Abstract
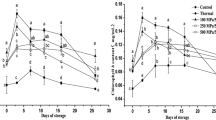
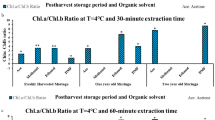
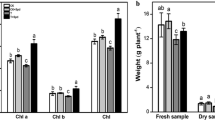
Chlorophylls and chlorophyll–protein complexes determine the color and other sensory properties of spinach. This study investigated the effects of high hydrostatic pressure (HHP) (100, 250, and 500 MPa for 5 min) treatments on structure, chlorophylls and soluble protein contents, protein peptide and fatty acid compositions, chlorophylls absorption spectra, emission, and excitation fluorescence spectra of thylakoid membrane in spinach, in order to better understand the changes in chlorophylls and chlorophyll–protein complexes under HHP. The result showed that HHP-treated samples showed a compact and stacked structure of thylakoid membrane in spinach. For the components of thylakoid membrane, both of chlorophylls and soluble protein contents were significantly (p<0.05) reserved by HHP treatments. The composition of proteins, peptides, and fatty acids was close to untreated samples, revealing higher stability under HHP. In addition, chlorophylls emission and excitation fluorescence spectra capacities of thylakoid membrane were better maintained under HHP treatments compared with thermal treatment, indicating higher light-harvesting and excitation efficiencies of Photosystem II (PS II). As the key functional component of thylakoid membrane, retention of PS II reflected the stability of thylakoid membrane functions under HHP. Hence, chlorophylls and chlorophyll–protein complexes were effectively sustained under HHP treatments, providing new opportunities to preserve the color quality of green vegetables.







Similar content being viewed by others
References
Steet JA, Tong CH (1996) Degradation kinetics of green color and chlorophylls in peas by colorimetry and HPLC. J Food Sci 61:924–927
Daum B, Kühlbrandt W (2011) Electron tomography of plant thylakoid membranes. J Exp Bot 62:2393–2402
Jarvis P, Chen LJ, Li H, Peto CA, Fankhause C, Chory J (1998) An Arabidopsis mutant defective in the plastid general protein import apparatus. Science 282:100–103
Wang RR, Xu Q, Yao J, Zhang YJ, Liao XJ, Hu XS, Wu JH, Zhang Y (2013) Post-effects of high hydrostatic pressure on green color retention and related properties of spinach puree during storage. Innov Food Sci Emerg 17:63–71
Kirchhoff H, Haferkamp S, Allen JF, Epstein DBA, Mullineaux CW (2008) Protein diffusion and macromolecular crowding in thylakoid membrane. Plant Physiol 146:1571–1578
Sharkey TD (2000) Perspective: some like it hot. Plant Biol 287:435–436
Norton T, Sun DW (2008) Recent advances in the use of high pressure as an effective processing technique in the food industry. Food Bioprocess Technol 1:2–34
Delgado FJ, Rodríguez-Pinilla J, Márquze G, Roa I, Ramírez R (2015) Physicochemical, proteolysis and texture changes during the storage of a mature soft cheese treated by high-pressure hydrostatic. Eur Food Res Technol 240:1167–1176
Jacobo-Velázquez DA, Hernández-Brenes C (2010) Biochemical changes during the storage of high hydrostatic pressure processed avocado paste. J Food Sci 75:264–270
Wang RR, Wang TT, Zheng Q, Hu XS, Zhang Y, Liao XJ (2012) Effects of high hydrostatic pressure on color of spinach puree and related properties. J Sci Food Agric 92:1417–1423
Schlüter O, Foerster J, Geyer M, Knorr D, Herppich WB (2009) Characterization of high-hydrostatic-pressure effects on fresh produce using chlorophyll fluorescence image analysis. Food Bioprocess Technol 2:291–299
Eggleston V, Tanner DJ (2005) Are carrots under pressure still alive?—the effect of high pressure processing on respiration rate of carrots. Acta Hortic 687:371–374
Wittig I, Braun HP, Schägger H (2006) Blue native PAGE. Nat Protoc 1:418–428
Black EP, Linton M, Mccall RD, Curran W, Fitzgerald GF, Kelly AL, Patterson MF (2008) The combined effects of high pressure and nisin on germination and inactivation of Bacillus spores in milk. J Appl Microbiol 105:78–87
Rudra SG, Sarkar BC, Shivhare US (2008) Thermal degradation kinetics of chlorophyll inpureed coriander leaves. Food Bioprocess Technol 1:91–99
Teng SS, Chen BH (1999) Formation of pyrochlorophylls and their derivatives in spinach leaves during heating. Food Chem 65:367–373
Lowry O, Rosebrough A, Farr A, Randall MR (1951) Protein measurement with Folin phenol reagent. J Biol Chem 193:680–685
Kim N, Son SH, Maeng JS, Cho YJ, Kim CT (2012) Enzymatic hydrolysis of anchovy fine powder at high and ambient pressure, and characterization of the hydrolyzates. J Sci Food Agric. doi:10.1002/jsfa.7173
Xu Y, Siegenthaler PA (1997) Low temperature treatments induce an increase in the relative content of both linolenic and △3-hexadecenoic acids in thylakoid membrane phosphatidylglycerol of squash cotyledons. Plant Cell Physiol 38:611–618
Ferroni L, Baldisseroto C, Pantaleni L, Fasulo MP, Fagioli P, Pancaldi S (2009) Degreening of the unicellular alga Euglena gracilis: thylakoid membrane composition, room temperature fluorescence spectra and chloroplast morphology. Plant Biol 11:631–641
Šiffel P, Braunová Z (1999) Release and aggregation of the light-harvesting complex in intact leaves subjected to strong CO2 deficit. Photosynth Res 61:217–226
Knorr D (1995) High pressure effects on plant derived foods. In: Ledward DA, Johnston DE, Earnshaw RG, Hastings APM (eds) High pressure processing of foods. Nottingham University Press, Loughborough, pp 123–135
Trejo Araya X, Hendrickx M, Verlinden BE, Van Buggenhout S, Smale NJ, Stewart C, John Mawson A (2007) Understanding texture changes of high pressure processed fresh carrots: a microstructural and biochemical approach. J Food Eng 80:873–884
Van Loey A, Ooms V, Weemaes C, Van den Broeck I, Ludikhuyze L, Indrawati DS, Hendrickx M (1998) Thermal and pressure-temperature degradation of chlorophyll in broccoli (Brassica oleracea L. Italica) juice: a kinetic study. J Agric Food Chem 46:5289–5294
Herendeen SL, Vanbogelen RA, Neidhardt FC (1979) Levels of major proteins of Escherichia coli during growth at different temperatures. J Bacteriol 139:185–194
Eshaghi S, Andersson B, Barber J (1999) Isolation of a highly active PS II–LHC II super-complex from thylakoid membrane by a direct method. FEBS Lett 446:23–26
Bricker TM (1990) The structure and function of CPa-1 and CPa-2 in photosystem II. Photosynth Res 24:1–13
Tang YL, Wen XG, Lu QT, Yang ZP, Cheng ZK, Lu CM (2007) Heat stress induces an aggregation of the light-harvesting complex of photosystem II in spinach plants. Plant Physiol 143:629–638
Waloszek A, Tulej M, Wieckowski S (1992) Spectroscopic analysis of chlorophyll bleaching in heat-pretreated thylakoid membranes isolated from cucumber cotyledons. Photosynthetica 27:109–118
Havaux M (1992) Stress tolerance of photosystem II in vivo. Antagonistic effects of water, heat, and photoinhibition stresses. Plant Physiol 100:422–424
Wei HM, Chen YW, Zhang NH, Zhao Y, Du LF (2005) Study on the spectra properties of cotyledon thylakoid membranes in chlorophyll-reduced rapeseed mutant Cr3529 and its wild type. Acta Botanica Boreali-Occidentalia Sinia 25:250–255
Naus J, Dvorak L, Kuropatwa R, Maslan M (1992) Transition in the thylakoid membranes of barley leaves studied by chlorophyll fluorescence temperature curve. Photosynthetica 27:563–570
Acknowledgments
This work was supported by National Natural Science Foundation of China (No. 31271910) and the Sci & Tech fund in the Twelfth Five-Year Plan of China (No. 2012BAD31B09).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Compliance with ethics requirements
The article does not contain any studies with human or animal subjects.
Rights and permissions
About this article
Cite this article
Wang, R., Ding, S., Hu, X. et al. Effects of high hydrostatic pressure on chlorophylls and chlorophyll–protein complexes in spinach. Eur Food Res Technol 242, 1533–1543 (2016). https://doi.org/10.1007/s00217-016-2654-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-016-2654-8