Abstract
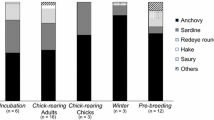
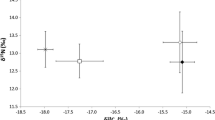
The diet of widely distributed species is influenced by the availability of food resources, which can vary according to local conditions. Thus, heterogeneity in diet patterns can help understand population structure and illustrate biogeographic boundaries. In this study, published and unpublished datasets of regurgitated material and stable isotopes of carbon (δ13C) and nitrogen (δ15N) from whole blood of brown boobies (Sula leucogaster) in the southwestern Atlantic Ocean (27°51′S to 0°55′N) were used to assess dietary spatial patterns. The variations in prey composition and isotopic niche breadth were associated with colony location (coastal/oceanic), genetic population structure, and marine biogeographic zonings—Large Marine Ecosystems, Longhurst’s Biogeochemical Provinces, and Spalding’s Provinces and Ecoregions. Boobies from coastal and southern colonies showed higher diversity in their diet compared with boobies from oceanic and northern colonies, given the presence of demersal taxa associated with bottom trawling discards. The variation in prey composition from regurgitates and stable isotopes was well framed with genetic structure and biogeographic boundaries, although less fitting with Spalding’s zoning. The brown booby showed trophic plasticity throughout the study area, suggesting that their diet is shaped by food resources available around the colonies, including those from fishery discards. These results demonstrate that highly mobile vertebrates are potentially useful samplers of the marine environment, able to indicate the diversity of prey organisms available in the foraging area through diet, and support biogeographic zonings. Furthermore, diet composition associated with population structure sheds light on local adaptation as a potential mechanism for promoting/disrupting gene flow in seabirds.
Graphical abstract






Similar content being viewed by others
Data availability
All data and the R script are publicly available through the GitHub repository: https://github.com/SeabirdEcologyUFRGS/DietBiogeography
References
Abelha MCF, Agostinho AA, Goulart E (2001) Plasticidade trófica em peixes de água doce. Acta Scientiarum Biol Sci 23(2):425–434
Alves VS, Soares ABA, Couto GS, Efe MA, Ribeiro ABB (2004) Aves marinhas de Abrolhos–Bahia, Brasil. In: Branco JO (ed) Aves marinhas e insulares brasileiras: bioecologia e conservação. Univali, Itajaí, pp 213–232
Amorim RB, Monteiro-Neto C (2016) Marine protected area and the spatial distribution of the gill net fishery in Copacabana, Rio de Janeiro, RJ, Brazil. Braz J Biol 76(1):1–9. https://doi.org/10.1590/1519-6984.06614
Araujo M, Cintra M (2009) Modelagem matemática da circulação oceânica na região equatorial. In: Viana DL, Hazin FHV (eds) O Arquipélago de São Pedro e São Paulo: 10 anos de Estação Científica. SECIRM, Brasília, pp 106–113
Barbraud C, Rolland V, Jenouvrier S, Nevoux M, Delord K, Weimerskirch H (2012) Effects of climate change and fisheries bycatch on Southern Ocean seabirds: a review. Mar Ecol Prog Ser 454:285–307. https://doi.org/10.3354/meps09616
Barnagaud J, Mazet N, Munoz F, Grenié M, Denelle P, Sobral M, Kissling WD, Şekercioğlu ÇH, Violle C (2019) Functional biogeography of dietary strategies in birds. Global Ecol Biogeogr 28(7):1004–1017. https://doi.org/10.1111/geb.12910
Baselga A, Orme D, Villeger S, De Bortoli J, Leprieur F, Logez M (2021) Betapart: partitioning beta diversity into turnover and nestedness components. R package version 1.5.4. https://CRAN.R-project.org/package=betapart. Accessed 25 May 2022
Bauer AB, Gomes Fischer L, Di Dario F, Mincarone MM (2017) Marine fishes (Elasmobranchii and Teleostei) from the Santana Archipelago, a marine protected area in the southwestern Atlantic. Mar Biol Res 13(8):813–831. https://doi.org/10.1080/17451000.2017.1302090
Bearhop S, Teece MA, Waldron S, Furness RW (2000) Influence of lipid and uric acid on δ13C and δ15N values of avian blood: implications for trophic studies. Auk 117(2):504–507. https://doi.org/10.1093/auk/117.2.504
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Series B 57(1):289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Bighetti GP, Padilha JA, Cunha LST, Kasper D, Malm O, Mancini PL (2021) Bioaccumulation of mercury is equal between sexes but different by age in seabird (Sula leucogaster) population from southeast coast of Brazil. Environ Pollut 285:117222. https://doi.org/10.1016/j.envpol.2021.117222
Bond AL, Jones IL (2009) A practical introduction to stable-isotope analysis for seabird biologists: approaches, cautions and caveats. Mar Ornithol 37(3):183–188
Branco JO, Verani JR (2006) Análise quali-quantitativa da ictiofauna acompanhante na pesca do camarão sete-barbas, na Armação do Itapocoroy, Penha, Santa Catarina. Rev Bras Zool 23(2):381–391. https://doi.org/10.1590/S0101-81752006000200011
Branco JO, Fracasso HAA, Machado IF, Bovendorp MS, Verani JR (2005) Dieta de Sula leucogaster Boddaert (Sulidae, Aves), nas Ilhas Moleques do Sul, Florianópolis, Santa Catarina, Brasil. Rev Bras Zool 22(4):1044–1049. https://doi.org/10.1590/S0101-81752005000400033
Briggs JC (1974) Marine zoogeography. McGraw-Hill, New York
Bugoni L, Vooren CM (2004) Feeding ecology of the common tern Sterna hirundo in a wintering area in southern Brazil. Ibis 146(3):438–453. https://doi.org/10.1111/j.1474-919X.2004.00277.x
Castillo-Guerrero JA, Lerma M, Mellink E, Suazo-Guillén E, Peñaloza-Padilla EA (2016) Environmentally-mediated flexible foraging strategies in brown boobies in the Gulf of California. Ardea 104(1):33–47. https://doi.org/10.5253/arde.v104i1.a3
Cattani AP, de Santos LO, Spach HL, Budel R, Gondim Guanais JHD (2011) Avaliação da ictiofauna da fauna acompanhante da pesca do camarão sete-barbas no município de Pontal do Paraná, Brasil. Bol Inst Pesca 37(2):247–260
Coelho EP, Alves VS, Soares ABA, Couto GS, Efe MA, Ribeiro ABB, Vielliard J, Gonzaga LP (2004) O atobá-marrom (Sula leucogaster) na ilha de Cabo Frio, Arraial do Cabo, Rio de Janeiro, Brasil. In: Branco JO (ed) Aves marinhas e insulares brasileiras: bioecologia e conservação. Univali, Itajaí, pp 233–254
Cunha L, Alves V, Rajão H, Lanna A (2013) Aves do Monumento Natural das Ilhas Cagarras. In: Moraes F, Bertoncini A, Aguiar A (eds) História, pesquisa e biodiversidade do Monumento Natural das Ilhas Cagarras. Museu Nacional, Rio de Janeiro, pp 178–206
Dalerum F, Angerbjörn A (2005) Resolving temporal variation in vertebrate diets using naturally occurring stable isotopes. Oecologia 144(4):647–658. https://doi.org/10.1007/s00442-005-0118-0
Danckwerts DK, Humeau L, Pinet P, McQuaid CD, Le Corre M (2021) Extreme philopatry and genetic diversification at unprecedented scales in a seabird. Sci Rep 11:6834. https://doi.org/10.1038/s41598-021-86406-9
Drymon JM, Powers SP, Carmichael RH (2012) Trophic plasticity in the Atlantic sharpnose shark (Rhizoprionodon terraenovae) from the north central Gulf of Mexico. Environ Biol Fish 95(1):21–35. https://doi.org/10.1007/s10641-011-9922-z
Duffy LM, Kuhnert PM, Pethybridge HR, Young JW, Olson RJ, Logan JM, Goñi N, Romanov E, Allain V, Staudinger MD, Abecassis M, Choy CA, Hobday AJ, Simier M, Galván-Magaña F, Potier M, Ménard F (2017) Global trophic ecology of yellowfin, bigeye, and albacore tunas: understanding predation on micronekton communities at ocean-basin scales. Deep Sea Res Part II 140:55–73. https://doi.org/10.1016/j.dsr2.2017.03.003
Floeter SR, Rocha LA, Robertson DR, Joyeux JC, Smith-Vaniz WF, Wirtz P, Edwards AJ, Barreiros JP, Ferreira CEL, Gasparini JL, Brito A, Falcón JM, Bowen BW, Bernardi G (2007) Atlantic reef fish biogeography and evolution. J Biogeogr 35(1):22–47. https://doi.org/10.1111/j.1365-2699.2007.01790.x
Fohringer C, Dudka I, Spitzer R, Stenbacka F, Rzhepishevska O, Cromsigt JPGM, Gröbner G, Ericsson G, Singh NJ (2021) Integrating omics to characterize eco-physiological adaptations: how moose diet and metabolism differ across biogeographic zones. Ecol Evol 11(7):3159–3183. https://doi.org/10.1002/ece3.7265
Forbes E (1859) The natural history of the European seas. John Van Voorst, London
Fricke R, Eschmeyer WN, Van der Laan R (2022) Eschmeyer’s catalog of fishes: genera, species, references. http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain. Accessed 30 May 2022
Friesen VL (2015) Speciation in seabirds: why are there so many species and why aren’t there more? J Ornithol 156(1):27–39. https://doi.org/10.1007/s10336-015-1235-0
Friesen VL, Burg TM, McCoy KD (2007) Mechanisms of population differentiation in seabirds. Mol Ecol 16(9):1765–1785. https://doi.org/10.1111/j.1365-294X.2006.03197.x
Fry B (2006) Stable isotope ecology. Springer, New York
Garvey JE, Whiles MR (2017) Trophic ecology. CRC Press, Boca Raton
Gerking SD (1994) Feeding ecology of fish. Academic Press, San Diego
Gilmour ME, Castillo-Guerrero JA, Fleishman AB, Hernández-Vázquez S, Young HS, Shaffer SA (2018) Plasticity of foraging behaviors in response to diverse environmental conditions. Ecosphere 9(7):e02301. https://doi.org/10.1002/ecs2.2301
Grant PR, Grant BR (2002) Unpredictable evolution in a 30-year study of Darwin’s finches. Science 296(5568):707–711. https://doi.org/10.1126/science.1070315
Harding AMA, Piatt JF, Schmutz JA, Shultz MT, Pelt TIV, Kettle AB, Speckman SG (2007) Prey density and the behavioral flexibility of a marine predator: the common murre (Uria aalge). Ecology 88(8):2024–2033. https://doi.org/10.1890/06-1695.1
Harrison CS, Hida TS, Seki MP (1983) Hawaiian seabird feeding ecology. Wildl Monogr 85:3–71
Harrison CS, Hida TS, Seki MP (1984) The diet of the brown booby Sula leucogaster and masked booby Sula dactylatra on Rose Atoll, Samoa. Ibis 126(4):588–590. https://doi.org/10.1111/j.1474-919X.1984.tb02082.x
Hartl DL, Clark AG (1997) Principles of population genetics. Sinauer Associates, Sunderland
Jablonski S, de Azevedo AF, Moreira LHA (2006) Fisheries and conflicts in Guanabara Bay, Rio de Janeiro, Brazil. Braz Arch Biol Technol 49(1):79–91. https://doi.org/10.1590/S1516-89132006000100010
Jackson AL, Inger R, Parnell AC, Bearhop S (2011) Comparing isotopic niche widths among and within communities: SIBER—stable isotope Bayesian ellipses in R. J Anim Ecol 80(3):595–602. https://doi.org/10.1111/j.1365-2656.2011.01806.x
Kreft H, Jetz W (2010) A framework for delineating biogeographical regions based on species distributions: global quantitative biogeographical regionalizations. J Biogeog 37(11):2029–2053. https://doi.org/10.1111/j.1365-2699.2010.02375.x
Krul R (2004) Aves marinhas costeiras do Paraná. In: Branco JO (ed) Aves marinhas e insulares brasileiras: bioecologia e conservação. Univali, Itajaí, pp 37–56
Larkin PA (1956) Interspecific competition and population control in freshwater fish. J Fish Res Board Can 13(3):327–342
Legendre P, Legendre L (2012) Numerical ecology, 3rd edn. Elsevier, Amsterdam
Lombal AJ, O’dwyer JE, Friesen V, Woehler EJ, Burridge CP (2020) Identifying mechanisms of genetic differentiation among populations in vagile species: historical factors dominate genetic differentiation in seabirds. Biol Rev 95(3):625–651. https://doi.org/10.1111/brv.12580
Longhurst A, Sathyendranath S, Platt T, Caverhill C (1995) An estimate of global primary production in the ocean from satellite radiometer data. J Plankton Res 17(6):1245–1271. https://doi.org/10.1093/plankt/17.6.1245
Lozano J, Moleon M, Virgos E (2006) Biogeographical patterns in the diet of the wildcat, Felis silvestris Schreber, in Eurasia: factors affecting the trophic diversity. J Biogeogr 33(6):1076–1085. https://doi.org/10.1111/j.1365-2699.2006.01474.x
Lucena FM, Vaske T, Ellis JR, O’Brien CM (2000) Seasonal variation in the diets of bluefish, Pomatomus saltatrix (Pomatomidae) and striped weakfish, Cynoscion guatucupa (Sciaenidae) in southern Brazil: implications of food partitioning. Environ Biol Fish 57(4):423–434. https://doi.org/10.1023/A:1007604424423
Magozzi S, Yoll A, Vander Zanden HB, Wunder MB, Trueman CN (2017) Using ocean models to predict spatial and temporal variation in marine carbon isotopes. Ecosphere 8(5):e01763. https://doi.org/10.1002/ecs2.1763
Magurran AE (2003) Measuring biological diversity. Wiley-Blackwell. Hoboken
Mallet-Rodrigues F (2010) Técnicas para amostragem da dieta e procedimentos para estudos do forrageamento de aves. Von Matter S, Straube FC, Accordi I, Piacentini V, Cândido-Jr JF (org) Ornitologia e conservação – ciência aplicada, técnicas de pesquisa e levantamento. Technical Books, Rio de Janeiro, pp 457–470
Mancini PL, Bugoni L (2014) Resources partitioning by seabirds and their relationship with other consumers at and around a small tropical archipelago. ICES J Mar Sci 71(9):2599–2607. https://doi.org/10.1093/icesjms/fsu105
Mancini PL, Hobson K, Bugoni L (2014) Role of body size in shaping the trophic structure of tropical seabird communities. Mar Ecol Prog Ser 497:243–257. https://doi.org/10.3354/meps10589
Mancini PL, Valim EEM, Bauer AB, Fischer LG (2023) Intraspecific trophic variation in brown booby (Sula leucogaster) from the Southwestern Atlantic. Mar Biol 170(1):1. https://doi.org/10.1007/s00227-022-04134-8
Mellink E, Domínguez J, Luévano J (2001) Diet of eastern Pacific brown boobies Sula leucogaster brewsteri on Isla San Jorge, north-eastern Gulf of California, and an April comparison with diets on the middle Gulf of California. Mar Ornithol 29(1):23–28
Menni RC, Jaureguizar AJ, Stehmann MFW, Lucifora LO (2010) Marine biodiversity at the community level: zoogeography of sharks, skates, rays and chimaeras in the southwestern Atlantic. Biodivers Conserv 19(3):775–796. https://doi.org/10.1007/s10531-009-9734-z
Miller MGR, Silva FRO, Machovsky-Capuska GE, Congdon BC (2018) Sexual segregation in tropical seabirds: drivers of sex specific foraging in the brown booby Sula leucogaster. J Ornithol 159(2):425–437. https://doi.org/10.1007/s10336-017-1512-1
Montevecchi WA, Myers A (1996) Dietary changes of seabirds indicate shifts in pelagic food webs. Sarsia 80(4):313–322. https://doi.org/10.1080/00364827.1996.10413606
Moraes F, Rebouças M, Jordão S, Pereira LA, Santos E, Amorim R, Lobo V, Silva-Jr L, Cabral D, Bertoncini A (2013) A pesca no entorno do Monumento Natural das Ilhas Cagarras. In: Moraes F, Bertoncini A, Aguiar A (eds) História, pesquisa e biodiversidade do Monumento Natural das Ilhas Cagarras. Museu Nacional, Rio de Janeiro, pp 246–268
Morris-Pocock JA, Anderson DJ, Friesen VL (2011) Mechanisms of global diversification in the brown booby (Sula leucogaster) revealed by uniting statistical phylogeographic and multilocus phylogenetic methods. Mol Ecol 20(13):2835–2850. https://doi.org/10.1111/j.1365-294X.2011.05132.x
Moseley C, Grémillet D, Connan M, Ryan PG, Mullers RHE, van der Lingen CD, Miller TW, Coetzee JC, Crawford RJM, Sabarros P, McQuaid CD, Pichegru L (2012) Foraging ecology and ecophysiology of Cape gannets from colonies in contrasting feeding environments. J Exp Mar Biol Ecol 422–423:29–38. https://doi.org/10.1016/j.jembe.2012.04.002
Nelson B (1978) The Sulidae: gannets and boobies. Oxford University Press, New York
Newsome SD, Rio CM, Bearhop S, Phillips DL (2007) A niche for isotope ecology. Front Ecol Environ 8(5):429–436. https://doi.org/10.1890/060150.1
Nunes GT, Bugoni L (2018) Local adaptation drives population isolation in a tropical seabird. J Biogeogr 45(2):332–341. https://doi.org/10.1111/jbi.13142
Nunes GT, Mancini PL, Bugoni L (2017) When Bergmann’s rule fails: evidences of environmental selection pressures shaping phenotypic diversification in a widespread seabird. Ecography 40(3):365–375. https://doi.org/10.1111/ecog.02209
Nunes GT, Bertrand S, Bugoni L (2018) Seabirds fighting for land: phenotypic consequences of breeding area constraints at a small remote archipelago. Sci Rep 8:665. https://doi.org/10.1038/s41598-017-18808-7
Nunes GT, Efe MA, Barreto CT, Gaiotto JV, Silva AB, Vilela F, Roy A, Bertrand S, Costa PG, Bianchini A, Bugoni L (2022) Ecological trap for seabirds due to the contamination caused by the Fundão dam collapse, Brazil. Sci Tot Environ 807:151486. https://doi.org/10.1016/j.scitotenv.2021.151486
Ogle DH, Doll JC, Wheeler P, Dinno A (2021) FSA: fisheries stock analysis. R package version 0.9.1. https://github.com/droglenc/FSA. Accessed 25 May 2022
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens HH, Szoecs E, Wagner H (2020) Vegan: community ecology package. R package version 2.5–7. https://CRAN.R-project.org/package=vegan. Accessed 25 May 2022
Parrish JD (2000) Behavioral, energetic and conservation: implications of foraging plasticity during migration. Stud Avian Biol 20:53–70
Pereira AN, Pantoja C, Luque JL, Timi JT (2014) Parasites of Urophycis brasiliensis (Gadiformes: Phycidae) as indicators of marine ecoregions in coastal areas of the South American Atlantic. Parasitol Res 113(11):4281–4292. https://doi.org/10.1007/s00436-014-4106-3
Piatt JF, Arimitsu ML, Sydeman WJ, Thompson SA, Renner H, Zador S, Douglas D, Hatch S, Kettle A, Williams J (2018) Biogeography of pelagic food webs in the North Pacific. Fish Oceanogr 27(4):366–380. https://doi.org/10.1111/fog.12258
Pilot M, Jędrzejewski W, Sidorovich VE, Meier-Augenstein W, Hoelzel AR (2012) Dietary differentiation and the evolution of population genetic structure in a highly mobile carnivore. PLoS ONE 7(6):e39341. https://doi.org/10.1371/journal.pone.0039341
Pinheiro HT, Rocha LA, Macieira RM, Carvalho-Filho A, Anderson AB, Bender MG, Di Dario F, Ferreira CEL, Figueiredo-Filho J, Francini-Filho R, Gasparini JL, Joyeux J-C, Luiz OJ, Mincarone MM, Moura RL, de Nunes J, Quimbayo JP, Rosa RS, Sampaio CLS, Sazima I, Simon T, Vila-Nova DA, Floeter SR (2018) South-western Atlantic reef fishes: zoogeographical patterns and ecological drivers reveal a secondary biodiversity centre in the Atlantic Ocean. Divers Distrib 24(7):951–965. https://doi.org/10.1111/ddi.12729
Piola AR, Campos EJD, Möller OO, Charo M, Martinez C (2000) Subtropical shelf front off eastern South America. J Geophys Res 105(C3):6565–6578. https://doi.org/10.1029/1999JC000300
Quillfeldt P, Cherel Y, Delord K, Weimerkirch H (2015) Cool, cold or colder? Spatial segregation of prions and blue petrels is explained by differences in preferred sea surface temperatures. Biol Lett 11(4):20141090. https://doi.org/10.1098/rsbl.2014.1090
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ramírez F, Vicente-Sastre D, Afán I, Igual J, Oro D, Forero M (2021) Stable isotopes in seabirds reflect changes in marine productivity patterns. Mar Ecol Prog Ser 662:169–180. https://doi.org/10.3354/meps13615
Reygondeau G, Maury O, Beaugrand G, Fromentin JM, Fonteneau A, Cury P (2012) Biogeography of tuna and billfish communities. J Biogeog 39(1):114–129. https://doi.org/10.1111/j.1365-2699.2011.02582.x
Richardson PL, Walsh D (1986) Mapping climatological seasonal variations of surface currents in the tropical Atlantic using ship drifts. J Geophys Res 91(C9):10537. https://doi.org/10.1029/JC091iC09p10537
Richardson JL, Urban MC, Bolnick DI, Skelly DK (2014) Microgeographic adaptation and the spatial scale of evolution. Trends Ecol Evol 29(3):165–176. https://doi.org/10.1016/j.tree.2014.01.002
Robinette DP, Howar J, Sydeman WJ, Nur N (2007) Spatial patterns of recruitment in a demersal fish as revealed by seabird diet. Mar Ecol Prog Ser 352:259–268. https://doi.org/10.3354/meps07079
Romano A, Séchaud R, Roulin A (2020) Global biogeographical patterns in the diet of a cosmopolitan avian predator. J Biogeogr 47(7):1467–1481. https://doi.org/10.1111/jbi.13829
Sanford E, Roth MS, Johns GC, Wares JP, Somero GN (2003) Local selection and latitudinal variation in a marine predator-prey interaction. Science 300(5622):1135–1137. https://doi.org/10.1126/science.1083437
Santos MCF, Almeida L, Silva CGM (2008) Avaliação quali-quantitativa da ictiofauna acompanhante na pesca do camarão sete-barbas, Xiphopenaeus kroyeri (Heller, 1862) no município de Caravelas (Bahia-Brasil). Bol Tec Cient CEPENE 16(1):99–106
Sazima I, Sazima C (2008) Occupational hazards: brown boobies (Sula leucogaster) as a nuisance to fishermen at Fernando de Noronha Island, with comments on injuries inflicted to the birds. Rev Bras Ornitol 16(3):250–251
Schreiber EA, Burger J (eds) (2002) Biology of marine birds. CRC Press, Boca Raton
Schreiber EA, Norton RL (2020) Brown booby (Sula leucogaster). In: Billerman SM (ed) Birds of the world. Cornell Lab of Ornithology, Ithaca. https://doi.org/10.2173/bow.brnboo.01
Sealey KS, Bustamante G (1999) Setting geographic priorities for marine conservation in Latin America and the Caribbean. The Nature Conservancy. Arlington
Sedrez MC, Barrilli GHC, de Moura ENF, Barreiros JP, Branco JO, Verani JR (2021) Feeding habits of Paralonchurus brasiliensis (Perciformes: Sciaenidae) from south of Brazil. Acta Biol Colomb 26(3):335–344. https://doi.org/10.15446/abc.v26n3.80609
Sexton JP, Hangartner SB, Hoffmann AA (2014) Genetic isolation by environment or distance: which pattern of gene flow is most common? Evolution 68(1):1–15. https://doi.org/10.1111/evo.12258
Sherman K (1994) Sustainability, biomass yields, and health of coastal ecosystems: an ecological perspective. Mar Ecol Prog Ser 112:277–301. https://doi.org/10.3354/meps112277
Sick H (1997) Ornitologia brasileira, 2nd edn. Nova Fronteira, Rio de Janeiro
Silva ER, Sancinetti GS, Fransozo A, Azevedo A, Costa RC (2016) Abundance and spatial-temporal distribution of the shrimp Xiphopenaeus kroyeri (Decapoda: Penaeidae): an exploited species in southeast Brazil. Braz J Biol 76(3):764–773. https://doi.org/10.1590/1519-6984.01814
Silva-Jr DR, Paranhos R, Vianna M (2016) Spatial patterns of distribution and the influence of seasonal and abiotic factors on demersal ichthyofauna in an estuarine tropical bay. J Fish Biol 89(1):821–846. https://doi.org/10.1111/jfb.13033
Soares L, Muto E, Lopez J, Clauzet G, Valiela I (2014) Seasonal variability of δ13C and δ15N of fish and squid in the Cabo Frio upwelling system of the southwestern Atlantic. Mar Ecol Prog Ser 512:9–21. https://doi.org/10.3354/meps10948
Spalding MD, Fox HE, Allen GR, Davidson N, Ferdaña ZA, Finlayson M, Halpern BS, Jorge MA, Lombana A, Lourie SA, Martin KD, McManus E, Molnar J, Recchia CA, Robertson J (2007) Marine ecoregions of the world: a bioregionalization of coastal and shelf areas. Bioscience 57(7):573–583. https://doi.org/10.1641/B570707
Spitz J, Richard E, Meynier L, Pusineri C, Ridoux V (2006) Dietary plasticity of the oceanic striped dolphin, Stenella coeruleoalba, in the neritic waters of the Bay of Biscay. J Sea Res 55(4):309–320. https://doi.org/10.1016/j.seares.2006.02.001
Valentin JL (2001) The Cabo Frio upwelling system, Brazil. In: Seeliger U, Kjerfve B (eds) Coastal marine ecosystems of Latin America. Springer, New York, pp 97–106. https://doi.org/10.1007/978-3-662-04482-7_8
Van Noord JE, Lewallen EA, Pitman RL (2013) Flyingfish feeding ecology in the eastern Pacific: prey partitioning within a speciose epipelagic community. J Fish Biol 83(2):326–342. https://doi.org/10.1111/jfb.12173
Viana DF, Hazin FHV, Andrade HA, Nunes M, Viana DL (2015) Fisheries in the Saint Peter and Saint Paul archipelago: 13 years of monitoring. Bol Inst Pesca 41(2):239–248
Vianna M, Almeida T (2005) Bony fish bycatch in the southern Brazil pink shrimp (Farfantepenaeus brasiliensis and F. paulensis) fishery. Braz Arch Biol Technol 48(4):611–623. https://doi.org/10.1590/S1516-89132005000500014
Votier SC, Sherley RB (2017) Seabirds. Curr Biol 27:R448–R450. https://doi.org/10.1016/j.cub.2017.01.042
Votier SC, Bearhop S, Witt MJ, Inger R, Thompson D, Newton J (2010) Individual responses of seabirds to commercial fisheries revealed using GPS tracking, stable isotopes and vessel monitoring systems. J Appl Ecol 47(2):487–497. https://doi.org/10.1111/j.1365-2664.2010.01790.x
Vries A, Ripley BD (2020) ggdendro: create dendrograms and tree diagrams using “ggplot2”. R package version 0.1.22. https://CRAN.R-project.org/package=ggdendro. Accessed 25 May 2022
Wang IJ, Bradburd GS (2014) Isolation by environment. Mol Ecol 23(23):5649–5662. https://doi.org/10.1111/mec.12938
Watanuki Y, Yamamoto M, Okado J, Ito M, Sydeman W (2022) Seabird reproductive responses to changing climate and prey communities are mediated by prey packaging. Mar Ecol Prog Ser 683:179–194. https://doi.org/10.3354/meps13943
Whitehead PJP (ed) (1985) Clupeoid fishes of the world (suborder Clupeoidei). FAO, Rome
Wickham H (2011) The split-apply-combine strategy for data analysis. J Stat Softw 40(1):1–29
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York
Widmann M, Kato A, Raymond B, Angelier F, Arthur B, Chastel O, Pellé M, Raclot T, Ropert-Coudert Y (2015) Habitat use and sex-specific foraging behavior of Adélie penguins throughout the breeding season in Adélie Land, East Antartica. Mov Ecol 3:30. https://doi.org/10.1186/s40462-015-0052-7
WoRMS Editorial Board (2022) World register of marine species. https://www.marinespecies.org. Accessed 25 Jun 2022
Acknowledgements
We are grateful to many researchers who studied the diet of brown boobies during the last 30 years in the Atlantic Ocean. We also thank E.E.M. Valim, T.P.X. Nascimento, L.R.M. Porto, G.P. Bighetti, F.P. Marques, C. Campolina, C. Barbraud, and K. Delord for their support in fieldwork. F.L. Rodrigues provided important contribution in prey identification and classification. We also thank the two anonymous reviewers for the comments and suggestions that contribute to improving the manuscript.
Funding
The unpublished datasets were obtained with funding from the IRD Young Associated Team Program (JEAI TABASCO), the IRD mixed international laboratory program (LMI TAPIOCA), CPER Celimer (France), Fundação Boticário (Brazil), the TRIATLAS project (European Union’s Horizon 2020 research and innovation program, Grant No. 817578), the PADDLE project (European Union’s Horizon 2020 RISE program (Grant No. 734271), and the “Projeto Costões Rochosos”, which is an environmental offset measure established through a Consent Decree/Conduct Adjustment Agreement between Petrorio and the Brazilian Ministry for the Environment, with the Brazilian Biodiversity Fund (FUNBIO) as an implementer. PLM received a PNPD scholarship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES) Finance Code 001. LB had been funded by National Research Council (CNPq; Grant No. 405497/2012-1) and is a fellowship from CNPq (No. 311409/2018-0). GTN is funded by CNPq (Grant No 443328/2019-6).
Author information
Authors and Affiliations
Contributions
GTN and JJ conceptualized the idea and designed the study. GTN, PLM, SLB and LB collected and shared data. GTN and JJ analyzed the data and wrote the manuscript and PLM, SLB, MAE, and LB contributed critically to the final revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The sampling of the unpublished datasets was approved by SISBIO 64234-6 and by the ethics committee at UFRGS.
Additional information
Responsible Editor: V. Paiva.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jacoby, J., Luciano Mancini, P., Bertrand, S.L. et al. Biogeographic variation on dietary aspects of a widely distributed seabird. Mar Biol 170, 21 (2023). https://doi.org/10.1007/s00227-022-04171-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-022-04171-3