Schmainda KM, Weisskoff RM (2006) Relative cerebral blood volume maps corrected for contrast agent extravasation significantly correlate with glioma tumor grade, whereas uncorrected maps do not. AJNR Am J Neuroradiol 27(4):859–867
Barajas RF Jr, Hodgson JG, Chang JS et al (2010) Glioblastoma multiforme regional genetic and cellular expression patterns: influence on anatomic and physiologic MR imaging. Radiology 254(2):564–576. https://doi.org/10.1148/radiol.09090663
Yang R, Nelson AC, Henzler C, Thyagarajan B, Silverstein KA (2015) ScanIndel: a hybrid framework for indel detection via gapped alignment, split reads and de novo assembly. Genome Med 7:127. https://doi.org/10.1186/s13073-015-0251-2
Saini J, Gupta RK, Kumar M et al (2019) Comparative evaluation of cerebral gliomas using rCBV measurements during sequential acquisition of T1-perfusion and T2*-perfusion MRI. PLoS One 14(4):e0215400. https://doi.org/10.1371/journal.pone.0215400
Barajas RF, Chang JS, Sneed PK, Segal MR, McDermott MW, Cha S (2009) Distinguishing recurrent intra-axial metastatic tumor from radiation necrosis following gamma knife radiosurgery using dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging. AJNR Am J Neuroradiol 30(2):367–372. https://doi.org/10.3174/ajnr.A1362
Essig M, Nguyen TB, Shiroishi MS et al (2013) Perfusion MRI: the five most frequently asked clinical questions. AJR Am J Roentgenol 201(3):W495–W510. https://doi.org/10.2214/AJR.12.9544
Mills SJ, du Plessis D, Pal P et al (2016) Mitotic activity in glioblastoma correlates with estimated extravascular extracellular space derived from dynamic contrast-enhanced MR imaging. AJNR Am J Neuroradiol 37(5):811–817. https://doi.org/10.3174/ajnr.A4623
Liu X, Mangla R, Tian W et al (2017) The preliminary radiogenomics association between MR perfusion imaging parameters and genomic biomarkers, and their predictive performance of overall survival in patients with glioblastoma. J Neuro-Oncol 135(3):553–560. https://doi.org/10.1007/s11060-017-2602-x
Binder ZA, Thorne AH, Bakas S et al (2018) Epidermal growth factor receptor extracellular domain mutations in glioblastoma present opportunities for clinical imaging and therapeutic development. Cancer Cell 34(1):163–177 e167. https://doi.org/10.1016/j.ccell.2018.06.006
Pandith AA, Qasim I, Zahoor W et al (2018) Concordant association validates MGMT methylation and protein expression as favorable prognostic factors in glioma patients on alkylating chemotherapy (Temozolomide). Sci Rep 8(1):6704. https://doi.org/10.1038/s41598-018-25169-2
Chahal M, Xu Y, Lesniak D et al (2010) MGMT modulates glioblastoma angiogenesis and response to the tyrosine kinase inhibitor sunitinib. Neuro-Oncology 12(8):822–833. https://doi.org/10.1093/neuonc/noq017
Nakada M, Kita D, Watanabe T et al (2011) Aberrant signaling pathways in glioma. Cancers (Basel) 3(3):3242–3278. https://doi.org/10.3390/cancers3033242
Heiland DH, Demerath T, Kellner E et al (2017) Molecular differences between cerebral blood volume and vessel size in glioblastoma multiforme. Oncotarget 8(7):11083–11093. https://doi.org/10.18632/oncotarget.11522
Moon WJ, Choi JW, Roh HG, Lim SD, Koh YC (2012) Imaging parameters of high grade gliomas in relation to the MGMT promoter methylation status: the CT, diffusion tensor imaging, and perfusion MR imaging. Neuroradiology 54(6):555–563. https://doi.org/10.1007/s00234-011-0947-y
Cha S, Lu S, Johnson G, Knopp EA (2000) Dynamic susceptibility contrast MR imaging: correlation of signal intensity changes with cerebral blood volume measurements. J Magn Reson Imaging 11(2):114–119. https://doi.org/10.1002/(sici)1522-2586(200002)11:2<114::aid-jmri6>3.0.co;2-s
Gupta A, Young RJ, Shah AD et al (2015) Pretreatment dynamic susceptibility contrast MRI perfusion in glioblastoma: prediction of EGFR gene amplification. Clin Neuroradiol 25(2):143–150. https://doi.org/10.1007/s00062-014-0289-3
Armocida D, Pesce A, Frati A, Santoro A, Salvati M (2020) EGFR amplification is a real independent prognostic impact factor between young adults and adults over 45yo with wild-type glioblastoma? J Neuro-Oncol 146(2):275–284. https://doi.org/10.1007/s11060-019-03364-z
Jain R, Narang J, Gutierrez J et al (2011) Correlation of immunohistologic and perfusion vascular parameters with MR contrast enhancement using image-guided biopsy specimens in gliomas. Acad Radiol 18(8):955–962. https://doi.org/10.1016/j.acra.2011.04.003
Pekmezci M, Rice T, Molinaro AM et al (2017) Adult infiltrating gliomas with WHO 2016 integrated diagnosis: additional prognostic roles of ATRX and TERT. Acta Neuropathol 133(6):1001–1016. https://doi.org/10.1007/s00401-017-1690-1
Hempel JM, Schittenhelm J, Klose U et al (2019) In vivo molecular profiling of human glioma : cross-sectional observational study using dynamic susceptibility contrast magnetic resonance perfusion imaging. Clin Neuroradiol 29(3):479–491. https://doi.org/10.1007/s00062-018-0676-2
Ryoo I, Choi SH, Kim JH et al (2013) Cerebral blood volume calculated by dynamic susceptibility contrast-enhanced perfusion MR imaging: preliminary correlation study with glioblastoma genetic profiles. PLoS One 8(8):e71704. https://doi.org/10.1371/journal.pone.0071704
Wetzel SG, Cha S, Johnson G et al (2002) Relative cerebral blood volume measurements in intracranial mass lesions: interobserver and intraobserver reproducibility study. Radiology 224(3):797–803. https://doi.org/10.1148/radiol.2243011014
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Kerem Ozturk, Esra Soylu and Zuzan Cayci. The first draft of the manuscript was written by Kerem Ozturk and Esra Soylu; and Zuzan Cayci commented on previous versions of the manuscript. All authors read and approved the final manuscript.
CRediT taxonomy:
Conceptualization: Kerem Ozturk, Esra Soylu, Zuzan Cayci; Methodology: Kerem Ozturk, Zuzan Cayci; Formal analysis and investigation: Kerem Ozturk, Esra Soylu; Writing - original draft preparation: Kerem Ozturk, Esra Soylu; Writing - review and editing: Zuzan Cayci; Funding acquisition: Zuzan Cayci; Resources: Zuzan Cayci; Supervision: Zuzan Cayci
Corresponding author
Ethics declarations
All procedures performed were in accordance with the ethical standards of the institutional research committee and with the 2013 revised Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declared no potential conflicts of interest for the research, authorship, and publication of this article.
Informed consent
Not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Ozturk, K., Soylu, E. & Cayci, Z. Correlation between dynamic susceptibility contrast perfusion MRI and genomic alterations in glioblastoma. Neuroradiology 63, 1801–1810 (2021). https://doi.org/10.1007/s00234-021-02674-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-021-02674-2