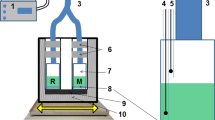
Abstract
Aerobic respiration is a biological energy generation process that consumes organic carbon and oxygen. In the ocean, the balance between photosynthesis and respiration is recognized as critical to understanding the ocean’s impact on the hydrospheric and atmospheric CO2. Techniques to determine respiration can be based on inorganic chemistry, electrochemistry, photochemistry, and enzymology. Here, for method comparison, physiological respiration was simultaneously measured by the Winkler method (W), O2 electrodes (E), and O2 optodes (O). These techniques detected respiratory O2 consumption (R), in situ, in dark incubation chambers. Respiratory electron transport system activity measurements detected potential respiration (Ф), biochemically. Leptomysis lingvura, a marine mysid, and Ulva rigida, a species of green algal sea lettuce, were the two organisms tested. Physiological respiration results from each technique were not statistically significantly different (multiple paired Student’s t tests, p value > 0.05) and were inside the range of similar published measurements. The mean dry-mass-specific respiration in L. lingvura and U. rigida was 0.147 ± 0.037 and 0.023 ± 0.008 µmol O2 h−1 (mg dry mass)−1, n = 9, respectively. The R-to-Ф ratios were different in the two organisms. However, linear regression between R and Ф for L. lingvura and U. rigida was stronger (r 2 = 0.814 and 0.313) than the linear regression between R and dry biomass (r 2 = 0.643 and 0.213). The application of Passing–Bablok regression analysis evidenced the high correlation between the results, and the Bland–Altman analysis examined the average difference (“bias”) and limits of agreement between the methods.




Similar content being viewed by others
References
Afonso J, Sansón M (1999) Algas, hongos y fanerógamas marinas de las Islas Canarias. Clave Analítica. Materiales didácticos universitarios. Serie Biología 2. Secretario de Publicaciones Universidad de La Laguna
Agardh CA (1823) Species algarum. Lund, Sweden
Aguiar-González B, Packard TT, Berdalet E, Roy S, Gómez M (2012) Respiration predicted from an enzyme kinetic model and the metabolic theory of ecology in two species of marine bacteria. J Exp Mar Biol Ecol 412:1–12. https://doi.org/10.1016/j.jembe.2011.09.018
Arístegui J, Harrison WG (2002) Decoupling of primary production and community respiration in the ocean: implications for regional carbon studies. Aquat Microb Ecol 29(2):199–209. https://doi.org/10.3354/ame029199
Atwood DK, Kinard WF, Barcelona MJ, Johnson EC (1977) Comparison of polarographic electrode and Winkler titration determinations of dissolved oxygen in oceanographic samples. Deep Sea Res 24(3):311–313. https://doi.org/10.1016/0146-6291(77)90000-5
Båmstedt U (1980) ETS activity as an estimator of respiratory rate of zooplankton populations. The significance of variations in environmental factors. J Exp Mar Biol Ecol 42(3):267–283. https://doi.org/10.1016/0022-0981(80)90181-1
Beardall J, Quigg A, Raven JA (2003) Oxygen consumption: photorespiration and chlororespiration. In: Larkum A, Douglas S, Raven JA (eds) Photosynthesis in algae. Advances in photosynthesis and respiration, vol 14. Springer, Dordrecht, pp 157–181
Bland JM, Altman D (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 327(8476):307–310
Burrows PA, Sazanov LA, Svab Z, Maliga P, Nixon PJ (1998) Identification of a functional respiratory complex in chloroplasts through analysis of tobacco mutants containing disrupted plastid ndh genes. EMBO J 17(4):868–876. https://doi.org/10.1093/emdoj/17.4.868
Cabello-Pasini A, Figueroa FL (2005) Effect of nitrate concentration on the relationship between photosynthetic oxygen evolution and electron transport rate in Ulva rigida (chlorophyta). J Phycol 41(6):1169–1177. https://doi.org/10.1111/j.1529-8817.2005.00144.x
Cabello-Pasini A, Macías-Carranza V, Abdala R, Korbee N, Figueroa FL (2011) Effect of nitrate concentration and UVR on photosynthesis, respiration, nitrate reductase activity, and phenolic compounds in Ulva rigida (Chlorophyta). J Appl Phycol 23(3):363–369. https://doi.org/10.1007/s10811-010-9548-0
Carpenter JH (1965) The accuracy of the Winkler method for dissolved oxygen analysis. Limnol Oceanogr 10(1):135–140. https://doi.org/10.4319/lo.1965.10.1.0135
Cleland WW (1967) Enzyme kinetics. Ann Rev Biochem 36(1):77–112. https://doi.org/10.1146/annurev.bi.36.070167.000453
Conover RJ (1960) The feeding behavior and respiration of some marine planktonic Crustacea. Biol Bull 119(3):399–415. https://doi.org/10.2307/1539258
Curl H, Sandberg J (1961) The measurement of dehydrogenase activity in marine organisms. J Mar Res 19(3):123–138
Dewitte K, Fierens C, Stöckl D, Thienpont LM (2002) Application of the Bland–Altman plot for interpretation of method-comparison studies: a critical investigation of its practice. Clin Chem 48(5):799–801
Dortch Q, Packard TT (1989) Differences in biomass structure between oligotrophic and eutrophic marine ecosystems. Deep Sea Res 36(2):223–240. https://doi.org/10.1016/0198-0149(89)90135-0
Duarte CM, Regaudie-de-Gioux A, Arrieta JM, Delgado-Huertas A, Agustí S (2013) The oligotrophic ocean is heterotrophic. Annu Rev Mar Sci 5:551–569. https://doi.org/10.1146/annurev-marine-121211-172337
Ducklow HW, Doney SC (2013) What is the metabolic state of the oligotrophic ocean? A debate. Annu Rev Mar Sci 5:525–533. https://doi.org/10.1146/annurev-marine-121211-172331
Fernández-Urruzola I, Osma N, Packard TT, Gómez M, Postel L (2014) Distribution of zooplankton biomass and potential metabolic activities across the northern Benguela upwelling system. J Mar Syst 140(B):138–149. https://doi.org/10.1016/j.jmarsys.2014.05.009
Friedrich J, Längin A, Kümmerer K (2013) Comparison of an electrochemical and luminescence-based oxygen measuring system for use in the biodegradability testing according to closed bottle test (OECD 301D). Clean (Weinh) 41(3):251–257. https://doi.org/10.1002/clen.201100558
Gatti S, Brey T, Müller W, Heilmayer O, Holst G (2002) Oxygen microoptodes: a new tool for oxygen measurements in aquatic animal ecology. Mar Biol 140(6):1075–1085. https://doi.org/10.1007/s00227-002-0786-9
Giavarina D (2015) Understanding bland altman analysis. Biochem Med Biochem Med 25(2):141–151
Goedheer JC (1963) A cooperation of two pigment systems and respiration in photosynthetic luminescence. Biochim Biophys Acta 66:61–71. https://doi.org/10.1016/0006-3002(63)91167-3
Gómez M (2000) Manual de prácticas de zoología Marina. Departamento de Biología, Universidad de Las Palmas de Gran Canaria, Servicio de Publicaciones y Producción Documental, Gran Canaria
Gómez M, Torres S, Hernández-León S (1996) Modification of the electron transport system (ETS) method for routine measurements of respiratory rates of zooplankton. S Afr J Mar Sci 17(1):15–20. https://doi.org/10.2989/025776196784158446
Green DE (1963) The mitochondrion. Sci Am 210:63–74
Guedeney G, Corneille S, Cuiné S, Peltier G (1996) Evidence for an association of ndh B, ndh J gene products and ferredoxin-NADP-reductase as components of a chloroplastic NAD(P)H dehydrogenase complex. FEBS Lett 378(3):277–280. https://doi.org/10.1016/0014-5793(95)01473-X
Hernández-León S, Gómez M (1996) Factors affecting the respiration/ETS ratio in marine zooplankton. J Plankton Res 18(2):239–255. https://doi.org/10.1093/plankt/18.2.239
Hernández-León S, Ikeda T (2005) Zooplankton respiration. In: Del Giorgio PA, Williams PJ (eds) Respiration in aquatic systems. Oxford University Press, New York, pp 57–82
Herrera A, Gómez M, Molina L, Otero F, Packard T (2011a) Rearing techniques and nutritional quality of two mysids from Gran Canaria (Spain). Aquacul Res 42(5):677–683. https://doi.org/10.1111/j.1365-2109.2010.02786.x
Herrera A, Packard T, Santana A, Gómez M (2011b) Effect of starvation and feeding on respiratory metabolism in Leptomysis lingvura (GO Sars 1866). J Exp Mar Biol Ecol 409(1):154–159. https://doi.org/10.1016/j.jembe.2011.08.016
Herrera-Ulibarri A (2013) Identification, abundance and rearing of mysids of Gran Canaria: aplication to laboratory and oceanographic respiratory metabolism studies. Ph.D. dissertation, Universidad de Las Palmas de Gran Canaria, Canary Island, Spain
Ikeda T (2013) Synthesis towards a global-bathymetric model of metabolism and chemical composition of mysid crustaceans. J Exp Mar Biol Ecol 445:79–87. https://doi.org/10.1016/j.jembe.2013.04.001
Ikeda T, Torres JJ, Hernández-León S, Geiger SP (2000) Metabolism. In: Harris RP, Wiebe PH, Lenz J, Skjoldal HR, Huntley M (eds) Zooplankton methodology manual. Academic Press, London, pp 455–515
Kanwisher J (1959) Polarographic oxygen electrode. Limnol Oceanogr 4:210–217. https://doi.org/10.4319/lo.1959.4.2.0210
Kenner RA, Ahmed SI (1975a) Measurements of electron transport activities in marine phytoplankton. Mar Biol 33(2):119–127. https://doi.org/10.1007/BF00390716
Kenner RA, Ahmed SI (1975b) Correlation between oxygen utilization and electron transport activity in marine phytoplankton. Mar Biol 33(2):129–133. https://doi.org/10.1007/BF00390717
King FD, Packard TT (1975) Respiration and the activity of the respiratory electron transport system in marine zooplankton. Limnol Oceanogr 20(5):849–854. https://doi.org/10.4319/lo.1975.20.5.0849
Klimant I, Meyer V, Kühl M (1995) Fiber-optic oxygen microsensors, a new tool in aquatic biology. Limnol Oceanogr 40(6):1159–1165. https://doi.org/10.4319/lo.1995.40.6.1159
Kuntz M (2004) Plastid terminal oxidase and its biological significance. Planta 218(6):896–899. https://doi.org/10.1007/s00425-004-1217-6
Lilley MKS, Lombard F (2015) Respiration of fragile planktonic zooplankton: extending the possibilities with a single method. J Exp Mar Biol Ecol 471:226–231. https://doi.org/10.1016/j.jembe.2015.06.013
Linnet K (1999) Limitations of the paired t-test for evaluation of method comparison data. Clin Chem 45(2):314–315
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Lubbers DW, Opitz N (1985) US Patent No. RE31, 879. Washington, DC: US Patent and Trademark Office
Lussier SM, Kuhn A, Chammas MJ, Sewall J (1988) Techniques for the laboratory culture of Mysidopsis species (Crustacea: Mysidacea). Environ Toxicol Chem 7(12):969–977. https://doi.org/10.1002/etc.5620071203
Maldonado F, Packard TT, Gómez M (2012) Understanding tetrazolium reduction and the importance of substrates in measuring respiratory electron transport activity. J Exp Mar Biol Ecol 434:110–118. https://doi.org/10.1016/j.jembe.2012.08.010
Marshall SM, Nicholls AG, Orr AP (1935) On the biology of Calanus finmarchicus. Part VI. Oxygen consumption in relation to environmental conditions. J Mar Biol Assoc UK (New Series) 20(01):1–27. https://doi.org/10.1017/S0025315400009991
Martinez I, Gómez M, Packard T (2010) Potential respiration is a better respiratory predictor than biomass in young, Artemia salina. J Exp Mar Biol Ecol 390(2):78–83. https://doi.org/10.1016/j.jembe.2010.05.011
Mazej Z, Gaberscik A (1999) ETS-activity as a measure of vitality of different macrophyte species. Plant Physiol 39(3):181–186
McDonald JH (2009) Handbook of biological statistics, vol 2. Sparky House Publishing, Baltimore, pp 191–197
Meland K, Mees J, Porter M, Wittmann KJ (2015) Taxonomic review of the orders mysida and stygiomysida (Crustacea, Peracarida). PLoS One 10(4):1–28. https://doi.org/10.1371/journal.pone.0124656
Møller IM (2001) Plant mitochondria and oxidative stress: electron transport, NADPH turnover, and metabolism of reactive oxygen species. Annu Rev Plant Biol 52(1):561–591. https://doi.org/10.1146/annurev.arplant.52.1.56
Nachlas MM, Margulies SI, Seligman AM (1960) A colorimetric method for the estimation of succinic dehydrogenase activity. J Biol Chem 235(2):499–503
Nelson D, Cox MM (2005) Lehninger principles of biochemistry. WH Freeman and Company, New York
Omori M, Ikeda T (1984) Methods in marine zooplankton ecology. xiii, 332 pp. John Wiley. J Mar Biol Assoc UK 65(2):562–562. https://doi.org/10.1017/S0025315400050669
Opitz N (1986) O2 Optodes for analyzing micro blood samples using thin sensor layers with small O2 capacities and special reflection properties for optical decoupling of sensor and sample. In: Longmuir IS (ed) Oxygen transport to tissue VIII. Advances in experimental medicine and biology, vol 200. Springer, Boston, MA
Osma N, Aristizabal M, Fernández-Urruzola I, Packard TT, Gómez M (2016a) Influence of starvation on respiratory metabolism and pyridine nucleotide levels in the marine dinoflagellate Oxyrrhis marina. Protist 167(2):136–147. https://doi.org/10.1016/j.protis.2016.01.002
Osma N, Fernández-Urruzola I, Gómez M, Montesdeoca-Esponda S, Packard TT (2016b) Predicting in vivo oxygen consumption rate from ETS activity and bisubstrate enzyme kinetics in cultured marine zooplankton. Mar Biol 163(7):1–14. https://doi.org/10.1007/s00227-016-2923-x
Osma N, Maldonado F, Fernández-Urruzola I, Packard TT, Gómez M (2016c) Variability of respiration and pyridine nucleotides concentration in oceanic zooplankton. J Plankton Res 38(3):537–550. https://doi.org/10.1093/plankt/fbw001
Owens TG, King FD (1975) The measurement of respiratory electron-transport-system activity in marine zooplankton. Mar Biol 30(1):27–36. https://doi.org/10.1007/BF00393750
Packard TT (1971) The measurement of respiratory electron transport activity in marine phytoplankton. J Mar Res 29(3):235–244. https://doi.org/10.1007/BF00393750
Packard TT (1985a) Measurement of electron transport activity of marine microplankton. In: Williams PJB, Jannasch HW (eds) Advances in aquatic microbiology. Academic Press, New York, pp 207–261
Packard TT (1985b) Oxygen consumption in the ocean: measuring and mapping with enzyme analysis. In: Zirino A (ed) Mapping strategies in chemical oceanography. American Chemical Society, Washington, D.C., pp 177–209
Packard TT, Christensen JP (2004) Respiration and vertical carbon flux in the Gulf of Maine water column. J Mar Res 62(1):93–115. https://doi.org/10.1357/00222400460744636
Packard TT, Codispoti LA (2007) Respiration, mineralization, and biochemical properties of the particulate matter in the southern Nansen Basin water column in April 1981. Deep-Sea Res Pt I 54(3):403–414
Packard TT, Gómez M (2008) Exploring a first-principles-based model for zooplankton respiration. ICES J Mar Sci Journal du Conseil 65(3):371–378. https://doi.org/10.1093/icesjms/fsn003
Packard TT, Williams PJL (1981) Rates of respiratory oxygen-consumption and electron-transport in surface seawater from the northwest Atlantic. Oceanol Acta 4(3):351–358
Packard TT, Garfield PC, Codispoti LA (1983) Oxygen consumption and denitrification below the Peruvian upwelling. In: Suess E, Thiede J (eds) Coastal upwelling: its sediment record. Plenum Press, New York, pp 147–173
Packard TT, Berdalet E, Blasco D, Roy SO, St-Amand L, Lagacé B (1996) CO2 production predicted from isocitrate dehydrogenase activity and bisubstrate enzyme. Aquat Microb Ecol 11:11–19. https://doi.org/10.1007/s002270100550
Passing H, Bablok W (1983) A new biometrical procedure for testing the equality of measurements from two different analytical methods. Application of linear regression procedures for method comparison studies in clinical chemistry, part I. Clin Chem Lab Med 21(11):709–720
Peltier G, Cournac L (2002) Chlororespiration. Annu Rev Plant Biol 53(1):523–550. https://doi.org/10.1146/annurev.arplant.53.100301.135242
Peñuelas J, Murillo J, Azcón-Bieto J (1988) Actual and potential dark respiration rates and different electron transport pathways in freshwater aquatic plants. Aquat Bot 30(4):353–362. https://doi.org/10.1016/0304-3770(88)90067-8
Posten HO (1984) Robustness of the two-sample t-test. In: Rasch D, Tiku ML (eds) Robustness of statistical methods and nonparametric statistics. D. Reidel Publishing Company, Dordrecht, pp 92–99
Raven JA, Beardall J (2005) Respiration in aquatic photolithotrophs. In: Del Giorgio PA, Williams PJB (eds) Respiration in aquatic ecosystems. Oxford University Press, New York, pp 36–46
Rumeau D, Peltier G, Cournac L (2007) Chlororespiration and cyclic electron flow around PSI during photosynthesis and plant stress response. Plant Cell Environ 30(9):1041–1051
Rutter WJ (1967) Protein determination in embryos. In: Wilt FH, Wessells NK (eds) Methods in developmental biology. Thomas Y. Crowell Co., New York, pp 671–683
Sars GO (1866) II. Beretning om en i Sommeren 1865 foretagen zoologisk Reise ved Kysterne af Christianias og Christiansands Stifter. Nyt Magazin for Naturvidenskaberne. 15:84–128
Satpati GG, Pal R (2011) Biochemical composition and lipid characterization of marine green alga Ulva rigida—a nutritional approach. J Algal Biomass Utln 2(4):10–13
Savenkoff C, Packard TT, Rodier M, Gérinno M, Lefèvre D, Denis M (1995) Relative contribution of dehydrogenases to overall respiratory ETS activity in some marine organisms. J Plankton Res 17(8):1593–1604. https://doi.org/10.1093/plankt/17.8.1593
Serret P, Robinson C, Aranguren-Gassis M, García-Martín EE, Gist N, Kitidis V, Lozano J, Stephens J, Harris C, Thomas R (2015) Both respiration and photosynthesis determine the scaling of plankton metabolism in the oligotrophic ocean. Nat Commun 6:6961
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika 52(3/4):591–611. https://doi.org/10.1093/biomet/52.3-4.591
Smith JJ, McFeters GA (1997) Mechanisms of INT (2-(4-iodophenyl)-3-(4-nitrophenyl)-5-phenyl tetrazolium chloride), and CTC (5-cyano-2, 3-ditolyl tetrazolium chloride) reduction in Escherichia coli K-12. J Microbiol Methods 29(3):161–175. https://doi.org/10.1016/S0167-7012(97)00036-5
Warkentin M, Freese HM, Karsten U, Schumann R (2007) New and fast method to quantify respiration rates of bacterial and plankton communities in freshwater ecosystems by using optical oxygen sensor spots. Appl Environ Microbiol 73(21):6722–6729
Williams PJ, Del Giorgio PA (2005) Respiration in aquatic ecosystems: history and background. In: Del Giorgio PA, Williams PJ (eds) Respiration in aquatic ecosystems. Oxford University Press, Oxford, pp 1–17
Williams PJLB, Quay PD, Westberry TK, Behrenfeld MJ (2013) The oligotrophic ocean is autotrophic. Annu Rev Mar Sci 5:535–549. https://doi.org/10.1146/annurev-marine-121211-172335
Winkler L (1888) Methods for measurement of dissolved oxygen. Ber Dtsch Chem Ges 21:2843–2855
Acknowledgements
The authors are grateful to I. Martínez for her help with the protein determinations. We acknowledge the comments from the reviewers which have contributed greatly to the improvement of this article. This work was accomplished thanks to the Spanish Ministry of Economy and Competitiveness and the Spanish Ministry of Education, Culture and Sports. The research was completed while the senior author was a Ph.D. student in the Doctoral Programme in Oceanography and Global Change at the University of Las Palmas de Gran Canaria.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was funded by the BIOMBA Project (CTM2012-32729/MAR) Granted to M. Gómez by the Spanish Ministry of Economy and Competitiveness. D. R. Bondyale-Juez received financial support from the FPU Grants from the Spanish Ministry of Education, Culture and Sports. T. T. Packard was supported by TIAA-CREF (USA), Social Security (USA), and by the Canary Islands CEI: Tricontinental Atlantic Campus.
Conflict of interest
The authors have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Responsible Editor: H.-O. Pörtner.
Reviewed by P. Serret and an undisclosed expert.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bondyale-Juez, D.R., Packard, T.T., Viera-Rodríguez, M.A. et al. Respiration: comparison of the Winkler technique, O2 electrodes, O2 optodes and the respiratory electron transport system assay. Mar Biol 164, 226 (2017). https://doi.org/10.1007/s00227-017-3271-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-017-3271-1