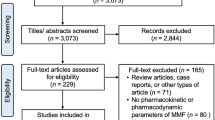
Abstract
Purpose
To investigate the associations of IMPDH and UGT1A9 polymorphisms with rejection in kidney transplant recipients taking mycophenolic acid (MPA).
Methods
PubMed, Web of Science, Embase, Cochrane Library, Wanfang Data, and the China Academic Journal Network Publishing Database were systematically searched for studies investigating the associations of IMPDH1, IMPDH2, and UGT1A9 polymorphisms with rejection in kidney transplant recipients taking MPA. Associations were evaluated by pooled odds ratios (ORs) and effect sizes (ESs) with 95% confidence intervals (CIs).
Results
Twelve studies were included in the analysis, including a total of 2342 kidney transplant recipients. The results showed that compared with the TC + CC variant genotypes, the TT genotype of IMPDH2 3757 T > C was significantly associated with a higher risk of rejection (ES = 1.60, 95% CI = 1.07–2.40, P = 0.021), while there was no significant association of the IMPDH2 3757 T > C polymorphism with acute rejection within 1 year in kidney transplant recipients (OR = 1.49, 95% CI = 0.79–2.80, P = 0.217; ES = 1.44, 95% CI = 0.88–2.36, P = 0.142). The GG genotypes of IMPDH1 125G > A and IMPDH1 106G > A were significantly associated with a higher risk of rejection (ES = 1.91, 95% CI = 1.11–3.28, P = 0.019) and acute rejection within 1 year (ES = 2.12, 95% CI = 1.45–3.10, P < 0.001) than the variant genotypes GA + AA. The TT genotype of UGT1A9 275 T > A showed a decreased risk of rejection compared with the variant genotypes TA + AA (ES = 0.44, 95% CI = 0.23–0.84, P = 0.013).
Conclusions
IMPDH1, IMPDH2, and UGT1A9 polymorphisms were associated with rejection in kidney transplant recipients, and the genetic backgrounds of patients should be considered when using MPA.





Similar content being viewed by others
Data availability
Data and materials will be available from the authors upon reasonable request.
References
Dalal P, Grafals M, Chhabra D, Gallon L (2009) Mycophenolate mofetil: safety and efficacy in the prophylaxis of acute kidney transplantation rejection. Ther Clin Risk Manag 5(1):139–149. https://doi.org/10.2147/tcrm.s3068
Sollinger HW (1995) Mycophenolate mofetil for the prevention of acute rejection in primary cadaveric renal allograft recipients. U.S. Renal Transplant Mycophenolate Mofetil Study Group. Transplantation 60(3):225–232. https://doi.org/10.1097/00007890-199508000-00003
Kaufman DB, Shapiro R, Lucey MR, CherikhR TB, Dyke DB, WS (2004) Immunosuppression: practice and trends. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg 4(Suppl 9):38–53. https://doi.org/10.1111/j.1600-6135.2004.00397.x
Picard N, Ratanasavanh D, Premaud A, Le Meur Y, Marquet P (2005) Identification of the UDP-glucuronosyltransferase isoforms involved in mycophenolic acid phase II metabolism. Drug Metab Dispos 33(1):139–146. https://doi.org/10.1124/dmd.104.001651
Bernard O, Guillemette C (2004) The main role of UGT1A9 in the hepatic metabolism of mycophenolic acid and the effects of naturally occurring variants. Drug Metab Dispos 32(8):775–778. https://doi.org/10.1124/dmd.32.8.775
Bernard O, Tojcic J, Journault K, Perusse L, Guillemette C (2006) Influence of nonsynonymous polymorphisms of UGT1A8 and UGT2B7 metabolizing enzymes on the formation of phenolic and acyl glucuronides of mycophenolic acid. Drug Metab Dispos 34(9):1539–1545. https://doi.org/10.1124/dmd.106.010553
Patel CG, Ogasawara K, Akhlaghi F (2013) Mycophenolic acid glucuronide is transported by multidrug resistance-associated protein 2 and this transport is not inhibited by cyclosporine, tacrolimus or sirolimus. Xenobiotica 43(3):229–235. https://doi.org/10.3109/00498254.2012.713531
Westley IS, Brogan LR, Morris RG, Evans AM, Sallustio BC (2006) Role of Mrp2 in the hepatic disposition of mycophenolic acid and its glucuronide metabolites: effect of cyclosporine. Drug Metab Dispos 34(2):261–266. https://doi.org/10.1124/dmd.105.006122
Picard N, Bergan S, Marquet P, van Gelder T, Wallemacq P, Hesselink DA, Haufroid V (2016) Pharmacogenetic biomarkers predictive of the pharmacokinetics and pharmacodynamics of immunosuppressive drugs. Ther Drug Monit 38(Suppl 1):S57-69. https://doi.org/10.1097/FTD.0000000000000255
Fujiyama N, Miura M, Kato S, Sone T, Isobe M, Satoh S (2010) Involvement of carboxylesterase 1 and 2 in the hydrolysis of mycophenolate mofetil. Drug Metab Dispos 38(12):2210–2217. https://doi.org/10.1124/dmd.110.034249
Kiang TKL, Partovi N, Shapiro RJ, Berman JM, Collier AC, Ensom MHH (2018) Regression and genomic analyses on the association between dose-normalized mycophenolic acid exposure and absolute neutrophil count in steroid-free, de novo kidney transplant recipients. Clin Drug Investig 38(11):1011–1022. https://doi.org/10.1007/s40261-018-0694-5
Neri A, Scalzotto E, Corradi V, Caprara C, Salin A, Cannone M, De Cal M, Romano G, Tulissi P, Cussigh AR, Montanaro D, Frigo A, Giavarina D, Chiaramonte S, Ronco C (2019) Acute rejection in kidney transplantation and the evaluation of associated polymorphisms (SNPs): the importance of sample size. Diagnosis 6(3):287–295. https://doi.org/10.1515/dx-2018-0110
Pouche L, Stojanova J, Marquet P, Picard N (2016) New challenges and promises in solid organ transplantation pharmacogenetics: the genetic variability of proteins involved in the pharmacodynamics of immunosuppressive drugs. Pharmacogenomics 17(3):277–296. https://doi.org/10.2217/pgs.15.169
Grinyó J, Vanrenterghem Y, Nashan B, Vincenti F, Ekberg H, Lindpaintner K, Rashford M, Nasmyth-Miller C, Voulgari A, Spleiss O, Truman M, Essioux L (2008) Association of four DNA polymorphisms with acute rejection after kidney transplantation. Transpl Int 21(9):879–891. https://doi.org/10.1111/j.1432-2277.2008.00679.x
Ciftci HS, Demir E, Karadeniz MS, Tefik T, Nane I, Oguz FS, Aydin F, Turkmen A (2018) Influence of uridine diphosphate-glucuronosyltransferases (1A9) polymorphisms on mycophenolic acid pharmacokinetics in patients with renal transplant. Ren Fail 40(1):395–402. https://doi.org/10.1080/0886022X.2018.1489285
Wang J, Yang JW, Zeevi A, Webber SA, Girnita DM, Selby R, Fu J, Shah T, Pravica V, Hutchinson IV, Burckart GJ (2008) IMPDH1 gene polymorphisms and association with acute rejection in renal transplant patients. Clin Pharmacol Ther 83(5):711–717. https://doi.org/10.1038/sj.clpt.6100347
Pazik J, Oldak M, Podgorska M, Lewandowski Z, Sitarek E, Ploski R, Szmidt J, Chmura A, Durlik M, Malejczyk J (2011) Lymphocyte counts in kidney allograft recipients are associated with IMPDH2 3757T>C gene polymorphism. Transpl Proc 43(8):2943–2945. https://doi.org/10.1016/j.transproceed.2011.08.037
Sombogaard F, van Schaik RH, Mathot RA, Budde K, van der Werf M, Vulto AG, Weimar W, Glander P, Essioux L, van Gelder T (2009) Interpatient variability in IMPDH activity in MMF-treated renal transplant patients is correlated with IMPDH type II 3757T > C polymorphism. Pharmacogenet Genomics 19(8):626–634. https://doi.org/10.1097/FPC.0b013e32832f5f1b
Woillard J-B, Picard N, Thierry A, Touchard G, Marquet P (2014) Associations between polymorphisms in target, metabolism, or transport proteins of mycophenolate sodium and therapeutic or adverse effects in kidney transplant patients. Pharmacogenet Genomics 24(5):256–262. https://doi.org/10.1097/fpc.0000000000000045
Kagaya H, Miura M, Saito M, Habuchi T, Satoh S (2010) Correlation of IMPDH1 gene polymorphisms with subclinical acute rejection and mycophenolic acid exposure parameters on day 28 after renal transplantation. Basic Clin Pharmacol Toxicol 107(2):631–636. https://doi.org/10.1111/j.1742-7843.2010.00542.x
Sohani ZN, Meyre D, de Souza RJ, Joseph PG, Gandhi M, Dennis BB, Norman G, Anand SS (2015) Assessing the quality of published genetic association studies in meta-analyses: the quality of genetic studies (Q-Genie) tool. BMC Genet 16:50. https://doi.org/10.1186/s12863-015-0211-2
Cheng L, Wang Y, Li X, Feng W, Weng B, Yuan Q, Xia P, Sun F (2020) Meta-analysis of the associations of CYP2B6-516G>T polymorphisms with efavirenz-induced central nervous system side effects and virological outcome in HIV-infected adults. Pharmacogenomics J 20(2):246–259. https://doi.org/10.1038/s41397-019-0112-2
Sun F, Chen Z, Yao P, Weng B, Liu Z, Cheng L (2021) Meta-analysis of ABCG2 and ABCB1 polymorphisms with sunitinib-induced toxicity and efficacy in renal cell carcinoma. Front Pharmacol 12:641075. https://doi.org/10.3389/fphar.2021.641075
Ciliao HL, Camargo-Godoy RBO, Souza MF, Zanuto A, Delfino VDA, Colus IMS (2018) Polymorphisms in IMPDH2, UGT2B7, and CES2 genes influence the risk of graft rejection in kidney transplant recipients taking mycophenolate mofetil. Mutat Res, Genet Toxicol Environ Mutagen 836(Pt B):97–102. https://doi.org/10.1016/j.mrgentox.2018.06.008
Sun M, He J, Qi X, Ding Z, Qiu Q, Zhang J, Wei X, Hu L, Zhang X, Hou J (2014) Correlation of inosine monophosphate dehydrogenase gene polymorphisms and acute rejection in renal transplantation. Chin J Exp Surg 31(12):2678–2781. https://doi.org/10.3760/ema.j.issn.1001-9030.2014.12.014
Gensburger O, Van Schaik RHN, Picard N, Le Meur Y, Rousseau A, Woillard J-B, Van Gelder T, Marquet P (2010) Polymorphisms in type I and II inosine monophosphate dehydrogenase genes and association with clinical outcome in patients on mycophenolate mofetil. Pharmacogenet Genomics 20(9):537–543. https://doi.org/10.1097/FPC.0b013e32833d8cf5
Michelon H, König J, Durrbach A, Quteineh L, Verstuyft C, Furlan V, Ferlicot S, Letierce A, Charpentier B, Fromm MF, Becquemont L (2010) SLCO1B1 genetic polymorphism influences mycophenolic acid tolerance in renal transplant recipients. Pharmacogenomics 11(12):1703–1713. https://doi.org/10.2217/pgs.10.132
Pazik J, Oldak M, Dabrowski M, Lewandowski Z, Sitarek E, Podgorska M, Wazna E, Ploski R, Szmidt J, Chmura A, Durlik M, Malejczyk J (2011) Association of UDP-glucuronosyltransferase 1A9 (UGT1A9) gene polymorphism with kidney allograft function. Ann Transplant 16(4):69–73. https://doi.org/10.12659/aot.882221
van Schaik RH, van Agteren M, de Fijter JW, Hartmann A, Schmidt J, Budde K, Kuypers D, Le Meur Y, van der Werf M, Mamelok R, van Gelder T (2009) UGT1A9 -275T>A/-2152C>T polymorphisms correlate with low MPA exposure and acute rejection in MMF/tacrolimus-treated kidney transplant patients. Clin Pharmacol Ther 86(3):319–327. https://doi.org/10.1038/clpt.2009.83
Little J, Higgins JP, Ioannidis JP, Moher D, Gagnon F, von Elm E, Khoury MJ, Cohen B, Davey-Smith G, Grimshaw J, Scheet P, Gwinn M, Williamson RE, Zou GY, Hutchings K, Johnson CY, Tait V, Wiens M, Golding J, van Duijn C, McLaughlin J, Paterson A, Wells G, Fortier I, Freedman M, Zecevic M, King R, Infante-Rivard C, Stewart A, Birkett Studies Stroga, N (2009) STrengthening the Reporting of Genetic Association Studies (STREGA): an extension of the STROBE statement. PLoS Med 6(2):e22. https://doi.org/10.1371/journal.pmed.1000022
Shah S, Harwood SM, Dohler B, Opelz G, Yaqoob MM (2012) Inosine monophosphate dehydrogenase polymorphisms and renal allograft outcome. Transplantation 94(5):486–491. https://doi.org/10.1097/TP.0b013e31825b7654
Winnicki W, Weigel G, Sunder-Plassmann G, Bajari T, Winter B, Herkner H, Sengoelge G (2010) An inosine 5′-monophosphate dehydrogenase 2 single-nucleotide polymorphism impairs the effect of mycophenolic acid. Pharmacogenomics J 10(1):70–76. https://doi.org/10.1038/tpj.2009.43
Wu TY, Peng Y, Pelleymounter LL, Moon I, Eckloff BW, Wieben ED, Yee VC, Weinshilboum RM (2010) Pharmacogenetics of the mycophenolic acid targets inosine monophosphate dehydrogenases IMPDH1 and IMPDH2: gene sequence variation and functional genomics. Br J Pharmacol 161(7):1584–1598. https://doi.org/10.1111/j.1476-5381.2010.00987.x
Na Takuathung M, Sakuludomkan W, Koonrungsesomboon N (2021) The impact of genetic polymorphisms on the pharmacokinetics and pharmacodynamics of mycophenolic acid: systematic review and meta-analysis. Clin Pharmacokinet 60(10):1291–1302. https://doi.org/10.1007/s40262-021-01037-7
Djebli N, Picard N, Rerolle JP, Le Meur Y, Marquet P (2007) Influence of the UGT2B7 promoter region and exon 2 polymorphisms and comedications on Acyl-MPAG production in vitro and in adult renal transplant patients. Pharmacogenet Genomics 17(5):321–330. https://doi.org/10.1097/FPC.0b013e32801430f8
Ting LS, Benoit-Biancamano MO, Bernard O, Riggs KW, Guillemette C, Ensom MH (2010) Pharmacogenetic impact of UDP-glucuronosyltransferase metabolic pathway and multidrug resistance-associated protein 2 transport pathway on mycophenolic acid in thoracic transplant recipients: an exploratory study. Pharmacotherapy 30(11):1097–1108. https://doi.org/10.1592/phco.30.11.1097
Pazik J, Oldak M, Lewandowski Z, Podgorska M, Sitarek E, Ploski R, Galazka Z, Kwiatkowski A, Malejczyk J, Durlik M (2013) Uridine diphosphate glucuronosyltransferase 2B7 variant p.His268Tyr as a predictor of kidney allograft early acute rejection. Transplant Proc 45(4):1516–1519. https://doi.org/10.1016/j.transproceed.2013.01.010
Dhaouadi T, Sfar I, Bardi R, Jendoubi-Ayed S, Abdallah TB, Ayed K, Gorgi Y (2013) Cytokine gene polymorphisms in kidney transplantation. Transpl Proc 45(6):2152–2157. https://doi.org/10.1016/j.transproceed.2012.12.006
Nafar M, Sahraei Z, Salamzadeh J, Samavat S, Vaziri ND (2011) Oxidative stress in kidney transplantation: causes, consequences, and potential treatment. Iran J Kidney Dis 5(6):357–372
Djamali A (2007) Oxidative stress as a common pathway to chronic tubulointerstitial injury in kidney allografts. Am J Physiol Renal Physiol 293(2):F445-455. https://doi.org/10.1152/ajprenal.00037.2007
Kim SK, Park HJ, Seok H, Jeon HS, Lee TW, Lee SH, Moon JY, Ihm CG, Kim TH, Kim YH, Kang SW, Park SJ, Jeong KH, Chung J-H (2014) Association studies of cytochrome P450, family 2, subfamily E, polypeptide 1 (CYP2E1) gene polymorphisms with acute rejection in kidney transplantation recipients. Clin Transplant 28(6):707–712. https://doi.org/10.1111/ctr.12369
Author information
Authors and Affiliations
Contributions
CL: conceptualization, methodology, investigation, data curation, formal analysis, writing; YP, WB: data curation; YM, WQ: writing—review and editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cheng, L., Yao, P., Weng, B. et al. Meta-analysis of the associations of IMPDH and UGT1A9 polymorphisms with rejection in kidney transplant recipients taking mycophenolic acid. Eur J Clin Pharmacol 78, 1227–1238 (2022). https://doi.org/10.1007/s00228-022-03311-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-022-03311-4