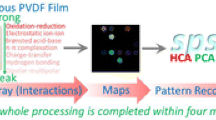
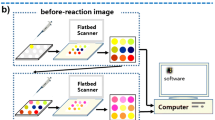
Abstract
Detection of human-generated volatile organic compounds (VOCs) is a new pathway for assessing health. Herein, a polyvinylidene fluoride (PVDF)-based colorimetric sensor array was designed for detecting disease-related VOCs (DVOCs) within 15 min, using a complex of Cu metal–organic framework, graphene aerogel, and dyes as response materials. Fingermaps derived from 28 DVOCs were obtained for further data processing. Pattern recognition was successfully employed in the correct discrimination of 28 DVOCs in low (10 μM), medium (100 μM), and high (300 μM) concentrations. Importantly, the sensor array also presented excellent discrimination ability and application potential when detecting VOCs produced by human cancer and normal cells. In general, VOC acquisition is noninvasive and harmless, and the PVDF-based sensor arrays are simple and visual. Such advantages expand their further application potential.









Similar content being viewed by others
References
Cho SM, Kim YJ, Heo GS, Shin S-M. Two-step preconcentration for analysis of exhaled gas of human breath with electronic nose. Sens Actuators, B Chem. 2006;117(1):50–7. https://doi.org/10.1016/j.snb.2005.10.050.
Kamal F, Kumar S, Edwards MR, Veselkov K, Belluomo I, Kebadze T, Romano A, Trujillo-Torralbo MB, Shahridan Faiez T, Walton R, Ritchie AI, Wiseman DJ, Laponogov I, Donaldson G, Wedzicha JA, Johnston SL, Singanayagam A, Hanna GB. Virus-induced volatile organic compounds are detectable in exhaled breath during pulmonary infection. Am J Respir Crit Care Med. 2021;204(9):1075–85. https://doi.org/10.1164/rccm.202103-0660OC.
Pauling L, Robinson AB, Teranishi R, Cary P. Quantitative analysis of urine vapor and breath by gas-liquid partition chromatography. Proc Natl Acad Sci U S A. 1971;68(10):2374–6. https://doi.org/10.1073/pnas.68.10.2374.
Jalal AH, Alam F, Roychoudhury S, Umasankar Y, Pala N, Bhansali S. Prospects and challenges of volatile organic compound sensors in human healthcare. ACS Sens. 2018;3(7):1246–63. https://doi.org/10.1021/acssensors.8b00400.
Ripszam M, Bruderer T, Biagini D, Ghimenti S, Lomonaco T, Di Francesco F. Biological studies with comprehensive 2D-GC-HRMS screening: exploring the human sweat volatilome. Talanta. 2023;257:124333. https://doi.org/10.1016/j.talanta.2023.124333.
Drabinska N, Flynn C, Ratcliffe N, Belluomo I, Myridakis A, Gould O, Fois M, Smart A, Devine T, Costello BL. A literature survey of all volatiles from healthy human breath and bodily fluids: the human volatilome. J Breath Res. 2021;15(3):034001. https://doi.org/10.1088/1752-7163/abf1d0.
Ding X, Zhang Y, Zhang Y, Ding X, Zhang H, Cao T, Qu ZB, Ren J, Li L, Guo Z, Xu F, Wang QX, Wu X, Shi G, Haick H, Zhang M. Modular assembly of MXene frameworks for noninvasive disease diagnosis via urinary volatiles. ACS Nano. 2022;16(10):17376–88. https://doi.org/10.1021/acsnano.2c08266.
Tyagi H, Daulton E, Bannaga AS, Arasaradnam RP, Covington JA. Urinary volatiles and chemical characterisation for the non-invasive detection of prostate and bladder cancers. Biosensors (Basel). 2021;11:437. https://doi.org/10.3390/bios11110437.
Ligor T, Adamczyk P, Kowalkowski T, Ratiu IA, Wenda-Piesik A, Buszewski B. Analysis of VOCs in urine samples directed towards of bladder cancer detection. Molecules. 2022;27(15):5023. https://doi.org/10.3390/molecules27155023.
Janssens E, van Meerbeeck JP, Lamote K. Volatile organic compounds in human matrices as lung cancer biomarkers: a systematic review. Crit Rev Oncol Hematol. 2020;153:103037. https://doi.org/10.1016/j.critrevonc.2020.103037.
Koureas M, Kirgou P, Amoutzias G, Hadjichristodoulou C, Gourgoulianis K, Tsakalof A. Target analysis of volatile organic compounds in exhaled breath for lung cancer discrimination from other pulmonary diseases and healthy persons. Metabolites. 2020;10(8):317. https://doi.org/10.3390/metabo10080317.
Pizzini A, Filipiak W, Wille J, Ager C, Wiesenhofer H, Kubinec R, Blasko J, Tschurtschenthaler C, Mayhew CA, Weiss G, Bellmann-Weiler R. Analysis of volatile organic compounds in the breath of patients with stable or acute exacerbation of chronic obstructive pulmonary disease. J Breath Res. 2018;12(3):036002. https://doi.org/10.1088/1752-7163/aaa4c5.
Saktiawati AMI, Putera DD, Setyawan A, Mahendradhata Y, van der Werf TS. Diagnosis of tuberculosis through breath test: a systematic review. EBioMedicine. 2019;46:202–14. https://doi.org/10.1016/j.ebiom.2019.07.056.
Barker M, Hengst M, Schmid J, Buers HJ, Mittermaier B, Klemp D, Koppmann R. Volatile organic compounds in the exhaled breath of young patients with cystic fibrosis. Eur Respir J. 2006;27(5):929–36. https://doi.org/10.1183/09031936.06.00085105.
van de Kant KDG, van der Sande LJTM, Jöbsis Q, van Schayck OCP, Dompeling E. Clinical use of exhaled volatile organic compounds in pulmonary diseases: a systematic review. Respir Res. 2012;13:117. https://doi.org/10.1186/1465-9921-13-117.
Vasilescu A, Hrinczenko B, Swain GM, Peteu SF. Exhaled breath biomarker sensing. Biosens Bioelectron. 2021;182:113193. https://doi.org/10.1016/j.bios.2021.113193.
Han B, Wang J, Yang W, Chen X, Wang H, Chen J, Zhang C, Sun J, Wei X. Hydrothermal synthesis of flower-like In2O3 as a chemiresistive isoprene sensor for breath analysis. Sens Actuators B Chem. 2020;309:127788. https://doi.org/10.1016/j.snb.2020.127788.
Tyas FH, Nikita JG, Apriyanto DK, Mitrayana A, Amin MN. The performance of CO2 laser photoacoustic spectrometer in concentration acetone detection as biomarker for diabetes mellitus type 2. J Phys Conf Ser. 2018;1011:012056. https://doi.org/10.1088/1742-6596/1011/1/012056.
Cao S, Xu Y, Yu Z, Zhang P, Xu X, Sui N, Zhou T, Zhang T. A dual sensing platform for human exhaled breath enabled by Fe-MIL-101-NH(2) metal-organic frameworks and its derived Co/Ni/Fe trimetallic oxides. Small. 2022;18(42):e2203715. https://doi.org/10.1002/smll.202203715.
Xu S, Yang C, Tian Y, Lu J, Jiang Y, Guo H, Zhao J, Peng H. Exploitation of Schottky-junction-based sensors for specifically detecting ppt-concentration gases. ACS Sens. 2022;7(12):3764–72. https://doi.org/10.1021/acssensors.2c01591.
Zou X, Zhou W, Lu Y, Shen C, Hu Z, Wang H, Jiang H, Chu Y. Exhaled gases online measurements for esophageal cancer patients and healthy people by proton transfer reaction mass spectrometry. J Gastroenterol Hepatol. 2016;31(11):1837–43. https://doi.org/10.1111/jgh.13380.
Kwon IJ, Jung TY, Son Y, Kim B, Kim SM, Lee JH. Detection of volatile sulfur compounds (VSCs) in exhaled breath as a potential diagnostic method for oral squamous cell carcinoma. BMC Oral Health. 2022;22(1):268. https://doi.org/10.1186/s12903-022-02301-3.
Phillips M, Cataneo RN, Ditkoff BA, Fisher P, Greenberg J, Gunawardena R, Kwon CS, Rahbari-Oskoui F, Wong C. Volatile markers of breast cancer in the breath. Breast J. 2003;9(3):184–91. https://doi.org/10.1046/j.1524-4741.2003.09309.x.
Altomare DF, Di Lena M, Porcelli F, Travaglio E, Longobardi F, Tutino M, Depalma N, Tedesco G, Sardaro A, Memeo R, de Gennaro G. Effects of curative colorectal cancer surgery on exhaled volatile organic compounds and potential implications in clinical follow-up. Ann Surg. 2015;262(5):862–6. https://doi.org/10.1097/SLA.0000000000001471. (discussion 866-867).
Wu X, Wang D, Shi L, Wang H, Wang J, Sun J, Li C, Tian X. A compact gas chromatography platform for detection of multicomponent volatile organic compounds biomarkers. Rev Sci Instrum. 2022;93(6):065003. https://doi.org/10.1063/5.0086618.
Ullah S, Sandqvist S, Beck O. A liquid chromatography and tandem mass spectrometry method to determine 28 non-volatile drugs of abuse in exhaled breath. J Pharm Biomed Anal. 2018;148:251–8. https://doi.org/10.1016/j.jpba.2017.10.003.
Lu Y, Niu W, Zou X, Shen C, Xia L, Huang C, Wang H, Jiang H, Chu Y. Glass bottle sampling solid phase microextraction gas chromatography mass spectrometry for breath analysis of drug metabolites. J Chromatogr A. 2017;1496:20–4. https://doi.org/10.1016/j.chroma.2017.03.061.
Gashimova E, Osipova A, Temerdashev A, Porkhanov V, Polyakov I, Perunov D, Dmitrieva E. Exhaled breath analysis using GC-MS and an electronic nose for lung cancer diagnostics. Anal Methods. 2021;13(40):4793–804. https://doi.org/10.1039/d1ay01163d.
Da Silva Sena CR, de Queiroz AE, de Gouveia BP, Percival E, Prangemeier B, Donoghue C, Terry S, Burke T, Gunning W, Murphy VE, Robinson PD, Sly PD, Gibson PG, Collison AM, Mattes J. Higher exhaled nitric oxide at 6 weeks of age is associated with less bronchiolitis and wheeze in the first 12 months of age. Thorax. 2022;77(11):1106. https://doi.org/10.1136/thoraxjnl-2021-217299.
Li Z, Askim JR, Suslick KS. The optoelectronic nose: colorimetric and fluorometric sensor arrays. Chem Rev. 2019;119(1):231–92. https://doi.org/10.1021/acs.chemrev.8b00226.
Shi C, Tang Y, Yang H, Yang J, Wu Y, Sun H, Yin S, Wang G. Capture and detection of Escherichia coli with graphene aerogels. J Mater Chem B. 2022;10(40):8211–7. https://doi.org/10.1039/d2tb01749k.
Wang S, Deng W, Yang L, Tan Y, Xie Q, Yao S. Copper-based metal-organic framework nanoparticles with peroxidase-like activity for sensitive colorimetric detection of staphylococcus aureus. ACS Appl Mater Interfaces. 2017;9(29):24440–5. https://doi.org/10.1021/acsami.7b07307.
Velusamy P, Su CH, Ramasamy P, Arun V, Rajnish N, Raman P, Baskaralingam V, Senthil Kumar SM, Gopinath SCB. Volatile organic compounds as potential biomarkers for noninvasive disease detection by nanosensors: a comprehensive review. Crit Rev Anal Chem. 2022;1–12. https://doi.org/10.1080/10408347.2022.2043145
Acknowledgements
This work was supported by the Chongqing Natural Science Foundation (CSTB2023NSCQ-BHX0078), Research projects of Wanzhou, China (wzstc20220314), Graduate Scientific Research and Innovation Foundation of Chongqing, China (CYS23136), National Facility for Translational Medicine (Shanghai) Open Research Fund (TMSK-2021-113), Chongqing Graduate Tutor Team Construction Project, Analytical and Testing Center of Chongqing University for TEM and the sharing fund of Chongqing University's large equipment.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hou, J., Liu, X., Hou, C. et al. A PVDF-based colorimetric sensor array for noninvasive detection of multiple disease-related volatile organic compounds. Anal Bioanal Chem 415, 6647–6661 (2023). https://doi.org/10.1007/s00216-023-04941-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-023-04941-y