Abstract
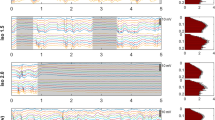
Volatile general anesthetics are used commonly in adults and children, yet their mechanisms of action are complex and the changes in single unit firing and synaptic activity that underlie the broad decreases in neuronal activity induced by these drugs have not been well characterized. Capturing such changes throughout the anesthesia process is important for comparing the effects of different anesthetics and gaining a better understanding of their mechanisms of action and their impact on different brain regions. Using chronically implanted electrodes in the rabbit somatosensory cortex, we compared the effects of two common general anesthetics, isoflurane, and sevoflurane, on cortical neurons. Single unit activity and local field potentials (LFP) were recorded continuously before and during anesthetic delivery at 1 MAC, as well as during recovery. Our findings show that although isoflurane and sevoflurane belong to the same class of volatile general anesthetics, their effects upon cortical single units and LFP were quite different. Overall, the suppression of neuronal firing was greater and more uniform under sevoflurane. Moreover, the changes in LFP frequency bands suggest that effect of anesthesia upon beta oscillations does not necessarily depend on the level of single unit activity, but rather on the changes in GABA/glutamate neurotransmission induced by each drug.



Similar content being viewed by others
References
Akeju O, Westover MB, Pavone KJ, Sampson AL, Hartnack KE, Brown EN, Purdon PL (2014) Effects of sevoflurane and propofol on frontal electroencephalogram power and coherence. Anesthesiology 121:990–998
Akrawi WP, Drummond JC, Kalkman CJ, Patel PM (1996) A comparison of the electrophysiologic characteristics of EEG burst-suppression as produced by isoflurane, thiopental, etomidate, and propofol. J Neurosurg Anesthesiol 8:40–46
Aksenov DP, Li L, Miller MJ, Iordanescu G, Wyrwicz AM (2015) Effects of anesthesia on BOLD signal and neuronal activity in the somatosensory cortex. J Cereb Blood Flow Metab 35:1819–1826
Alkire MT, Haier RJ, Fallon JH (2000) Toward a unified theory of narcosis: brain imaging evidence for a thalamocortical switch as the neurophysiologic basis of anesthetic-induced unconsciousness. Conscious Cogn 9(3):370–386
Anderson PM, Jones NC, O’Brien TJ, Pinault D (2017) The N-methyl d-aspartate glutamate receptor antagonist ketamine disrupts the functional state of the corticothalamic pathway. Cereb Cortex 27:3172–3185
Buzsaki G, Anastassiou CA, Koch C (2012) ‘The origin of extracellular fields and currents–EEG, ECoG, LFP and spikes. Nat Rev Neurosci 13:407–420
Chae YJ, Zhang J, Au P, Sabbadini M, Xie GX, Yost CS (2010) Discrete change in volatile anesthetic sensitivity in mice with inactivated tandem pore potassium ion channel TRESK. Anesthesiology 113:1326–1337
Christie SB, Miralles CP, De Blas AL (2002) GABAergic innervation organizes synaptic and extrasynaptic GABAA receptor clustering in cultured hippocampal neurons. J Neurosci 22:684–697
Constant I, Dubois MC, Piat V, Moutard ML, McCue M, Murat I (1999) Changes in electroencephalogram and autonomic cardiovascular activity during induction of anesthesia with sevoflurane compared with halothane in children. Anesthesiology 91:1604–1615
Douglas RJ, Martin KA (2004) Neuronal circuits of the neocortex. Annu Rev Neurosci 27:419–451
Drummond JC (1985) MAC for halothane, enflurane, and isoflurane in the New Zealand white rabbit: and a test for the validity of MAC determinations. Anesthesiology 62:336–338
Franks NP, Lieb WR (1988) Volatile general anaesthetics activate a novel neuronal K+ current. Nature 333:662–664
Freye E, Bruckner J, Latasch L (2004) No difference in electroencephalographic power spectra or sensory-evoked potentials in patients anaesthetized with desflurane or sevoflurane. Eur J Anaesthesiol 21:373–378
Hagihira S (2015) Changes in the electroencephalogram during anaesthesia and their physiological basis. Br J Anaesth 115(Suppl 1):i27–i31
Hartikainen K, Rorarius M, Makela K, Perakyla J, Varila E, Jantti V (1995a) Visually evoked bursts during isoflurane anaesthesia. Br J Anaesth 74:681–685
Hartikainen K, Rorarius M, Makela K, Yli-Hankala A, Jantti V (1995b) Propofol and isoflurane induced EEG burst suppression patterns in rabbits. Acta Anaesthesiol Scand 39:814–818
Herrmann CS, Struber D, Helfrich RF, Engel AK (2016) EEG oscillations: from correlation to causality. Int J Psychophysiol 103:12–21
Hight D, Voss LJ, Garcia PS, Sleigh J (2017) Changes in alpha frequency and power of the electroencephalogram during volatile-based general anesthesia. Front Syst Neurosci 11:36
Hudetz AG, Imas OA (2007) Burst activation of the cerebral cortex by flash stimuli during isoflurane anesthesia in rats. Anesthesiology 107:983–991
Ishizawa Y, Ahmed OJ, Patel SR, Gale JT, Sierra-Mercado D, Brown EN, Eskandar EN (2016) Dynamics of propofol-induced loss of consciousness across primate neocortex. J Neurosci 36:7718–7726
Kroeger D, Amzica F (2007) Hypersensitivity of the anesthesia-induced comatose brain. J Neurosci 27:10597–10607
Larsen M, Valo ET, Berg-Johnsen J, Langmoen IA (1998) Isoflurane reduces synaptic glutamate release without changing cytosolic free calcium in isolated nerve terminals. Eur J Anaesthesiol 15:224–229
Luethy A, Boghosian JD, Srikantha R, Cotten JF (2017) Halogenated ether, alcohol, and alkane anesthetics activate TASK-3 tandem pore potassium channels likely through a common mechanism. Mol Pharmacol 91:620–629
Nishikawa K, Harrison NL (2003) The actions of sevoflurane and desflurane on the gamma-aminobutyric acid receptor type A: effects of TM2 mutations in the alpha and beta subunits. Anesthesiology 99:678–684
Patel AJ, Honore E, Lesage F, Fink M, Romey G, Lazdunski M (1999) Inhalational anesthetics activate two-pore-domain background K+ channels. Nat Neurosci 2:422–426
Prabhakar NR, Semenza GL (2015) Oxygen sensing and homeostasis. Physiology (Bethesda) 30:340–348
Purdon PL, Sampson A, Pavone KJ, Brown EN (2015) Clinical Electroencephalography for anesthesiologists: Part I: background and basic signatures. Anesthesiology 123:937–960
Quiroga RQ, Nadasdy Z, Ben-Shaul Y (2004) Unsupervised spike detection and sorting with wavelets and superparamagnetic clustering. Neural Comput 16:1661–1687
Schwender D, Daunderer M, Klasing S, Finsterer U, Peter K (1998) Power spectral analysis of the electroencephalogram during increasing end-expiratory concentrations of isoflurane, desflurane and sevoflurane. Anaesthesia 53:335–342
Sheng M, Liu P, Mao D, Ge Y, Lu H (2017) The impact of hyperoxia on brain activity: a resting-state and task-evoked electroencephalography (EEG) study. PLoS One 12:e0176610
Shushruth S (2013) Exploring the neural basis of consciousness through anesthesia. J Neurosci 33:1757–1758
Sitdikova G, Zakharov A, Janackova S, Gerasimova E, Lebedeva J, Inacio AR, Zaynutdinova D, Minlebaev M, Holmes GL, Khazipov R (2014) Isoflurane suppresses early cortical activity. Ann Clin Transl Neurol 1:15–26
Stachnik J (2006) Inhaled anesthetic agents. Am J Health Syst Pharm 63:623–634
Swadlow HA (1989) Efferent neurons and suspected interneurons in S-1 vibrissa cortex of the awake rabbit: receptive fields and axonal properties. J Neurophysiol 62:288–308
van Lier H, Drinkenburg WH, van Eeten YJ, Coenen AM (2004) Effects of diazepam and zolpidem on EEG beta frequencies are behavior-specific in rats. Neuropharmacology 47:163–174
Vinje ML, Moe MC, Valo ET, Berg-Johnsen J (2002) The effect of sevoflurane on glutamate release and uptake in rat cerebrocortical presynaptic terminals. Acta Anaesthesiol Scand 46:103–108
Williams DC, Aleman MR, Brosnan RJ, Fletcher DJ, Holliday TA, Tharp B, Kass PH, Steffey EP, LeCouteur RA (2016) Electroencephalogram of healthy horses during inhaled anesthesia. J Vet Intern Med 30:304–308
Wyrwicz AM, Chen N-K, Li L, Weiss C, Disterhoft JF (2000) fMRI of visual system activation in the conscious rabbit. Magn Res Med 44:474–478
Yao C, Li Y, Shu S, Yao S, Lynch C, Bayliss DA, Chen X (2017) TASK channels contribute to neuroprotective action of inhalational anesthetics. Sci Rep 7:44203
Yin Y, Yan M, Zhu T (2012) Minimum alveolar concentration of sevoflurane in rabbits with liver fibrosis. Anesth Analg 114:561–565
Funding
This work was supported by the National Institute of General Medical Sciences (R01GM112715).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declared no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aksenov, D.P., Miller, M.J., Dixon, C.J. et al. The effect of sevoflurane and isoflurane anesthesia on single unit and local field potentials. Exp Brain Res 237, 1521–1529 (2019). https://doi.org/10.1007/s00221-019-05528-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-019-05528-9