Abstract
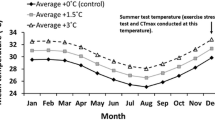
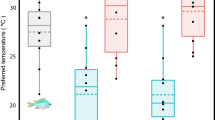
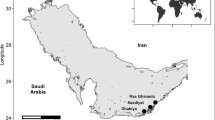
There is increasing empirical evidence of the shifting range distributions of tropical reef fishes as a response to persistent climatic warming. However, the link between the observed disparity of closely related species’ occurrence in temperate waters and their trait-based responses to winter thermal conditions is unclear. This study focuses on congeneric tropical reef fishes with similar dispersal potential (pelagic larval duration) but with varying occurrence in Kochi, southwestern Japan. The studied species include the abundantly occurring, overwintering, and reproductively established species in Kochi (adapted) and the less abundant species with no recorded overwintering and adult populations (non-adapted). This study assessed the responses of congeneric adapted and non-adapted species from two genera of Pomacentridae (Pomacentrus and Dascyllus) and one from Chaetodontidae (Chaetodon) to the winter seawater thermal range in Kochi by subjecting them to the decreasing temperature from 25 to 15 °C (1 °C d−1) in the laboratory. Both the adapted and non-adapted species demonstrated swimming and feeding reductions when temperatures approached 18 °C. However, stress-related behaviors were observed at 17 °C in the non-adapted species compared with 16 °C in their adapted congeners. The non-adapted species also exhibited relatively lower growth rates and higher mortality rates of > 50% during the 3-d exposure to 15 °C, indicating that the minimum winter temperature in Kochi is often lethal to these species. Thus, the present study highlights the species-specific cold thermal tolerance of tropical reef fishes to winter conditions at the leading edge as a useful predictor of a given specie’s range-shift capacity.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Beitinger TL, McCauley, (1990) Whole-animal physiological processes for the assessment of stress in fishes. J Great Lakes Res 16(4):542–575. https://doi.org/10.1016/S0380-1330(90)71445-1
Beitinger TL, Bennett WA, McCauley RW (2000) Temperature tolerances of North American freshwater fishes exposed to dynamic changes in temperature. Environ Biol Fish 58(3):237–275. https://doi.org/10.1023/a:1007676325825
Bennett WA, Beitinger TL (1997) Temperature tolerance of sheepshead minnow, Cyprinodon variegatus. Copeia 1:77–87. https://doi.org/10.2307/1447842
Bennett WA, Currie RJ, Wagner PF, Beitinger TL (1997) Cold tolerance and potential overwintering of the red-bellied piranha Pygocentrus nattereri in the United States. Trans Am Fish Soc 126(5):841–849. https://doi.org/10.1577/1548-8659(1997)126%3c0841:CTAPOO%3e2.3.CO;2
Black EC (1955) Blood levels of hemoglobin and lactic acid in some freshwater fishes following exercise. J Fish Res Bd Can 12(6):917–929. https://doi.org/10.1139/f55-048
Booth DJ, Figueira WF, Gregson MA, Brown L, Beretta G (2007) Occurrence of tropical fishes in temperate southeastern Australia: role of the East Australian Current. Estuar Coast Shelf Sci 72:102–114. https://doi.org/10.1016/j.ecss.2006.10.003
Booth DJ, Bond N, Macreadie P (2011) Detecting range shifts among Australian fishes in response to climate change. Mar Freshw Res 62(9):1027–1042. https://doi.org/10.1071/mf10270
Booth DJ, Feary D, Kobayashi D, Luiz O, Nakamura Y (2018) Tropical marine fishes and fisheries and climate change. In: Phillips BF, Perez-Ramirez M (eds) Climate change impacts on fisheries and aquaculture: a global analysis, vol II. Wiley, West Sussex, pp 875–896. https://doi.org/10.1002/9781119154051.ch26
Cheung WWL, Lam VWY, Sarmiento JL, Kearney K, Watson R, Pauly D (2009) Projecting global marine biodiversity impacts under climate change scenarios. Fish Fish 10:235–251. https://doi.org/10.1111/j.1467-2979.2008.00315.x
Cunjak RA (1988) Physiological consequences of overwintering in streams: the cost of acclimatization? Can J Fish Aquat Sci 45(3):443–452. https://doi.org/10.1139/f88-053
Djurichkovic LD, Donelson JM, Fowler AM, Feary DA, Booth DJ (2019) The effects of water temperature on the juvenile performance of two tropical damselfishes expatriating to temperate reefs. Sci Rep 9:13937. https://doi.org/10.1038/s41598-019-50303-z
Easterling DR, Meehl GA, Parmesan C, Changnon SA, Karl TR, Mearns LO (2000) Climate extremes: observations, modeling, and impacts. Science 289:2068–2074. https://doi.org/10.1126/science.289.5487.2068
Elliot JM (1981) Some aspects of thermal stress in freshwater teleosts. In: Pickering AD (ed) Stress and fish. Academic Press, New York, pp 209–245
Elliot JM (1991) Tolerance and resistance to thermal stress in juvenile Atlantic salmon, Salmo salar. Freshwat Biol 25:61–70. https://doi.org/10.1111/j.1365-2427.1991.tb00473.x
Emme J, Bennett WA (2008) Low temperature as a limiting factor for introduction and distribution of Indo-Pacific damselfishes in the eastern United States. J Therm Biol 33:62–66. https://doi.org/10.1016/j.jtherbio.2007.10.003
Emme J, Bennett WA (2009) Critical thermal tolerance polygons of tropical marine fishes. J Therm Biol 34:220–225. https://doi.org/10.1016/j.jtherbio.2009.02.005
Factors influencing thermal tolerances of individual organisms. In: Esch GW, McFarlane RM (eds) Thermal Ecology, vol. II, ERDA Symp Ser CONF-750425 Natl Inf Serv, Springfield, Va, pp 10–26
Feary DA, Pratchett MS, Emslie MJ, Fowler AM, Figueira WF, Luiz OJ, Nakamura Y, Booth DJ (2014) Latitudinal shifts in coral reef fishes: why some species do and others don’t shift. Fish Fish 15:593–615. https://doi.org/10.1111/faf.12036
Figueira WF, Booth DJ (2010) Increasing ocean temperature allow tropical fishes to survive overwinter in temperate waters. Glob Change Biol 16:506–516. https://doi.org/10.1111/j.1365-2486.2009.01934.x
Figueira WF, Biro PA, Booth DJ, Valenzuela VC (2009) Performance of tropical fishes recruiting into temperate habitats: role of ambient temperature and implications for climate change. Mar Ecol Prog Ser 384:231–239. https://doi.org/10.3354/meps08057
Finch C, Pike M, Witten M (1990) Slow mortality rate accelerations during aging in some animals approximate that of humans. Science 249:902. https://doi.org/10.1126/science.2392680
Fry FEJ (1947) Effects of the environment on animal activity. Univ. Toronto Studies in Biol., Series No. 55, Publ Ont Fish Res Lab 68:1–62.
Fry FEJ (1971) The effect of environmental factors on the physiology of fish. In: Hoar WS, Randall DJ (eds) Fish physiology, environmental relations and behavior, vol 6. Academic Press, New York, pp 1–98
Girden ER (1992) ANOVA: repeated measures. Sage University Paper Series on quantitative applications in the social sciences, 07-084. Newbury Park, CA: Sage.
Guderley (2004) Metabolic responses to low temperature in fish muscle. Biol Rev 79(2):409–427. https://doi.org/10.1017/s1464793103006328
Guderley H, Houle-Leroy P, Gagne A (2001) Thermal acclimation, growth and burst swimming of threespine stickleback: enzymatic correlates and influence of photoperiod. Physiol Biochem Zool 74:66–74. https://doi.org/10.1086/319313
Hammer O, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis, version 3.08 (8/2015). Paleontol Electron 4(1):9. https://palaeo-electronica.org/2001_1/past/issue1_01.htm
Hirata T, Oguri S, Hirata S, Fukami H, Nakamura Y, Yamaoka K (2011) Seasonal changes in fish assemblages in an area of hermatypic corals in Yokonami, Tosa Bay, Japan. Jpn J Ichthyol 58:9–64
Hoar WS (1956) Photoperiodism and thermal resistance of goldfish. Nature 178:4529. https://doi.org/10.1038/178364a0
Hongo C, Yamano H (2013) Species-specific responses of corals to bleaching events on anthropogenically turbid reefs on Okinawa Island, Japan, over a 15-year Period (1995–2009). PLoS ONE 8(4):e60952. https://doi.org/10.1371/journal.pone.0060952
Hurst TP (2007) Causes and consequences of winter mortality in fishes. J Fish Biol 71:315–345. https://doi.org/10.1111/j.1095-8649.2007.01596.x
Hurst TP, Conover DO (2002) Effects of temperature and salinity on survival of young-of-the-year Hudson River striped bass (Morone saxatilis): implications for optimal overwintering habitat. Can J Fish Aquat Sci 59(5):787–795. https://doi.org/10.1139/f02-051
IBM Corp (2015) IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Corp.
Johansen JL, Steffensen JF, Jones GP (2015) Winter temperatures decrease swimming performance and limit distributions of tropical damselfishes. Conserv Physiol 3:39. https://doi.org/10.1093/conphys/cov039
Johnson TP, Bennett AF (1995) The thermal acclimation of burst escape performance in fish: an integrated study of molecular and cellular physiology and organismal performance. J Exp Biol 198:2165–2175
Johnson TB, Evans DO (1996) Temperature constraints on overwinter survival of age-0 white perch. Trans Am Fish Soc 125:466–471. https://doi.org/10.1577/1548-8659(1996)125%3c0466:ntcoos%3e2.3.co;2
Johnston IA, Dunn J (1987) Temperature acclimation and metabolism in ectotherms with particular reference to teleost fish. Symp Soc Exp Biol 41:67–93
Kimball ME, Miller JM, Whitfield PE, Hare JA (2004) Thermal tolerance and potential distribution of invasive lionfish (Pterois volitans/miles complex) on the east coast of the United States. Mar Ecol Prog Ser 283:269–278. https://doi.org/10.3354/meps283269
Kingsbury KM, Gillanders BM, Booth DJ, Coni EOC, Nagelkerken I (2019) Range-extending coral reef fishes trade-off growth for maintenance of body condition in cooler waters. Sci Total Environ 703:134598. https://doi.org/10.1016/j.scitotenv.2019.134598
Kleinbaum DG, Klein M (2012) Survival analysis: a self-learning text (3rd edn). Statistics for biology and health. Springer, New York, pp 201–240
Last PR, White WT, Gledhill DC, Hobday AJ, Brown R, Edgar GJ, Pecl G (2011) Long-term shifts in abundance and distribution of a temperate fish fauna: a response to climate change and fishing practices. Glob Ecol Biogeogr 20:58–72. https://doi.org/10.1111/j.1466-8238.2010.00575.x
Leriorato JC, Nakamura Y (2019) Unpredictable extreme cold events: a threat to range-shifting tropical reef fishes in temperate waters. Mar Biol 166:110. https://doi.org/10.1007/s00227-019-3557-6
McBride RS, Able KW (1998) Ecology and fate of butterflyfishes, Chaetodon spp, in the temperate, western North Atlantic. Bull Mar Sci 63(2):401–416
Moles A, Korn S, Rice S (1997) Effects of low temperatures and starvation on resistance to stress in presmolt coho salmon. In: Reynolds J (ed) Fish Ecology in Arctic North America, AFS Symp 19, Bethesda, MD, pp 148‒154
Munday PL, Jones GP, Pratchett MS, Williams AJ (2008) Climate change and the future for coral reef fishes. Fish Fish 9:261–285. https://doi.org/10.1111/j.1467-2979.2008.00281.x
Nakamura Y, Feary DA, Kanda M, Yamaoka K (2013) Tropical fishes dominate temperate reef fish communities within western Japan. PLoS ONE 8:e81107. https://doi.org/10.1371/journal.pone.0081107
Nielsen HM, Ødegård J, Olesen I, Gjerde B, Ardo L, Jeney G, Jeney Z (2010) Genetic analysis of common carp (Cyprinus carpio) strains I: genetic parameters and heterosis for growth traits and survival. Aquaculture 304:14–21. https://doi.org/10.1016/j.aquaculture.2010.03.016
Nilsson GE, Crawley N, Lunde IG, Munday PL (2009) Elevated temperature reduces the respiratory scope of coral reef fishes. Glob Change Biol 15:1405–1412. https://doi.org/10.1111/j.1365-2486.2008.01767.x
Nitani H (1972) Beginning of the Kuroshio. In: Stommel H, Yoshida K (eds) Kuroshio–its physical aspects. University of Tokyo Press, Tokyo, pp 129–163
Osborne J (2010) Improving your data transformations: applying the Box-Cox transformation. Pract Assess Res Eval 15(12):1–9. https://doi.org/10.7275/qbpc-gk17
Pearce A, Hutchins B, Hoschke A, Fearns P (2016) Record high damselfish recruitment at Rottnest Island, Western Australia, and the potential for climate-induced range extension. Reg Stud Mar Sci 8:77–88. https://doi.org/10.1016/j.rsma.2016.09.009
Pike TW, Samanta M, Lindström J, Royle NJ (2008) Behavioural phenotype affects social interactions in an animal network. Proc R Soc B 275:2515–2520. https://doi.org/10.1098/rspb.2008.0744
Pitchaimani M, Eakin T (2008) Unique estimation of Gompertz parameters with mortality deceleration rate. Math Comput Model 47:104–114. https://doi.org/10.1016/j.mcm.2007.02.005
Pratchett MS, Munday PL, Wilson S, Graham NAJ, Cinner JE, Bellwood DR, Jones GP, Polunin NVC, McClanahan TR (2008) Effects of climate-induced coral bleaching on coral-reef fishes-ecological and economic consequences. Oceanogr Mar Biol Annu Rev 46:251–296. https://doi.org/10.1201/9781420065756.ch6
Prchal M, Kause A, Vandeputte M, Gela D, Allamellou JM, Kumar G, Bestin A, Bugeon J, Zhao J, Kocour M (2018) The genetics of overwintering performance in two-year old common carp and its relation to performance until market size. PLoS ONE 13:e0191624. https://doi.org/10.1371/journal.pone.0191624
Réale D, Reader SM, Sol D, McDougall PT, Dingemanse NJ (2007) Integrating animal temperament within ecology and evolution. Biol Rev 82:291–318. https://doi.org/10.1111/j.1469-185X.2007.00010.x
Rummer JL, Couturier CS, Stecyk JAW, Gardiner NM, Kinch JP, Nilsson GE, Munday PL (2014) Life on the edge: thermal optima for aerobic scope of equatorial reef fishes are close to current day temperatures. Glob Change Biol 20:1055–1066. https://doi.org/10.1111/gcb.12455
Sakai K, Singh T, Iguchi A (2019) Bleaching and post-bleaching mortality of Acropora corals on a heat-susceptible reef in 2016. PeerJ 7:e8138. https://doi.org/10.7717/peerj.8138
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682. https://doi.org/10.1038/nmeth.2019 (PMID 22743772)
Schultz ET, Conover DO (1999) The allometry of energy reserve depletion: test of a mechanism for size-dependent winter mortality. Oecologia 119:474–483. https://doi.org/10.1007/s004420050810
Seatemperature.net (2020) Sea temperature in Manila, Philippines. World Sea temperature and water surface temperature. Available: https://seatemperature.net/current/philippines/manila-sea-temperature Accessed Oct 2020
Sidell BD, Moerland TS (1989) Effects of temperature on muscular function and locomotory performance in teleost fish. Adv Comp Environ Physiol 5:115–156. https://doi.org/10.1007/978-3-642-74510-2_5
Smit H, Amelink-Koutstaak JM, Vijverberg J, Von Vaupel-Klein JC (1971) Oxygen consumption and efficiency of swimming goldfish. Comp Biochem Physiol 39:1–28. https://doi.org/10.1016/0300-9629(71)90343-4
Soeparno, Nakamura Y, Shibuno T, Yamaoka K (2012) Relationship between pelagic larval duration and abundance of tropical fishes on temperate coasts of Japan. J Fish Biol 80:346–357. https://doi.org/10.1111/j.1095-8649.2011.03175.x
Stevens GC (1989) The latitudinal gradient in geographical range: how so many species coexist in the tropics. Am Nat 133:240–256. https://doi.org/10.1086/284913
Su JL, Guan BX, Jiang JZ (1990) The Kuroshio. Part I. Physical features. Oceonogr Mar Biol Annu Rev 28:11–71
Tabachnick BG, Fidell LS (2013) Using multivariate statistics (6th ed). Pearson, Upper Saddle River, NJ
Tanaka K, Taino S, Haraguchi H, Prendergast G, Hiraoka M (2012) Warming off southwestern Japan linked to distributional shifts of subtidal canopy-forming seaweeds. Ecol Evol 2:2854–2865. https://doi.org/10.1002/ece3.391
Templeton GF (2011) A two-step approach for transforming continuous variables to normal: implications and recommendations for IS Research. Commun Assoc Inf Syst 28(4):41–58
Tose K, Hirata T, Kotera Y, Kanda M, Nakamura Y (2017) Occurrence and reproduction of tropical fishes in ocean warming hotspots of Japanese temperate reefs. Environ Biol Fish 100:617–630. https://doi.org/10.1007/s10641-017-0590-5
Uchiyama Y, Odani S, Kashima M, Kamidaira Y, Mitarai S (2018) Influences of the Kuroshio on interisland remote connectivity of corals across the Nansei Archipelago in the East China Sea. J Geophys Res Oceans 123(12):9245–9265. https://doi.org/10.1029/2018JC014017
Vehviläinen H, Kause A, Quinton C, Koskinen H, Paananen T (2008) Survival of the currently fittest: genetics of rainbow trout survival across time and space. Genetics 180:507–516. https://doi.org/10.1534/genetics.108.089896
Vergés A, Steinberg PD, Hay ME, Poore AG, Campbell AH, Ballesteros E, Heck KL Jr, Booth DJ, Coleman MA, Feary DA, Figueira W, Langlois T, Marzinelli EM, Mizerek T, Mumby PJ, Nakamura Y, Roughan M, van Sebille E, Gupta AS, Smale DA, Tomas F, Wernberg T, Wilson SK (2014) The tropicalization of temperate marine ecosystems: climate-mediated changes in herbivory and community phase shifts. Proc Royal Soc B 281:20140846. https://doi.org/10.1098/rspb.2014.0846
Wainwright PC, Bellwood DR (2002) Ecomorphology of feeding in coral reef fishes. In: Sale PS (ed) Coral reef fishes: dynamic and diversity in a complex ecosystem. Elsevier, San Diego, pp 33–55
Ward AJW, Webster MM, Hart PJB (2006) Intraspecific food competition in fishes. Fish Fish 7:231–261. https://doi.org/10.1111/j.1467-2979.2006.00224.x
Wellington GM, Victor BC (1989) Planktonic larval duration of one hundred species of Pacific and Atlantic damselfishes (Pomacentridae). Mar Biol 101:557–567. https://doi.org/10.1007/bf00541659
Wernberg T, Smale DA, Tuya F, Thomsen MS, Langlois TJ, de Bettignies T, Bennett S, Rousseaux CS (2013) An extreme climatic event alters marine ecosystem structure in a global biodiversity hotspot. Nat Clim Change 3:78–82. https://doi.org/10.1038/nclimate1627
Wernberg T, Bennett S, Babcock RC, de Bettignies T, Cure K, Depczynski M, Dufois F, Fromont J, Fulton CJ, Hovey RK, Harvey ES, Holmes TH, Kendrick GA, Radford B, Santana-Garcon J, Saunders BJ, Smale DA, Thomsen MS, Tuckett CA, Tuya F, Vanderklift MA, Wilson S (2016) Climate-driven regime shift of a temperate marine ecosystem. Science 353:169–172. https://doi.org/10.1126/science.aad8745
Wilson D, McCormick M (1999) Microstructure of settlement-marks in the otoliths of tropical reef fishes. Mar Biol 134:29–41. https://doi.org/10.1007/s002270050522
Yamano H, Sugihara K, Nomura K (2011) Rapid poleward range expansion of tropical reef corals in response to rising sea surface temperatures. Geophys Res Lett 38:L04061. https://doi.org/10.1029/2010GL046474
Yara Y, Oshima K, Fujii M, Yamano H, Yamanaka Y, Okada N (2011) Projection and uncertainty of the poleward range expansion of coral habitats in response to sea surface temperature warming: a multiple climate model study. Galaxea JCRS 13:1–11. https://doi.org/10.3755/galaxea.13.11
Acknowledgements
We are grateful to the Mindanao State University at Naawan (MSUN) for supporting the logistics during the field-based fish surveys in Mindanao and to Eugene Moleño, Darwin Baslot, and Daryl Baslot of MSUN for their kind assistance. This study was implemented under the auspices of the research collaboration between MSUN and Kochi University. We thank the anonymous reviewers for their helpful comments and Enago (www.enago.jp) for the English language review.
Funding
This study was supported by a grant from the Japan Society for the Promotion of Science (KAKENHI 15K07529) through the Climate Change Impacts Project.
Author information
Authors and Affiliations
Contributions
JCL and YN designed the study. JCL and YN performed the laboratory experiment, and JCL analyzed the data. JCL, YN, and WHU conducted the field survey. JCL wrote the paper with the help of YN and WHU. All the authors have approved the contents of this paper and have agreed to its submission to Marine Biology.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Ethical approval
Although the Animal Experiment Committee of the Kochi University ruled that no ethical clearance or approval of research ethics was required for this study because experimental work was conducted with an unregulated fish species, we followed all applicable institutional and/or national guidelines for the care and use of animals in this study. A minimum number of fish were used to test the hypothesis. After the experiment, fish were kept in home aquariums where they demonstrated normal conditions (e.g., unstressed body color and actively swimming).
Additional information
Responsible Editor: S. Hamilton.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Reviewers: undisclosed experts.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Leriorato, J.C., Nakamura, Y. & Uy, W.H. Cold thermal tolerance as a range-shift predictive trait: an essential link in the disparity of occurrence of tropical reef fishes in temperate waters. Mar Biol 168, 93 (2021). https://doi.org/10.1007/s00227-021-03875-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-021-03875-2