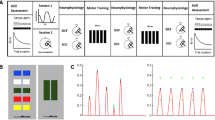
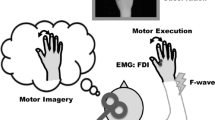
Abstract
Muscle synergy is important for simplifying functional movement, which constitutes spatiotemporal patterns of activity across muscles. To execute selective finger movements that are independent of synergistic movement patterns, we hypothesized that inhibitory neural activity is necessary to suppress enslaved finger movement caused by synergist muscles. To test this hypothesis, we focused on a pair of synergist muscles used in the hand opening movement, namely the index finger abductor and little finger abductor (abductor digiti minimi; ADM), and examined whether inhibitory neural activity in ADM occurs during selective index finger abduction/adduction movements and/or its imagery using transcranial magnetic stimulation and F-wave analysis. During the index finger adduction movement, background EMG activity, F-wave persistence, and motor evoked potential (MEP) amplitude in ADM were elevated. However, during the index finger abduction movement, ADM MEP amplitude remained unchanged despite increased background EMG activity and F-wave persistence. These results suggest that increased spinal excitability in ADM is counterbalanced by cortical-mediated inhibition only during selective index finger abduction movement. This assumption was further supported by the results of motor imagery experiments. Although F-wave persistence in ADM increased only during motor imagery of index finger abduction, ADM MEP amplitude during motor imagery of index finger abduction was significantly lower than that during adduction. Overall, our findings indicate that cortical-mediated inhibition contributes to the execution of selective finger movements that are independent of synergistic hand movement patterns.




Similar content being viewed by others
References
Aoyama T, Kaneko F, Ohashi Y, Kohno Y (2019) Dissociation between cortical and spinal excitability of the antagonist muscle during combined motor imagery and action observation. Sci Rep 9:13120
Beck S, Richardson SP, Shamim EA, Dang N, Schubert M, Hallett M (2008) Short intracortical and surround inhibition are selectively reduced during movement initiation in focal hand dystonia. J Neurosci 28:10363–10369
Capaday C, Devanne H, Bertrand L, Lavoie BA (1998) Intracortical connections between motor cortical zones controlling antagonistic muscles in the cat: a combined anatomical and physiological study. Exp Brain Res 120:223–232
Capaday C, Ethier C, Brizzi L, Sik A, van Vreeswijk C, Gingras D (2009) On the nature of the intrinsic connectivity of the cat motor cortex: evidence for a recurrent neural network topology. J Neurophysiol 102:2131–2141
Case LK, Pineda J, Ramachandran VS (2015) Common coding and dynamic interactions between observed, imagined, and experienced motor and somatosensory activity. Neuropsychologia 79:233–245
Cengiz B, Vuralli D, Zinnuroglu M et al (2018) Analysis of mirror neuron system activation during action observation alone and action observation with motor imagery tasks. Exp Brain Res 236:497–503
Chang WH, Fried PJ, Saxena S, Jannati A, Gomes-Osman J, Kim YH, Pascual-Leone A (2016) Optimal number of pulses as outcome measures of neuronavigated transcranial magnetic stimulation. Clin Neurophysiol 127:2892–2897
Connolly KJ, Forssberg H (1997) Neurophysiology and neuropsychology of motor development. Cambridge University Press, Cambridge
de Winter JC, Gosling SD, Potter J (2016) Comparing the Pearson and Spearman correlation coefficients across distributions and sample sizes: a tutorial using simulations and empirical data. Psychol Methods 21:273–290
Decety J, Grezes J (1999) Neural mechanisms subserving the perception of human actions. Trends Cogn Sci 3:172–178
Di Lazzaro V, Oliviero A, Saturno E et al (2001) The effect on corticospinal volleys of reversing the direction of current induced in the motor cortex by transcranial magnetic stimulation. Exp Brain Res 138:268–273
Eaves DL, Riach M, Holmes PS, Wright DJ (2016) Motor imagery during action observation: a brief review of evidence, theory and future research opportunities. Front Neurosci 10:514
Ehrsson HH, Kuhtz-Buschbeck JP, Forssberg H (2002) Brain regions controlling nonsynergistic versus synergistic movement of the digits: a functional magnetic resonance imaging study. J Neurosci 22:5074–5080
Ethier C, Brizzi L, Giguere D, Capaday C (2007) Corticospinal control of antagonistic muscles in the cat. Eur J Neurosci 26:1632–1641
Fadiga L, Buccino G, Craighero L, Fogassi L, Gallese V, Pavesi G (1999) Corticospinal excitability is specifically modulated by motor imagery: a magnetic stimulation study. Neuropsychologia 37:147–158
Goldsworthy MR, Hordacre B, Ridding MC (2016) Minimum number of trials required for within- and between-session reliability of TMS measures of corticospinal excitability. Neuroscience 320:205–209
Hager-Ross C, Schieber MH (2000) Quantifying the independence of human finger movements: comparisons of digits, hands, and movement frequencies. J Neurosci 20:8542–8550
Hasegaw Y, Kasai T, Kinoshita H, Yahagi S (2001) Modulation of a motor evoked response to transcranial magnetic stimulation by the activity level of the first dorsal interosseous muscle in humans when grasping a stationary object with different grip widths. Neurosci Lett 299:1–4
Jacobs KM, Donoghue JP (1991) Reshaping the cortical motor map by unmasking latent intracortical connections. Science 251:944–947
Jeannerod M (1995) Mental imagery in the motor context. Neuropsychologia 33:1419–1432
Kaneko K, Kawai S, Fuchigami Y, Morita H, Ofuji A (1996) The effect of current direction induced by transcranial magnetic stimulation on the corticospinal excitability in human brain. Electroencephalogr Clin Neurophysiol 101:478–482
Kaneko F, Murakami T, Onari K, Kurumadani H, Kawaguchi K (2003) Decreased cortical excitability during motor imagery after disuse of an upper limb in humans. Clin Neurophysiol 114:2397–2403
Kaneko N, Masugi Y, Yokoyama H, Nakazawa K (2018) Difference in phase modulation of corticospinal excitability during the observation of the action of walking, with and without motor imagery. NeuroReport 29:169–173
Kazennikov O, Solopova I, Talis V, Ioffe M (2008) Anticipatory postural adjustment: the role of motor cortex in the natural and learned bimanual unloading. Exp Brain Res 186:215–223
Malouin F, Richards CL, Jackson PL, Lafleur MF, Durand A, Doyon J (2007) The kinesthetic and visual imagery questionnaire (KVIQ) for assessing motor imagery in persons with physical disabilities: a reliability and construct validity study. J Neurol Phys Ther 31:20–29
McMorland AJ, Runnalls KD, Byblow WD (2015) A neuroanatomical framework for upper limb synergies after stroke. Front Hum Neurosci 9:82
Mouthon A, Ruffieux J, Walchli M, Keller M, Taube W (2015) Task-dependent changes of corticospinal excitability during observation and motor imagery of balance tasks. Neuroscience 303:535–543
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Park J, Xu D (2017) Multi-finger interaction and synergies in finger flexion and extension force production. Front Hum Neurosci 11:318
Reilly KT, Hammond GR (2000) Independence of force production by digits of the human hand. Neurosci Lett 290:53–56
Reilly KT, Schieber MH (2003) Incomplete functional subdivision of the human multitendoned finger muscle flexor digitorum profundus: an electromyographic study. J Neurophysiol 90:2560–2570
Rozand V, Lebon F, Papaxanthis C, Lepers R (2015) Effect of mental fatigue on speed-accuracy trade-off. Neuroscience 297:219–230
Sakamoto M, Muraoka T, Mizuguchi N, Kanosue K (2009) Combining observation and imagery of an action enhances human corticospinal excitability. Neurosci Res 65:23–27
Santello M, Baud-Bovy G, Jörntell H (2013) Neural bases of hand synergies. Front Comput Neurosci 7:23
Schieber MH (1991) Individuated finger movements of rhesus monkeys: a means of quantifying the independence of the digits. J Neurophysiol 65:1381–1391
Schieber MH, Hibbard LS (1993) How somatotopic is the motor cortex hand area? Science 261:489–492
Schneider C, Devanne H, Lavoie BA, Capaday C (2002) Neural mechanisms involved in the functional linking of motor cortical points. Exp Brain Res 146:86–94
Shinoda Y, Yokota J, Futami T (1981) Divergent projection of individual corticospinal axons to motoneurons of multiple muscles in the monkey. Neurosci Lett 23:7–12
Slobounov S, Johnston J, Chiang H, Ray WJ (2002) Motor-related cortical potentials accompanying enslaving effect in single versus combination of fingers force production tasks. Clin Neurophysiol 113:1444–1453
Sohn YH, Hallett M (2004) Surround inhibition in human motor system. Exp Brain Res 158:397–404
Stinear CM, Byblow WD (2003) Role of intracortical inhibition in selective hand muscle activation. J Neurophysiol 89:2014–2020
Stinear CM, Byblow WD (2004) Impaired modulation of intracortical inhibition in focal hand dystonia. Cereb Cortex 14:555–561
Suzuki T, Bunno Y, Onigata C, Tani M, Uragami S, Yoshida S (2014) Excitability of spinal neural function during motor imagery in Parkinson’s disease. Funct Neurol 29:263–267
Thirugnanasambandam N, Khera R, Wang H, Kukke SN, Hallett M (2015) Distinct interneuronal networks influence excitability of the surround during movement initiation. J Neurophysiol 114:1102–1108
Wright DJ, Williams J, Holmes PS (2014) Combined action observation and imagery facilitates corticospinal excitability. Front Hum Neurosci 8:951
Yahagi S, Shimura K, Kasai T (1996) An increase in cortical excitability with no change in spinal excitability during motor imagery. Percept Mot Skills 83:288–290
Yahagi S, Takeda Y, Ni Z et al (2005) Modulations of input-output properties of corticospinal tract neurons by repetitive dynamic index finger abductions. Exp Brain Res 161:255–264
Zatsiorsky VM, Li ZM, Latash ML (1998) Coordinated force production in multi-finger tasks: finger interaction and neural network modeling. Biol Cybern 79:139–150
Acknowledgements
This work was supported by JSPS KAKENHI [Grant #16K16433 (T.A.)].
Author information
Authors and Affiliations
Contributions
TA: contributed to the design of the study, data collection, data analysis, and manuscript preparation; FK: contributed to the design of the study, data analysis, and manuscript preparation; YO: contributed to data analysis and manuscript preparation; YK: contributed to data collection, data analysis, and manuscript preparation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no financial and non-financial competing interests.
Additional information
Communicated by Winston D Byblow.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aoyama, T., Kaneko, F., Ohashi, Y. et al. Neural mechanism of selective finger movement independent of synergistic movement. Exp Brain Res 237, 3485–3492 (2019). https://doi.org/10.1007/s00221-019-05693-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-019-05693-x