Abstract
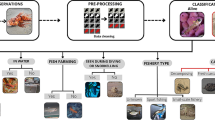
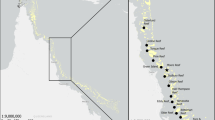
Information about the composition of pelagic communities generally lags far behind that of coastal communities, and largely derives from fisheries data that do not reflect small and non-target species. Spatiotemporal vertebrate diversity and variability associated with environmental drivers across a 3,486 km2 area of a highly productive pelagic marine ecosystem off Pacific Panama (7.583°N, 78.200°W) were described between August 2019 and March 2020 using drifting pelagic baited remote underwater video stations (BRUVS), a non-invasive fishery-independent sampling technique. From ~ 240 h of footage, we observed 26 taxa from 17 families, including 1 mammal, 3 reptile, 5 elasmobranch, and 17 teleost species. Community composition varied spatiotemporally with higher taxa richness and total abundance during the wet (April–December) than dry season (January–March). Total abundance also increased with increasing distance from shore/seafloor depth. Spatiotemporal assemblage differences were largely driven by the relative abundance of the small zooplanktivorous fishes: Psenes cyanophrys, Caranx caballus, and Decapterus spp. Psenes cyanophrys and juvenile C. caballus were commonly observed during the wet season and their abundances increased with distance from shore/seafloor depth. Decapterus spp. were observed year-round but were observed more often and in greater numbers with increasing with distance from shore/seafloor depth. This study demonstrates the practical application of drifting pelagic BRUVS to detect a diverse range of pelagic vertebrates and environmentally driven changes in pelagic communities to fill knowledge gaps in data-limited species and regions.






Similar content being viewed by others
Data availability
The datasets generated during/or analyzed during this study are available from the corresponding author on reasonable request.
References
Abreu-Grobois A, Plotkin P (2008) Lepidochelys olivacea. The IUCN Red list of threatened species. e.T11534A3292503. https://doi.org/10.2305/IUCN.UK.2008.RLTS.T11534A3292503.en
Arias A, Pressley RL (2016) Combatting illegal, unreported, and unregulated fishing with information: a case study of probable illegal fishing in the Tropical Eastern Pacific. Front Mar Sci 3:13. https://doi.org/10.3389/fmars.2016.00013
Barber RT, Chavez FP (1986) Regulation of primary productivity rate in the equatorial Pacific. Limnol Oceanogr 36:1803–1815. https://doi.org/10.4319/lo.1991.36.8.1803
Barber RT, Smith RL (1981) Coastal upwelling ecosystems. In: Longhurst A (ed) Analysis of marine ecosystems. Academic Press, New York, pp 31–68
Bouchet PJ, Meeuwig JJ (2015) Drifting baited stereo-videography: a novel sampling tool for surveying pelagic wildlife in offshore marine reserves. Ecosphere 6:137. https://doi.org/10.1890/ES14-00380.1
Bouchet PJ, Letessier TB, Caley MJ, Nichol SL, Hemmi JM, Meeuwig JJ (2020) Submerged carbonate banks aggregate pelagic megafauna in offshore tropical Australia. Front Mar Sci 7:530. https://doi.org/10.3389/fmars.2020.00530
Bullis HR, Klima EF (1972) The marine fisheries of Panama. In: Jones ML (ed) The Panamic Biota: some observations prior to a sea-level canal. Bulletin of the Biological Society of Washington No. 2, Washington, pp 167–177
Cambra M, Lara-Lizardi F, Peñaherrera C, Hearn A, Ketchum JT, Zarate P, Chacón C, Suárez-Moncada J, Herrera E, Espinoza M (2021) A first assessment of the distribution and abundance of large pelagic species at Cocos Ridge seamounts (Eastern Tropical Pacific) using drifting pelagic baited remote cameras. PLoS ONE 16:e0244343. https://doi.org/10.1371/journal.pone.0244343
Cappo M, Speare P, Death G (2004) Comparison of baited remote underwater video stations (BRUVS) and prawn (shrimp) trawls for assessments of fish biodiversity in inter-reefal areas of the Great Barrier Reef Marine Park. J Exp Mar Biol Ecol 302:123–152. https://doi.org/10.1016/j.jembe.2003.10.006
Cisneros-Montemayor AM, Harper S, Tai TC (2018) The market and shadow value of informal fish catch: a framework and application to Panama. Nat Resour Forum 42:83–92. https://doi.org/10.1111/1477-8947.12143
Collette BB, Di Natale A, Fox W, Graves J, Juan Jorda M, Pohlot B, Restrepo V, Schratwieser J (2022a) Istiophorus platypterus. The IUCN Red List of Threatened Species. e.T170338A46649664. https://doi.org/10.2305/IUCN.UK.2022-1.RLTS.T170338A46649664.en
Collette BB, Di Natale A, Fox W, Graves J, Juan Jorda M, Pohlot B, Schratwieser J. (2022b). Makaira nigricans. The IUCN Red List of Threatened Species. e.T170314A46936155. https://doi.org/10.2305/IUCN.UK.2022-1.RLTS.T170314A46936155.en
Ellis DM, DeMartini EE (1995) Evaluation of a video camera technique for indexing abundances of juvenile pink snapper, Pristipomoides filamentosus, and other Hawaiian insular shelf fishes. Fish Bull 93:67–77
Espinoza M, Cappo M, Heupel MR, Tobin AJ, Simpfendorfer CA (2014) Quantifying shark distribution patterns and species-habitat associations: implications of Marine Park Zoning. PLoS ONE 9:e106885. https://doi.org/10.1371/journal.pone.0106885
Fernández-Álamo MA, Färber-Lorda J (2006) Zooplankton and the oceanography of the eastern tropical Pacific: a review. Prog Oceanogr 69:318–359. https://doi.org/10.1016/j.pocean.2006.03.003
Fiedler PC, Lavín MF (2017) Oceanographic conditions of the Eastern Tropical Pacific. In: Glynn P, Manzello D, Enochs I (eds) Coral reefs of the Eastern Tropical Pacific. Coral reefs of the world, vol 8. Springer, Dordrecht, pp 59–83. https://doi.org/10.1007/978-94-017-7499-4_3
Forrest JAH, Bouchet PJ, Barley SC, McLennan AG, Meeuwig JJ (2021) True blue: temporal and spatial stability of pelagic wildlife at a submarine canyon. Ecosphere 12:e03423. https://doi.org/10.1002/ecs2.3423
Genin A (2004) Bio-physical coupling in the formation of zooplankton and fish aggregations over abrupt topographies. J Mar Syst 50:3–20. https://doi.org/10.1016/j.jmarsys.2003.10.008
Gooding RM, Magnuson JJ (1967) Ecological significance of a drifting object to pelagic fishes. Pac Sci 21:486–497
Harper S, Guzmán HM, Zylich K, Zeller D (2014) Reconstructing Panama’s total fisheries catches from 1950 to 2010: highlighting data deficiencies and management needs. MarFish Rev 76:51–65. https://doi.org/10.7755/MFR.76.1_2.3
Hartig F (2020) DHARMa: residual diagnostics for hierarchical (Multi-Level/Mixed) regression models. R package version 0.3.3.0. https://CRAN.R-project.org/package=DHARMa. Accessed 4 Oct 2023
Haulsee DE, Blondin HE, Logan RK, Crowder LB (2022) Where do the billfish go? Using recreational catch data to relate local and basin scale environmental conditions to billfish occurrence in the Eastern Tropical Pacific. Fish Oceanogr 31:135–148. https://doi.org/10.1111/fog.12567
Huang B, Liu C, Banzon VF, Freeman E, Graham G, Hankins B, Smith TM, Zhang H (2020): NOAA 0.25-degree daily optimum interpolation sea surface temperature (OISST), Version 2.1. NOAA National Centers for Environmental Information. https://doi.org/10.25921/RE9P-PT57. Accessed July 14, 2020.
Hubbs C (1929) Psenes cyanophrys, an Oceanic Fish, New for the U.S. Copeia 171:34–36. https://doi.org/10.2307/1436611
IATTC (2011) Tunas and billfishes in the Eastern Tropical Pacific Ocean in 2010. Fishery Status Report 9. https://iattc.org/GetAttachment/2daf4658-7f00-4c3e-a371-dc4daeb74880/IATTC-82-05_Tunas-and-billfishes-in-the-eastern-Pacific-Ocean-2010.pdf
Johnson KS, Chavez FP, Friederich GE (1999) Continental-shelf sediment as a primary source of iron for coastal phytoplankton. Nature 398:697–700. https://doi.org/10.1038/19511
Karstensen J, Stramma L, Visbeck M (2008) Oxygen minimum zones in the eastern tropical Atlantic and Pacific oceans. Prog Oceanogr 77:331–350. https://doi.org/10.1016/j.pocean.2007.05.009
Kawamura G, Nishimura W, Ueda S, Nishi T (1981) Vision in tunas and marlins. Mem Kagoshima Univ Res Senter S Pacific 1:5–47
Kilibarda M, Hengl T, Heuvelink GBM, Graeler B, Pebesma E, Perčec Tadić M, Bajat B (2014) Spatio-temporal interpolation of daily temperatures for global land areas at 1 km resolution. J Geophys Res Atmos 119:2294–2313. https://doi.org/10.1002/2013JD020803
Kilibarda M, Perčec Tadić M, Hengl T, Luković J, Bajat B (2015) Global geographic and feature space coverage of temperature data in the context of spatio-temporal interpolation. Spat Stat 14:22–38. https://doi.org/10.1016/j.spasta.2015.04.005
Ladino F, Cardeñosa D, Bessudo S, Cuellar A, Muriel F, Carvajal J, Amariles D, Duarte A (2023) Monitoreo de fauna pelágica de los Montes submarinos del Pacífco colombiano usando BRUVS. Biota Colombiana. 24:e1103. https://doi.org/10.21068/2539200x.1103
Lennert-Cody CE, Buckland ST, Gerrodette T, Webb A, Barlow J, Fretwell PT, Maunder MN, Kitakado T, Moore JE, Scott MD, Skaug HJ (2018) Review of potential line-transect methodologies for estimating abundance of dolphin stocks in the eastern tropical Pacific. J Cetacean Res Manag 19:9–21. https://doi.org/10.47536/jcrm.v19i1.411
Letessier TB, Mouillot D, Bouchet PJ, Vigliola L, Fernandes MC, Thompson C, Boussarie G, Turner J, Juhel J-B, Maire E, Caley MJ, Koldewey HJ, Friedlander A, Sala E, Meeuwig JJ (2019) Remote reefs and seamounts are the last refuges for marine predators across the Indo-Pacific. PLoS Biol 17:e3000366. https://doi.org/10.1371/journal.pbio.3000366
Lezama-Ochoa N, Murua H, Hall M, Román M, Ruiz J, Vogel N, Caballero A, Sancristobal I (2017) Biodiversity and habitat characteristics of the bycatch assemblages in fish aggregating devices (FADs) and school sets in the eastern Pacific Ocean. Front Mar Sci 4:265. https://doi.org/10.3389/fmars.2017.00265
Logan RK, Vaudo JJ, Wetherbee BM, Shivji MS (2023) Patrolling the border: billfish exploit the hypoxic boundary created by the world’s largest oxygen minimum zone. J Anim Ecol.
Mair JM, Cipriani R, Guzman HM, Usan D (2012) Fishery of the green jack Caranx caballus (Osteichytes: Carangidae) in Las Perlas Archipelago. Pacific Panama. Rev Biol Trop. 60:1271–1288. https://doi.org/10.15517/rbt.v60i3.1806
Mallet D, Pelletier D (2014) Underwater video techniques for observing coastal marine biodiversity: a review of sixty years of publications (1952–2012). Fish Res 154:44–62. https://doi.org/10.1016/j.fishres.2014.01.019
Marshall A, Barreto R, Carlson J, Fernando D, Fordham S, Francis MP, Derrick D, Herman K, Jabado RW, Liu KM, Rigby CL, Romanov E (2022) Mobula birostris. The IUCN Red List of Threatened Species. e.T198921A214397182. https://doi.org/10.2305/IUCN.UK.2022-1.RLTS.T198921A214397182.en
Martin SL, Ballance LT, Groves T (2016) An ecosystem services perspective for the oceanic eastern tropical Pacific Ocean: commercial fisheries, carbon storage, recreational fishing, and biodiversity. Front Mar Sci 3:50. https://doi.org/10.3389/fmars.2016.00050
Mclean DL, Green M, Harvey ES, Williams A, Daley R, Graham KJ (2015) Comparison of baited longlines and baited underwater cameras for assessing the composition of continental slope deepwater fish assemblages off southeast Australia. Deep Sea Res Part 1 Oceanogr 98:10–20. https://doi.org/10.1016/j.dsr.2014.11.013
Meeuwig JJ, Thompson CDH, Forrest JA, Christ HJ, Letessier TB, Meeuwig DJ (2021) Pulling back the Blue curtain: a Pelagic monitoring program for the Blue Belt. Front Mar Sci 8:649123. https://doi.org/10.3389/fmars.2021.649123
Metillo EB, Aspiras-Eya AA (2014) Fatty acids in six small pelagic fish species and their crustacean prey from the Mindano Sea, Southern Phillipines. Trop Life Sci Res 25:105–115
Miloslavich P, Klein E, Díaz JM, Hernández CE, Bigatti G, Campos L, Artigas F, Castillo J, Penchaszadeh PE, Neill PE, Carranza A, Retana MV, de Astarloa D, Lewis M, Yorio P, Piriz ML, Rodríguez D, Yoneshigue-Valentin Y, Gamboa L, Martín A (2011) Marine biodiversity in the Atlantic and Pacific coasts of South America: knowledge and gaps. PLoS ONE 6:e14631. https://doi.org/10.1371/journal.pone.0014631
Oksanen J, Simpson G, Blanchet F, Kindt R, Legendre P, Minchin P, O'Hara R, Solymos P, Stevens M, Szoecs E, Wagner H, Barbour M, Bedward M, Bolker B, Borcard D, Carvalho G, Chirico M, De Caceres M, Durand S, Evangelista H, FitzJohn R, Friendly M, Furneaux B, Hannigan G, Hill M, Lahti L, McGlinn D, Ouellette M, Ribeiro Cunha E, Smith T, Stier A, Ter Braak C, Weedon J (2022) vegan: Community Ecology Package. R package version 2.6.4. https://CRAN.R-project.org/package=vegan. Accessed 4 Oct 2023
Olson RJ, Watters GM (2003) A model of the pelagic ecosystem in the eastern tropical Pacific Ocean. Inter-Am Trop Tuna Comm Bull 22:135–219
Olson RJ, Duffy LM, Kuhnert PM, Galván-Magaña F, Bocanegra-Castillo N, Altatorre-Ramírez V (2014) Decadal diet shift in yellowfin tuna Thunnus albacares suggests broad-scale food web changes in the eastern tropical Pacific Ocean. Mar Ecol Prog Ser 497:157–178. https://doi.org/10.3354/meps10609
Pennington JT, Mahoney KL, Kuwahara VS, Kolber DD, Calienes R, Chavez FP (2006) Primary production in the eastern tropical Pacific: a review. Prog Oceanogr 69:285–317. https://doi.org/10.1016/j.pocean.2006.03.012
QGIS Development Team (2020) QGIS Geographic Information System. v3.6.0 Open Source Geospatial Foundation Project. http://qgis.osgeo.org. Accessed 4 Oct 2023
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/. Accessed 4 Oct 2023
Randall JE, Carlson BA (1999) Caranx caballus, a new immigrant carangid fish to the Hawaiian islands from the Tropical Eastern Pacific. Pac Sci 53:357–360
Rigby CL, Dulvy NK, Barreto R, Carlson J, Fernando D, Fordham S, Francis MP, Herman K, Jabado RW, Liu KM, Marshall A, Pacoureau N, Romanov E, Sherley RB, Winker H (2019) Sphyrna lewini. The IUCN Red List of Threatened Species. e.T39385A2918526. https://www.iucnredlist.org/es/species/39385/2918526
Rigby CL, Carlson J, Chin A, Derrick D, Dicken M, Pacoureau N (2021a) Carcharhinus limbatus. The IUCN Red List of Threatened Species. e.T3851A2870736. https://doi.org/10.2305/IUCN.UK.2021-2.RLTS.T3851A2870736.en
Rigby CL, Sherman CS, Chin A, Simpfendorfer C (2021b) Carcharhinus falciformis. The IUCN Red List of Threatened Species. e.T39370A205782570. https://doi.org/10.2305/IUCN.UK.2021-3.RLTS.T39370A205782570.en
Santana-Garcon J, Newman SJ, Harvey ES (2014a) Development and validation of a mid-water baited stereo-video technique for investigating pelagic fish assemblages. J Exp Mar Biol Ecol 452:82–90. https://doi.org/10.1016/j.jembe.2013.12.009
Santana-Garcon J, Newman SJ, Langlois TJ, Harvey ES (2014b) Effects of a spatial closure on highly mobile fish species: an assessment using pelagic stereo-BRUVs. J Exp Mar Biol Ecol 460:153–161. https://doi.org/10.1016/j.jembe.2014.07.003
Saucedo-Lozano M, Bernal-Ornelas H, Espino-Barr E, Garcia-Boa A, Cabral-Solís EG, Puente-Gómez M (2012) Feeding habits of the Green Jack (Caranx caballus Günther, 1868) on the coast of Manzanillo, Colima, México. Open Mar Biol J 6:28–37. https://doi.org/10.2174/1874450801206010028
Seminoff JA (2004) Chelonia mydas. The IUCN Red List of Threatened Species. e.T4615A11037468. https://doi.org/10.2305/IUCN.UK.2004.RLTS.T4615A11037468.en
Shimose T, Yokawa K, Saito H, Tachihara K (2008) Seasonal occurrence and feeding habits of black marlin, Istiompax indica, around Yonaguni Island, southwestern Japan. Ichthyol Res 55:90–94. https://doi.org/10.1007/s10228-007-0004-3
Similä T, Holst JC, Christensen I (1996) Occurrence and diet of killer whales in northern Norway: seasonal patterns relative to the distribution and abundance of Norwegian spring-spawning herring. Can J Fish Aquat Sci 53:769–779. https://doi.org/10.1139/f95-253
Stevens DL Jr, Olsen AR (2004) Spatially balanced sampling of natural resources. J Am Stat Assoc 99:262–278. https://doi.org/10.1198/016214504000000250
Thompson CDH, Meeuwig JJ, Brown J, Richardson AJ, Friedlander AM, Miller PI, Weber SB (2021) Spatial variation in Pelagic wildlife assemblages in the ascension island marine protected area: implications for monitoring and management. Front Mar Sci 8:634599. https://doi.org/10.3389/fmars.2021.634599
Vilchis LI, Balance LT, Watson W (2009) Temporal variability of neustonic ichthyoplankton assemblages of the eastern Pacific warm pool: can community structure be linked to climate variability? Deep-Sea Res i: Oceanogr Res Pap 56:125–140. https://doi.org/10.1016/j.dsr.2008.08.004
Vogel EF, Biuw M, Blanchet MA, Jonsen ID, Mul E, Johnsen E, Hjøllo SS, Olsen MT, Dietz R, Rikardsen A (2021) Killer whale movements on the Norwegian shelf are associated with herring density. Mar Ecol Prog Ser 665:217–231. https://doi.org/10.3354/meps13685
Whitmarsh SK, Fairweather PG, Huveneers C (2017) What is Big BRUVver up to? Methods and uses of baited underwater video. Rev Fish Biol Fish 27:53–73. https://doi.org/10.1007/s11160-016-9450-1
Wood S (2017) Generalized additive models: an introduction with R, 2nd edn. Chapman and Hall, CRC
Acknowledgements
This research was made possible by funding from the Guy Harvey Foundation, the Guardians of the Eastern Tropical Pacific Seascape donor group, and Nova Southeastern University, and substantial in-kind logistical support in the field provided by Tropic Star Lodge. We are grateful to the employees of Tropic Star Lodge, especially the captains and mates for their assistance with fieldwork. We also thank Dr. Tracey Sutton for his help identifying pelagic fishes, and Drs. Erin Seney and Kate Mansfield for their help in identifying marine turtle species.
Funding
This work was supported by the Guy Harvey Foundation, the Guardians of the Eastern Tropical Pacific Seascape donor group, and Nova Southeastern University. Substantial in-kind logistical support in the field was provided by Tropic Star Lodge.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. TSP: conducted the fieldwork and video analysis. JJV: analyzed the data. JJV and TSP: wrote the manuscript. All authors provided comments on drafts of the manuscript and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Ethical approval
This was an observational study performed using procedures approved by Nova Southeastern University’s Institutional Animal Care and Use Committee (2017.11.MS1). Work in Panama was conducted under a permit from the Ministerio de Ambiente, República de Panamá (SE/A-64–19).
Additional information
Responsible Editor: S. Hamilton.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vaudo, J.J., Plum, T.S., Logan, R.K. et al. Diversity and spatiotemporal variation of an Eastern Tropical Pacific pelagic vertebrate community assessed with drifting pelagic BRUVS. Mar Biol 170, 157 (2023). https://doi.org/10.1007/s00227-023-04308-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-023-04308-y