Abstract
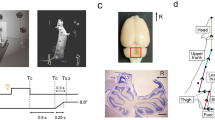
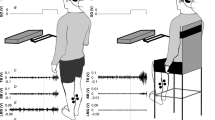
When an electrical stimulus is applied to perturb the vestibular system, a postural response is generated orthogonal to head orientation. It has previously been shown that there is a convergence of neck proprioceptive and vestibular input within the cerebellum to provide a head-on-body reference frame (Manzoni et al. in Neuroscience 93:1095–1107, 1999). The objective of this experiment was to determine whether the direction of the postural response to a vestibular perturbation is modulated when function of the cerebellar vermis is temporarily depressed. Twenty participants were randomly assigned to a SHAM group (paired-pulse transcranial magnetic stimulation) or a TEST group (continuous theta burst stimulation). Stochastic vestibular stimulation (SVS) was applied to standing subjects with their head facing forward or over their left shoulder. Cumulant density traces were established between the SVS and shear force over 180°, and the peak amplitude determined the direction of sway. There were no significant changes in sway direction when the head was facing forward for either stimulation (TEST or SHAM; p = 0.889) or when the head was facing over the shoulder for the SHAM condition (p = 0.954). There was, however, a significant change in sway direction when the head was turned with a depressed cerebellum (p = 0.018); from the expected antero-posterior direction, orthogonal to head orientation, to one slightly more mediolateral with respect to the feet. These results suggest the cerebellum plays a role in the integration of input to generate an appropriately directed postural response relative to the head position.



Similar content being viewed by others
References
Aimonetti J-M, Hospod V, Roll J-P, Ribot-Ciscar E (2007) Cutaneous afferents provide a neuronal population vector that encodes the orientation of human ankle movements. J Physiol 580:649–658
Angelaki DE, Yakusheva TA, Green AM et al (2010) Computation of egomotion in the macaque cerebellar vermis. Cerebellum 9:174–182
Apps R, Garwicz M (2005) Anatomical and physiological foundations of cerebellar information processing. Nat Rev Neurosci 6:297–311
Arasanz CP, Staines WR, Roy EA, Schweizer TA (2012) The cerebellum and its role in word generation: a cTBS study. Cortex 48:718–724
Bent LR, McFadyen BJ, French Merkley V et al (2000) Magnitude effects of galvanic vestibular stimulation on the trajectory of human gait. Neurosci Lett 279:157–160
Boyd CAR (2010) Cerebellar agenesis revisited. Brain 133:941–944
Black FO, Wall C, Nashner LM (1983) Effects of visual and support surface orientation references upon postral control in vestibular deficient subjects. Acta Otolaryngol 95:199–210
Blum PS, Gilman S (1979) Vestibular, somatosensory, and auditory input to the thalamus of the cat. Exp Neurol 65:343–354
Britton TC, Day BL, Brown P et al (1993) Postural electromyographic responses in the arm and leg following galvanic vestibular stimulation in man. Exp Brain Res 94:143–151
Cathers I, Day BL, Fitzpatrick RC (2005) Otolith and canal reflexes in human standing. J Physiol 563:229–234
Chadderton P, Schaefer AT, Williams SR, Margrie TW (2014) Sensory-evoked synaptic integration in cerebellar and cerebral cortical neurons. Nat Rev Neurosci 15:71–83
Chen R (2000) Studies of human motor physiology with transcranial magnetic stimulation. Muscle Nerve 9:S26–S32
Chen R (2004) Interactions between inhibitory and excitatory circuits in the human motor cortex. Exp Brain Res 154:1–10
Dakin CJ, Son GML, Inglis JT, Blouin J-S (2007) Frequency response of human vestibular reflexes characterized by stochastic stimuli. J Physiol 583:1117–1127
Day BL, Cole J (2002) Vestibular-evoked postural responses in the absence of somatosensory information. Brain 125:2081–2088
Demirtas-Tatlidede A, Freitas C, Cromer JR et al (2010) Safety and proof of principle study of cerebellar vermal theta burst stimulation in refractory schizophrenia. Schizophr Res 124:91–100
Denoth F, Magherini PC, Pompeiano O, Stanojević M (1979) Responses of purkinje cells of the cerebellar vermis to neck and macular vestibular inputs. Pflüg Arch Eur J Physiol 381:87–98
Edin BB, Vallbo AB (1990) Dynamic response of human muscle spindle afferents to stretch. J Neurophysiol 63:1297–1306
Fitzpatrick R, McCloskey DI (1994) Proprioceptive, visual and vestibular thresholds for the perception of sway during standing in humans. J Physiol 478:173–186
Fitzpatrick RC, Day BL (2004) Probing the human vestibular system with galvanic stimulation. J Appl Physiol 96:2301–2316
Grace Gaerlan M, Alpert PT, Cross C et al (2012) Postural balance in young adults: the role of visual, vestibular and somatosensory systems. J Am Acad Nurse Pract 24:375–381
Green AM, Angelaki DE (2010) Multisensory integration: resolving sensory ambiguities to build novel representations. Curr Opin Neurobiol 20:353–360
Gurfinkel VS, Popov KE, Smetanin BN, Shlykov VY (1989) Changes in the direction of vestibulomotor response in the course of adaptation to protracted static head turning in man. Neirofiziologiia 21(2):210–217
Huang Y-Z, Edwards MJ, Rounis E et al (2005) Theta burst stimulation of the human motor cortex. Neuron 45:201–206
Ito M (1984) The cerebellum and neural control. Raven Press, New York
Kammermeier S, Kleine JF, Eggert T et al (2013) Disturbed vestibular-neck interaction in cerebellar disease. J Neurol 260:794–804
Koch G, Mori F, Marconi B et al (2008) Changes in intracortical circuits of the human motor cortex following theta burst stimulation of the lateral cerebellum. Clin Neurophysiol 119:2559–2569
Lopez C, Blanke O (2011) The thalamocortical vestibular system in animals and humans. Brain Res Rev 67:119–146
Lund S, Broberg C (1983) Effects of different head positions on postural sway in man induced by a reproducible vestibular error signal. Acta Physiol Scand 117:307–309
Manzoni D, Pompeiano O, Bruschini L, Andre P (1999) Neck input modifies the reference frame for coding labyrinthine signals in the cerebellar vermis: a cellular analysis. Neuroscience 93:1095–1107
Marsden JF, Castellote J, Day BL (2002) Bipedal distribution of human vestibular-evoked postural responses during asymmetrical standing. J Physiol 542:323–331
Mian OS, Day BL (2009) Determining the direction of vestibular-evoked balance responses using stochastic vestibular stimulation. J Physiol 587:2869–2873
Mian OS, Day BL (2014) Violation of the craniocentricity principle for vestibularly evoked balance responses under conditions of anisotropic stability. J Neurosci 34:7696–7703
Nashner LM, Wolfson P (1974) Influence of head position and proprioceptive cues on short latency postural reflexes evoked by galvanic stimulation of the human labyrinth. Brain Res 67:255–268
Newton RA (1982) Joint receptor contributions to reflexive and kinesthetic responses. Phys Ther 62:22–29
Pettorossi VE, Schieppati M (2014) Neck proprioception shapes body orientation and perception of motion. Front Hum Neurosci 8:895
Popa T, Russo M, Meunier S (2010) Long-lasting inhibition of cerebellar output. Brain Stimul 3:161–169
Pozzo T, Berthoz A, Lefort L (1990) Head stabilization during various locomotor tasks in humans. I. Normal subjects. Exp Brain Res 82:97–106
Roy JE, Cullen KE (2001) Selective processing of vestibular reafference during self-generated head motion. J Neurosci 21:2131–2142
Sherman SM, Guillery RW (2002) The role of the thalamus in the flow of information to the cortex. Philos Trans R Soc Lond B Biol Sci 357:1695–1708
Silveri MC, Di Betta AM, Filippini V et al (1998) Verbal short-term store-rehearsal system and the cerebellum. Evidence from a patient with a right cerebellar lesion. Brain 121:2175–2187
Stiles L, Smith PF (2015) The vestibular-basal ganglia connection: balancing motor control. Brain Res 1597:180–188. doi:10.1016/j.brainres
Ugawa Y, Rothwell JC, Day BL, Thompson PD, Marsden CD (1991) Percutaneous electrical stimulation of corticospinal pathways at the level of the pyramidal decussation in humans. Ann Neurol 29:418–427
Wiestler T, McGonigle DJ, Diedrichsen J (2011) Integration of sensory and motor representations of single fingers in the human cerebellum. J Neurophysiol 105:3042–3053
Yakusheva TA, Shaikh AG, Green AM et al (2007) Purkinje cells in posterior cerebellar vermis encode motion in an inertial reference frame. Neuron 54:973–985
Acknowledgments
This work was supported by funding from the Natural Science and Engineering Research Council of Canada Discovery Grant (NSERC DG to L. R. B). The authors would also like to thank Gagan Gill for help with data collection and recruitment and Dr. Ryan Peters for his technical support and guidance on data analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lam, C.K., Tokuno, C.D., Staines, W.R. et al. The direction of the postural response to a vestibular perturbation is mediated by the cerebellar vermis. Exp Brain Res 234, 3689–3697 (2016). https://doi.org/10.1007/s00221-016-4766-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-016-4766-6