Abstract
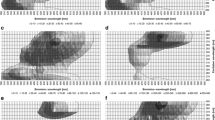
Fluorochromes can impair analysis results or, due to their natural frequency (molecular vibrations) completely superimpose important signals and make measurements impossible. This is especially true for non-invasive, optical measuring methods such as Raman microspectroscopy, which reach their limits when fluorochromes are present. Beer contains many fluorescent substances, e.g., amino acids, vitamins and phenolic compounds. Therefore, in this study, eight different beers (lager beer, dark lager, Pilsner beer, Radler beer, wheat beer, dark wheat beer, alcohol-free lager beer, and alcohol-free wheat beer) were examined for their fluorescent ingredients. Using the PARAFAC model, the fluorescence of the eight beers could be explained for three components. By comparing with existing database literature, these components could be qualitatively assigned to the organic substances DOM (dissolved organic matter), DOC (dissolved organic carbon) or CDOM (colored dissolved organic matter) and OC (organic matter). The emission and extinction spectra helped to establish these organic substances as the three aromatic amino acids phenylalanine, tryptophan and tyrosine, iso-α-acid, phenolic compounds and the vitamin B group. In addition, correlations with the fluorescence intensities from the EEM (excitation–emission matrix) data were detected in combination with beer analyses. A correlation between the fluorescence intensity of origin and iso-α-acid could therefore be shown. The fluorescent amino acids phenylalanine, tryptophan and tyrosine showed only slight correlations with the fluorescent intensity. The calculated data showed that fluorescence spectroscopy with the obtained EEM represents a powerful real-time measurement, which offers sensitive monitoring for the identification of fluorescent substances in beer samples. Qualitative analysis combined with the PARAFAC analysis is also advantageous to identify the main components.




Similar content being viewed by others
References
Steiner E, Gastl M, Becker T (2011) Die Identifizierung von Trübungen in Bier (2). Brauwelt 7:193–205
Steiner E, Gastl M, Becker T (2011) Die Identifizierung von Trübungen in Bier (1). Brauwelt 05(06):161–166
Steiner E, Arendt EK, Gastl M, Becker T (2011) Influence of the malting parameters on the haze formation of beer after filtration. Eur Food Res Technol 233(4):587–597
Sikorska E, Górecki T, Khmelinskii IV, Sikorski M, de Keukeleire D (2004) Fluorescence spectroscopy for characterization and differentiation of beers. J Inst Brew 110(4):267–275
Christensen J, Nørgaard L, Bro R, Engelsen SB (2006) Multivariate autofluorescence of intact food systems. Chem Rev 106(6):1979–1994
Apperson K, Leiper KA, McKeown IP, Birch D (2002) Beer fluorescence and the isolation, characterisation and silica adsorption of haze-active beer proteins. J Inst Brew 108(2):193–199
Noack D, Lachenmeier D (2019) Nachweis einer Megasphaera-Kontamination bei Schankbier mittels fluoreszenzmarkiertertypes Gensonden
Demčenko AP (2015) Introduction to fluorescence sensing, 2nd edn. Cham, Heidelberg, New York, Dordrecht, London
Baschong W, Landmann L (2006) Fluorescence microscopy. In: Baschong W, Landmann L (Hrsg.) Cell biology Chapter 1—Fluorescence microscopy
Huang YY, Beal CM, Cai WW, Ruoff RS, Terentjev EM (2010) Micro-Raman spectroscopy of algae. Composition analysis and fluorescence background behavior. Biotechnol Bioeng 105(5):889–898
Huang Y-S, Karashima T, Yamamoto M, Ogura T, Hamaguchi H (2004) Raman spectroscopic signature of life in a living yeast cell. J Raman Spectrosc 35(7):525–526
Borlinghaus RT (2016) Konfokale Mikroskopie in Weiß. Optische Schnitte in allen Farben, Berlin, Heidelberg
Kubitscheck U (2017) Fluorescence microscopy. From principles to biological applications. Weinheim
Brand L, Johnson ML (Hrsg.) (2008) Fluorescence spectroscopy. Amsterdam
Lakowicz JR (2010) Principles of fluorescence spectroscopy, Third edition, corrected at 4. printing. New York, NY
Wedler G, Freund H-J (2012) Lehrbuch der physikalischen Chemie, 6., vollst. überarb. und aktualisierte Aufl. Weinheim
Valeur B, Berberan-Santos MN (2012) Molecular fluorescence. Principles and applications, 2. ed. Weinheim
Gordon R, Cozzolino D, Chandra S, Power A, Roberts J, Chapman J (2017) Analysis of Australian beers using fluorescence spectroscopy. Beverages 3(4):57
Guilbault, G. G.: Practical fluorescence (1999)
Ndou TT, Warner IM (1991) Applications of multidimensional absorption and luminescence spectroscopies in analytical chemistry. Chem Rev 91(4):493–507
Christensen J, Ladefoged AM, Nørgaard L (2005) Rapid determination of bitterness in beer using fluorescence spectroscopy and chemometrics. J Inst Brew 111(1):3–10
Friedrich W (1988) Vitamin B12 und verwandte Corrinoide. In: Walter de Gruyter, Berlin, New York
Gorinstein S, Goshev I, Moncheva S, Zemser M, Weisz M, Caspi A, Libman I, Lerner HT, Trakhtenberg S (2000) Intrinsic tryptophan fluorescence of human serum proteins and related conformational changes. J Protein Chem 19(8):637–642
Gorinstein S, Zemser M, Vargas-Albores F, Ochoa J-L, Paredes-Lopez O, Scheler C, Salnikow J, Martin-Belloso O, Trakhtenberg S (1999) Proteins and amino acids in beers, their contents and relationships with other analytical data. Food Chem 67(1):71–78
Sikorska E, Gliszczyńska-Swigło A, Insińska-Rak M, Khmelinskii I, Keukeleire de D, Sikorski M (2008) Simultaneous analysis of riboflavin and aromatic amino acids in beer using fluorescence and multivariate calibration methods. Anal Chim Acta 613(2):207–217
Chen J, LeBoeuf EJ, Dai S, Gu B (2003) Fluorescence spectroscopic studies of natural organic matter fractions. Chemosphere 50(5):639–647
Viñas P, Balsalobre N, López-Erroz C, Hernández-Córdoba M (2004) Liquid chromatographic analysis of riboflavin vitamins in foods using fluorescence detection. J Agric Food Chem 52(7):1789–1794
Sikorska E, Górecki T, Khmelinskii IV, Sikorski M, de Keukeleire D (2006) Monitoring beer during storage by fluorescence spectroscopy. Food Chem 96(4):632–639
Duyvis MG, Hilhorst R, Laane C, Evans DJ, Schmedding DJM (2002) Role of riboflavin in beer flavor instability. Determination of levels of riboflavin and its origin in beer by fluorometric apoprotein titration. J Agric Food Chem 50(6):1548–1552
Shahidi F, Naczk M (2004) Phenolics in food and nutraceuticals. Fla, Boca Raton
Sikorska E, Górecki T, Khmelinskii IV, Sikorski M, Kozioł J (2005) Classification of edible oils using synchronous scanning fluorescence spectroscopy. Food Chem 89(2):217–225
Narziß L, Back W (2012) Die Bierbrauerei. Band 2: Die Technologie der Würzebereitung
Andrés-Lacueva C, Mattivi F, Tonon D (1998) Determination of riboflavin, flavin mononucleotide and flavin–adenine dinucleotide in wine and other beverages by high-performance liquid chromatography with fluorescence detection. J Chromatogr A 823(1–2):355–363
Lawaetz AJ, Stedmon CA (2009) Fluorescence intensity calibration using the Raman scatter peak of water. Appl Spectrosc 63(8):936–940
MEBAK (Hrsg.) (2012) Würze, Bier, Biermischgetränke (Band 2): Methodensammlung der Mitteleuropäischen Brautechnischen Analysenkommission
Anger H-M (2006) Brautechnische Analysenmethoden. Rohstoffe
Convention EB (1992) Analytica EBC. Method 9.12. 3. J Inst Brew 98:21–22
Geddes CD (Hrsg.) (2018) Reviews in fluorescence 2017. Cham
Harris DC, Werner G, Werner T (Hrsg.) (2014) Lehrbuch der Quantitativen Analyse. Berlin
Bro R, Kiers HAL (2003) A new efficient method for determining the number of components in PARAFAC models. J Chemom J Chemom Soc 17(5):274–286
Stedmon CA, Bro R (2008) Characterizing dissolved organic matter fluorescence with parallel factor analysis. A tutorial. Limnol Oceanogr Methods 6(11):572–579
Murphy KR, Stedmon CA, Graeber D, Bro R (2013) Fluorescence spectroscopy and multi-way techniques. PARAFAC. Anal Methods 5(23):6557–6566
Andersen CM, Bro R (2003) Practical aspects of PARAFAC modeling of fluorescence excitation-emission data. J Chemom J Chemom Soc 17(4):200–215
Prasad M, Kumar A, Atta DK, Ramanathan L (2014) Spectrofluorometric analysis of organic matter in the Sundarban mangrove, Bangladesh
Lablicate GmbH 2019: openfluor data base. URL: https://openfluor.lablicate.com/. Abrufdatum 12.06.2019
Leenheer JA, Croué J-P (2003) Peer reviewed. Characterizing aquatic dissolved organic matter
Walmsley TA, Lever M (1982) Fluorometric measurement of furfural and 5-hydroxymethylfurfural. Anal Biochem 124(2):446–451
Acknowledgements
Special thanks go to Dr. Carolin Heim from the Chair of Urban Water Systems Engineering, Garching (TUM), as we were able to carry out our measurements on the fluorescence spectroscope there.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Eva-Maria Kahle, Martin Zarnkow and Fritz Jacob declare that they have no conflict of interest.
Compliance with ethics requirements
The authors Eva-Maria Kahle, Martin Zarnkow and Fritz Jacob hereby confirm that this manuscript was prepared according to and follows the COPE guidelines and has not already been published nor is it under consideration for publication elsewhere. This article does not contain any studies with human or animal subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kahle, EM., Zarnkow, M. & Jacob, F. Substances in beer that cause fluorescence: evaluating the qualitative and quantitative determination of these ingredients. Eur Food Res Technol 245, 2727–2737 (2019). https://doi.org/10.1007/s00217-019-03394-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-019-03394-x