Abstract
Recent years showed a boost in knowledge about the presence and fate of micropollutants in the environment. Instrumental and methodological developments mainly in liquid chromatography coupled to mass spectrometry hold a large share in this success story. These techniques soon complemented gas chromatography and enabled the analysis of more polar compounds including pesticides but also household chemicals, food additives, and pharmaceuticals often present as traces in surface waters. In parallel, sample preparation techniques evolved to extract and enrich these compounds from biota and water samples. This review article looks at very polar and ionic compounds using the criterion log P ≤ 1. Considering about 240 compounds, we show that (simulated) log D values are often even lower than the corresponding log P values due to ionization of the compounds at our reference pH of 7.4. High polarity and charge are still challenging characteristics in the analysis of micropollutants and these compounds are hardly covered in current monitoring strategies of water samples. The situation is even more challenging in biota analysis given the large number of matrix constituents with similar properties. Currently, a large number of sample preparation and separation approaches are developed to meet the challenges of the analysis of very polar and ionic compounds. In addition to reviewing them, we discuss some trends: for sample preparation, preconcentration and purification efforts by SPE will continue, possibly using upcoming mixed-mode stationary phases and mixed beds in order to increase comprehensiveness in monitoring applications. For biota analysis, miniaturization and parallelization are aspects of future research. For ionic or ionizable compounds, we see electromembrane extraction as a method of choice with a high potential to increase throughput by automation. For separation, predominantly coupled to mass spectrometry, hydrophilic interaction liquid chromatography applications will increase as the polarity range ideally complements reversed phase liquid chromatography, and instrumentation and expertise are available in most laboratories. Two-dimensional applications have not yet reached maturity in liquid-phase separations to be applied in higher throughput. Possibly, the development and commercial availability of mixed-mode stationary phases make 2D applications obsolete in semi-targeted applications. An interesting alternative will enter routine analysis soon: supercritical fluid chromatography demonstrated an impressive analyte coverage but also the possibility to tailor selectivity for targeted approaches. For ionic and ionizable micropollutants, ion chromatography and capillary electrophoresis are amenable but may be used only for specialized applications such as the analysis of halogenated acids when aspects like desalting and preconcentration are solved and the key advantages are fully elaborated by further research.

Graphical abstract
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Already in 1985, Richardson and Bowron [1] discussed the fate of pharmaceuticals and other micropollutants in the (aquatic) environment. Over the last 35 years, the interest in their fate steadily increased as can be seen by a statistic for the keywords environmental and most common substance classes (antibiotics, metabolites, but also industrial compounds, herbicides, see Fig. 1a). Recent years showed improvements throughout the whole analytical process. However, due to the different physicochemical properties of micropollutants caused by a variety of functional groups, it is obvious that there is no analytical method for the successful simultaneous analysis of all contaminants in a sample. Whereas for the analysis of non-polar to medium polar compounds (log P > 1) successful and reliable analysis methods already exist, there is a lack of methods for the analysis of very polar and ionic compounds (vPICs). Reemtsma et al. [2] even pointed out that—at present—an analytical gap exists for these substances. Despite great effort in the past years in environmental analysis, visible from the rising number of publications (see Fig. 1a, enlarged section), still little is known about the occurrence, fate, and potential ecological effects of vPICs and their mostly even more polar metabolites in the (aquatic) environment. Since these compounds are highly polar, often mobile and possibly also persistent, they have the potential to spread through the water cycle and even reach drinking water. For ionic compounds, the charge influences sorption and uptake, which is not well understood. With increasing awareness of their possible ecotoxicological relevance, e.g., effects on aquatic organisms and health threats, the demand of general public for tightened regulation is rising. This requires further research to increase knowledge and understanding of occurrence and effects. Consequently, sensitive and precise methods for the analysis of these mostly small organic compounds are needed.
a Non-exhaustive trend of numbers of published articles over the last 35 years listed in “Web of Science” using the keywords environmental + most common substance classes (metabolites, industrial compounds, herbicides, or antibiotics, see also b) and additionally to it “ionic” in a second search (see enlarged section). b Classification of the 237 very polar and ionic compounds (vPICs, log P ≤ 1) analyzed in 63 cited articles. Details of the compounds can be found in Table S1 in the ESM. Only substance classes with ≥ 3 members are included. c Scatterplot with marginal boxplot of log P and the corresponding log D (pH 7.4) values of the compounds. Log P and log D data were extracted from Chemicalize provided by ChemAxon (10/05/2020). Colors code for different charge numbers (given as absolute number (modulus)). Selected analytes with large differences between log P and log D (see also ESM Table S1) are (1) Gd-BT-DO3A, (2) neomycin, (3) tobramycin, (4) Gd-DTPA, (5) cephalosporin C, (6) gentamicin, (7) glyphosate, (8) cephalozin, (9) 3-methylphosphinic acid, (10) itaconate, and (11) 3,4-dihydroxyphenylacetic acid
We compiled data from recent articles dealing with the analysis of vPICs (log P ≤ 1) in the environment. Figure 1 b classifies 237 different vPICs that were analyzed in 63 published cited here with regard to their application. Clearly, vPICs are present in all fields of applications. It is important to mention that this group also contains other micropollutants than pharmaceuticals and their metabolites (which themselves are mostly more polar, e.g., abacavir and its metabolite abacavir carboxylate with log D (pH 7.4) values of 0.38 and − 2.72, respectively (see also Table S1 in the Electronic Supplementary Material, ESM). In fact, only less than about 40% of the compounds analyzed here are pharmaceuticals. Other substance classes like food supplements (e.g., (artificial) sweetener) or industrial compounds in general are quite common representatives for vPICs. In order to get a feeling for the polarity of the substances, Fig. 1c shows the distribution of their log P values. Having in mind that at pH 7.4 over 70% of these compounds have charge numbers equal to or higher than 0.5, it is crucial to consider their corresponding log D values at pH 7.4 which are also summarized in Fig. 1c (details are presented in Table S1 in the ESM). A Welch’s t test showed a statistically significant difference between the log P values and their corresponding log D (pH 7.4) values. Details on the correlation of log P and log D (pH 7.4) are also presented in Fig. 1 c. As expected, the average polarity of these compounds increases (log D decreases) which will result in different behavior in the environment. Several compounds such as gadolinium-based contrast agents or aminoglycoside antibiotics have large differences between log P and log D (pH 7.4) (see Fig. 1c).
Distribution of log P and log D (pH 7.4) values of the compounds listed in ESM Table S1 (classification according to application as in Fig. 1b). Details of the correlation between log P and log D (7.4) is shown in Fig. 1c). Contours show estimated log P scopes of the sample preparation and separation techniques as discussed in this article. DLLME, dispersive liquid-liquid microextraction; EC, evaporative concentration; EME, electromembrane concentration; CNT, carbon nanotubes; HLB, hydrophilic-lipophilic balance; RPLC, reversed phase liquid chromatography; HILIC, hydrophilic interaction liquid chromatography; SFC, supercritical fluid chromatography; MMLC, mixed-mode liquid chromatography; IC, ion chromatography; (NA)CE, (nonaqueous) capillary electrophoresis
Mass spectrometry (MS) is the detection method of choice to reach the required detection limits, as it provides identification strength and an additional separation dimension when coupled to the various separation techniques [3]. Current screening and environmental monitoring strategies for micropollutants often rely on solid-phase extraction (SPE) for sample preparation and liquid chromatography (LC), mostly C18 reversed phase (RP) stationary phases, for separation. Both SPE and reversed phase liquid chromatography (RPLC) have limitations for vPICs. This is due to insufficient retention, leading to poor extraction efficiencies and poor separation of early eluting peaks in RPLC. Figure 2 provides an overview on current analytical methods (sample preparation and separation) applicable for vPIC analysis and the range of log P/log D values, which can be covered (in part, however, of course not the whole range with a single analysis). Limitations regarding the polarity range covered are evident in many up-to-date reviews [4,5,6,7]. Various analytical approaches were tested to address polar micropollutants with hydrophilic interaction liquid chromatography (HILIC) being the most prominent method, followed by supercritical fluid chromatography (SFC), mixed-mode liquid chromatography (MMLC), and combinations thereof. We will discuss the applicability of ion chromatography (IC) and capillary electrophoresis (CE) suited for ionic and ionizable compounds (as the majority of polar compounds is ionizable). New instrumental developments, e.g., for IC-MS, will be discussed and also innovations, e.g., improved stationary phases with a column material particle size less than 2 μm for ultra-high performance SFC (UHPSFC).
Environmental samples, especially biota samples, have complex matrices. Matrix effects seem to be relevant especially for very polar analytes [8]. They have to be addressed enhancing selectivity (both for separation and detection) and using sample preparation strategies. While common sample preparation techniques like SPE are still advancing quickly, multiresidue methods such as QuEChERS (quick, easy, cheap, effective, rugged, and safe) found their way from food to environmental soil and biota analysis. We here want to discuss some alternatives, too, such as electromembrane extraction (EME), some of them only applicable for ionic and ionizable analytes.
In the past, there have been some reviews or trend articles already addressing the analysis of polar compounds [3,4,5,6, 9]. In this review article, we will focus on liquid-phase separations coupled to MS and sample preparation strategies for the analysis of vPICs (log P ≤ 1) in environmental samples including water and biota. We critically discuss new developments in the field differentiating between targeted and non-targeted methods. We here want to discuss sample preparation in combination with different separation methods, e.g., IC, CE, and SFC, and to elaborate recent trends and emphasize their potential for the analysis of vPICs in environmental samples in the future.
Trends in sample preparation
Sample preparation for environmental matrices
Beside robustness and reproducibility, the main objectives of sample preparation are the removal of matrix compounds, analyte recovery, and especially analyte preconcentration, whereas the latter two are also often combined in the term enrichment factor. Since most micropollutants in environmental samples are present at trace levels (ng/l to μg/l range), separation techniques alone are not able to reach these concentration levels, and they can be utilized only if sample enrichment is applied [10,11,12,13]. Moreover, smaller initial sample sizes (especially important in biota analysis of invertebrates), improvement in extraction selectivity for targeted analysis, and also coverage in non-target analysis are essential [3]. Coupling to analytical separation methods plays an important role [14]. Another aspect is green analytical chemistry, which has emerged in the 1990s, due to many analytical methods themselves generating a significant amount of chemical waste, resulting in a great environmental impact. It is required to reduce the amount and toxicity of applied solvents and reagents, especially by automation and miniaturization [15]. In this section, we focus on modern approaches for the sample preparation of polar and ionic analytes in water and biota samples.
Solid-phase extraction
Conventional offline SPE is the method of choice for isolating and enriching organic micropollutants from environmental samples with a wide range of sorbents available [16]. Based on the nature of the analytes, a careful choice of the sorbent material allows obtaining high recoveries and enrichment factors, typically in the range of 2–1000. Of all sorbents available for SPE, Oasis HLB is the most common sorbent used for environmental samples. It is a copolymeric sorbent with hydrophilic and lipophilic properties and can be used for a wide range of target compounds. It is water-wettable and stable over the whole pH range. In environmental analysis, it was used for the extraction of pharmaceuticals of various therapeutic classes from different matrices including wastewater [17, 18], sludge [19], and fish tissue [20]. However, the study of Boulard et al. [21] showed limitations of this material for polar pharmaceuticals and their transformation products with a log D (pH 7) close to or below 0. Alternatives to Oasis HLB are carbonaceous SPE sorbents, as they combine polar and non-polar interactions, making them suitable for a large polarity range (see Zahn et al. [3] for further discussion). In environmental analysis, they were applied for the analysis of polar pesticides [22] and herbicides [23].
Mixed-mode SPE sorbents became commercially available, which are able to retain compounds through both hydrophobic interactions and ionic interactions so that ionic or ionizable compounds can also be extracted [6]. For example, Scheurer et al. [24] used Strata X-CW columns to extract the ionic antidiabetic metformin from sewage and surface water with a relative recovery ≥ 90%. However, even SPE materials intended for polar compounds are often unable to retain the most polar compounds and ion exchange materials are limited to charged compounds. Accordingly, either combinations of different SPE sorbents or alternative methods such as evaporative concentration (EC) are needed to enrich these compounds from matrices: Köke et al. [25] used mixed-bed SPE originally developed by Kern et al. [26] combining three SPE materials to enrich polar compounds from various environmental waters. The results were compared with a sample preparation method based on EC. The study showed that both approaches provided a higher coverage of analytes compared with Oasis HLB. Mean recoveries for the enriched analytes were similar; however, lower matrix effects were observed for the HLB method in HILIC-MS/MS. Mechelke et al. [27] compared EC with mixed-bed multilayer SPE for the enrichment of 590 organic substances from river water and wastewater. The results showed, that overall, EC was better suited for the enrichment of polar analytes (see also [3]), albeit considerable signal suppression was observed for the EC-enriched samples. However, there is still no method that covers the complete range of vPICs; therefore, further research is necessary to increase the analyte coverage and to further reduce matrix effects especially for polar analytes in biota samples, which may have more naturally occurring interferents.
SPE can be applied online and offline, with specific benefits and drawbacks of these two approaches. A major advantage of online SPE is that it analyzes the entire eluate from the SPE extract, hence providing better preconcentration factors, sensitivity, and recovery than most offline SPE approaches. In addition, online SPE has low solvent consumption requirements thereby decreasing the costs for organic solvents waste disposal [28]. Problems may arise from the compatibility with subsequent RPLC due to an insufficient elution strength [16]. However, Huntscha et al. [29] demonstrated that this problem can be overcome by adding water after the enrichment step to improve compatibility with the separation of polar compounds. In their study, a multiresidue method for the analysis of 88 polar organic micropollutants (with a broad range of physicochemical properties: log D (pH 7) ranging from − 4.2 to 4.2) was applied to analyze ground, surface, and wastewater using online mixed-bed multilayer SPE coupled to HPLC-MS. The majority of the compounds (~ 80%) could be quantified below 10 ng/l in groundwater and surface water and below 100 ng/l in wastewater using a sample volume of 20 ml. The method showed good relative recoveries.
Major drawbacks of SPE are e.g. limited sorption capacity and clogging of sorbent pores by suspended particles/matrix compounds. An alternative extraction mode that prevents these problems is dispersive SPE (d-SPE). It provides a large active surface for sorption. Recently, many novel materials were developed. Cai et al. [30] showed a successful application of vortex-assisted dispersive micro-SPE based on a novel porous metal organic framework for the determination of amphenicols and its metabolite in aquaculture water. Under optimal conditions, the relative recoveries were > 70%. Since d-SPE requires two centrifugation steps, automation of the technique is difficult, thus may not be suitable for higher throughput. A further development of d-SPE is magnetic SPE (m-SPE) using sorbents with superparamagnetic properties and high adsorption capacity, so that filtration or centrifugation steps can be avoided. Among the most recent functionalized magnetic materials are magnetite (Fe3O4) nanoparticles coated with silica, different polymers, nanomaterials, and ionic liquids [31]. In a study of Luo et al. [32], a magnetic composite made of graphene and Fe3O4 @ SiO2 was prepared by simple adsorption. It was used as an extraction medium for the effective and efficient enrichment of six sulfonamide antibiotics in environmental water samples. However, SPE cannot be regarded a green method given its relatively high solvent consumption.
Another mode of SPE is solid-phase microextraction (SPME). It is fast, versatile, sensitive, and solvent-free. The isolation and preconcentration of the analytes occurs in a single step, providing a simple sample preparation. In its most popular configuration, the SPME device consists of a fused-silica rod coated with a thin layer of a suitable polymeric coating (e.g., polydimethylsiloxane, polyacrylate, and carbowax). Since its introduction in 1990 by Pawliszyn [33], SPME applications significantly broadened also for the analysis of environmental samples [34,35,36]. Aresta et al. [37], for example, developed an SPME-LC-UV method for the determination of the polar antibiotic chloramphenicol in urine and environmental water samples. For SPME, polar carbowax fibers were used and provided sufficient extraction recoveries. However, SPME was shown to be less sensitive compared with SPE [38, 39]. Carbon nanotubes (CNTs) gained great interest [35], especially multi-walled CNTs with multiple layers of graphene [40] with their large surface-to-volume ratio and increased loadability. The extraction of non-polar, polar, and even ionic species is possible via both hydrophobic and ionic interactions [35], e.g., for the analysis of polar sulfonylurea herbicides in environmental water samples [41]. Recent SPME approaches also focused on a lower ecological impact [42, 43].
As well as SPME, microextraction by packed sorbent (MEPS) is an equivalent to SPE and can be expected to become a very promising sample preparation technique in the future for several reasons: it is fast and simple to use, it can be fully automated, it can cope with much smaller sample volumes than full-scale SPE (as small as 10 μl), which is of interest especially for the analysis of, e.g., plasma, urine, or biota samples like insects. MEPS sorbents can be used more than 100 times (even for complex samples). MEPS uses the same sorbents as conventional SPE in cartridges. Thus, downscaling of most existing SPE methods is possible. In MEPS, 1–4 mg sorbent is either inserted into the syringe (100–250 μl) as a plug or between the needle and the barrel in a cartridge. Sample extraction, enrichment, and clean-up are accomplished directly on the packed sorbent [44]. The number of extraction cycles can be increased by drawing and ejecting the sample through the needle into the syringe several times (draw-eject), leading to a higher recovery [45]. Another key aspect of MEPS is the small solvent volume used for the elution of the analytes, which makes it good choice for online coupling with LC [46] and CE [47] separations. Mostly, pharmaceuticals in wastewater were analyzed in environmental applications [48]. Morales-Cid et al. [47] demonstrated a new and innovative way to integrate MEPS into commercial CE equipment. This method provided automated sample clean-up and preconcentration from only a few microliters of sample, which is interesting in biota analysis. The robustness of the proposed technical implementation was demonstrated by the use of a (MEPS)-nonaqueous capillary electrophoresis (NACE)-MS method used to determine the very polar fluoroquinolones in urine. The detection limits (LODs) were in the range of 6.3–10.6 μg/l and absolute recoveries were in the range of 71–109%.
Dispersive liquid-liquid microextraction
As a mode of liquid-liquid extraction (LLE), dispersive liquid-liquid microextraction (DLLME) was introduced by Rezaee et al. [49] for the extraction and preconcentration of organic analytes from water samples. A dispersed liquid phase is used to facilitate extraction from mostly small volumes (tens of μl range) of extraction solvent by fast equilibration due to the increased interfacial surface area between sample and extraction solvent. Many combinations with other sample preparation methods like SPE, single-drop microextraction, or DLLME itself allowed the purification and enrichment of a broad range of analytes (pharmaceuticals, pesticides, personal care products) in different samples (environmental samples, food, biological fluids) [50]. For hydrophilic compounds, recoveries have to be further improved. So far, the main strategy for the extraction of vPICs is compensating poor recoveries by high enrichment factors [51]. However, optimization strategies such as using ionic liquids as extraction solvents and ultrasound assistance in the extraction process led to the successful extraction of the polar fluoroquinolones (7 out of 8 with log P < 1) from groundwater samples [52]. In order to enable automated methods, there are new approaches to omit the dispenser solvent which could eliminate the centrifugation step and enable higher throughput [53].
QuEChERS extraction
According to the current trend in analytical chemistry to develop environmentally friendly methods, QuEChERS extraction has become increasingly popular in many fields [54]. The extraction method was originally developed by Anastassiades in 2003 [55] to determine pesticides in fruits and vegetables. Since then, the method has been optimized and adjusted for different analytes and environmental matrices. For example, QuEChERS was used to extract pharmaceuticals from sewage and surface water [56], drinking water, treatment plant sludge [57], soil [58], invertebrates [59], and fish tissue [60, 61]. The method is based on LLE with mainly acetonitrile (partly also acetone or ethyl acetate) and water, often followed by a d-SPE for clean-up. Due to the miscibility of acetonitrile and water, a mixture of salts (NaCl and MgSO4) must be added to induce phase separation [55]. The ratio/volume of solvents and salts is used to optimize phase polarities and thus extraction efficiencies. For highly polar compounds, methanol can be added to enhance the recovery: for the quantification of quinolones and tetracyclines from fish tissue, an extraction solution based on a mixture of acetonitrile and methanol (75:25, v/v) resulted in relative recoveries ranging from 69 to 125% [62]. In contrast to food samples, the size and quantity of environmental biota samples is often limited. Therefore, the sample extraction techniques originally developed for food analysis have to be adapted. Often, miniaturization is necessary for invertebrate samples [59], e.g., carbamazepine and fluoxetine were quantified in single individuals of benthic invertebrates with relative recoveries > 85% [60]. Many non-polar and fatty compounds present in the mollusk matrix were removed using hexane as a third liquid phase during extraction. Overall, with its simplicity, environmental sustainability, and its compatibility with all relevant separation techniques, we suppose QuEChERS extraction to be increasingly applied in the future. However, we expect that not all vPICs can be extracted with acceptable efficiency.
Electric field-driven sample preparation
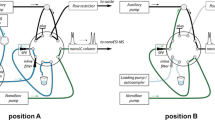
There are new approaches using electric fields as auxiliary force to accelerate mass transfer of ionic analytes during extraction. Among them, electromembrane extraction (EME) has probably gained highest attention so far due to its simple setup. Introduced in 2006 by Pedersen-Bjergaard et al. [63], the usually aqueous sample and acceptor solutions are separated by a membrane, which can either be a supported liquid membrane (SLM) or a polymer-imprinted membrane. Application of an electric field across the two compartments results in migration of analytes according to their charge and electrophoretic mobility (both in the donor and acceptor solution but also in the membrane). This strategy has three benefits: (1) by using a smaller acceptor solution volume, enrichment of the analytes occurs together with (2) a clean-up from non-charged and oppositely charged matrix compounds, and (3) selectivity tuning by the membrane composition is possible (see below). This approach is only applicable for the extraction of ionizable and ionic compounds from various liquid samples or extracts. High matrix tolerance and compatibility with common analytical techniques like LC, GC, and CE have already been demonstrated [64]. Selectivity regarding the polarity of compounds can be tuned with different membrane compositions. In 2014, for example, Koruni et al. [65] presented the simultaneous extraction of both acidic and basic drugs over a broad range of polarities using two SLMs of different composition. However, further improvements to also target highly polar charged compounds are necessary and are addressed in current research [66]. In principle, EME is very environmentally friendly and potentially automatable, so we expect increased attention in the future, especially for water analysis. For extracts of biota or soil samples, this method is not (yet) established as mostly organic solvents are used for extraction. One possibility may be to evaporate the organic solvent and reconstituting the residue in water. Further research needs to address shortcomings such as low speed, low degree of automation, sensitivity, and recovery. Most studies so far used optimized methods for selected analytes; they have not yet been implemented for screening purposes.
Trends in separation techniques
Chromatographic techniques
In this chapter, we focus on trends in chromatographic techniques especially useful for the analysis of vPICs or for a broad analyte coverage in environmental samples, such as HILIC, SFC, and MMLC. Their advantages, disadvantages, and application to environmental samples are elaborated.
Hydrophilic interaction liquid chromatography
In the last decade, HILIC-MS established itself as a valuable complementary approach to RPLC for the determination of highly hydrophilic, polar, and ionic compounds. This is also confirmed by the increase of HILIC applications [4] in various fields such as biology [67], food [68] and environmental science [24, 69]. Based on a combination of a polar stationary phase with a low aqueous and high organic content in the mobile phase, HILIC is able to improve chromatographic retention, resolution, and thus sensitivity for polar compounds [5], which may otherwise elute in or close to the void volume in RPLC. Further advantages over RPLC are higher applicable flow rates due the high organic content of the mobile phase and hence its lower viscosity. It is well suited for large volume injections of extracts with high organic content, often of interest in biota analysis. For the same reason, HILIC is also well compatible with online SPE as demonstrated by Fontanals et al. [16] with their first fully automated method based on online SPE coupled to HILIC-MS to determine a group of polar drugs and pharmaceuticals (log D (pH 7 ranging from − 1.8 to 1.38) in environmental water samples. The method had analyte recoveries near 100% and LODs ≤ 2 ng/l for most of the compounds. However, the high organic content of the mobile phase is a disadvantage when water samples are analyzed: the direct injection of large volumes of water as routinely done in RPLC (up to 100 μl) is not possible because of the high elution strength of water in HILIC [70]. Instead, dilution to ca. 80% acetonitrile is often required, significantly increasing LODs. In environmental analysis, HILIC was applied for the targeted analysis of polar herbicides [71], pesticides [72], and pharmaceuticals, such as antibiotics [17], drugs of abuse [16], cytostatics [73], antidiabetics [24, 74], and contrast agents [75, 76]. HILIC was successfully used as a complementary method to RPLC for the non-target screening of emerging polar organic compounds in wastewater [77]. To further extend the applicability of HILIC to a wider range of pharmaceutical compounds, various new HILIC stationary phases may be applied. Silica-based materials, in particular bare and amide-bonded silica, zwitterionic, and diol stationary phases, remain by far the most widely used stationary phases for HILIC in environmental applications with MS-compatible mobile phases [5]. In terms of green chemistry, HILIC is surely not advantageous due to the large amount of acetonitrile needed, necessitating a search for eco-friendlier alternatives. An example was given by dos Santos Pereira et al. [78], who demonstrated that biodegradable ethanol successfully replaced acetonitrile in the separation of vPICs. Another possibility to reduce the amount of organic solvents could be miniaturization of the analytical column, which is already a trend in bioanalysis [79]. However, this may be at the cost of a lowered sample loadability and is thus an option for biota but not for water analysis. Overall, we expect HILIC applications to increase as its polarity range almost ideally complements RPLC analysis (see Fig. 2), while the same equipment can be used. Multidimensional applications combining RPLC and HILIC are discussed in Section 2D applications.
Supercritical fluid chromatography
In recent years, SFC experienced a significant revival due to improvements in instrumentation, resulting in higher reliability and robustness [80]. Moreover, the advent of sub-2-μm particles and superficially porous particles in the stationary phases encouraged the use of UHPSFC. This gave rise to a further improvement of resolution and faster analyses, however, without the need for high pump pressure as encountered in ultra-high performance LC. Hyphenation to MS is increasingly used, which opened the way to new application fields such as bioanalysis, omics sciences, plant, food, and environmental analyses [80]. The mobile phase of modern SFC usually consists of compressed carbon dioxide, making it a “green method” [81]. Small volumes of modifiers such as methanol are added to tailor selectivity addressing different polarity ranges and optimize resolution. Regarding stationary phase chemistries, a large variety of stationary phases, from the most polar silica to the least polar well-endcapped or densely bonded alkyl-bonded silica, is now available, which enables to tailor selectivity [82].
In a seminal paper, two chromatographic separation strategies were compared, a serial RPLC-HILIC coupling vs. SFC [81]. For SFC, a zwitterionic HILIC column and a binary gradient of CO2 and 20 mM ammonium acetate in methanol (modifier) were used. Both systems were able to retain nearly all the 274 environmentally relevant compounds with very different polarities (log D (pH 7) ranging from − 7.71 to 7.67) providing a more comprehensive analysis than common RPLC-MS (see also Fig. 3). Such at least partially orthogonal methods can be used for cross-validation.
Plots of log D (pH 7) vs. retention time of standard compounds analyzed by RPLC-HILIC-TOF-MS (a) and SFC-TOF-MS (b). Very polar compounds (log D < − 2.5, blue rectangles) are mainly retained by HILIC in the RPLC-HILIC coupling, while non-polar compounds (log D > 2.0, red triangles) are exclusively retained by RPLC. Polar compounds are retained in both HILIC and RPLC, but retention in HILIC seems to be more likely with increased polarity. In SFC (b), non-polar compounds are retained less than very polar compounds. The retention patterns in the RPLC-HILIC coupling (a) show two groups that represent HILIC-(RT < 16 min) and RPLC-retained compounds (RT > 16 min). Compound log D increases with RT in RPLC, while the opposite occurs in HILIC. This retention behavior known from normal phase LC can partly be observed in SFC separations, too (b). Reprinted with permission from [81]. © 2020 American Chemical Society
Mixed-mode liquid chromatography
Mixed-mode stationary phases are gaining attention as more and more applications are published and new commercial columns appear on the market [83]. The main advantage of MMLC is that it provides more than one type of interaction between the stationary phase and the analytes, allowing the simultaneous determination of compounds with a wide range of physicochemical properties (e.g., ionic, basic, acidic and neutral chemicals, or hydrophilic and hydrophobic) in one run [84, 85]. The three most common types of MMLC are RP-ion exchange, HILIC-ion exchange, and RP-HILIC [86], which extends the application range of these methods but partially also combines their weaknesses and drawbacks [3]. Stationary phases are silica or polymer based; different hybrid and monolithic mixed-mode stationary phases are available [87]. The particle size of the current commercial sorbents is usually in the range of 3–7 μm and the stationary phases are synthesized on fully porous particles. In the near future, it can be expected that commercial sub-2-μm particles and superficially porous materials will be developed. This will lead to better separation efficiency and analysis time. Initially, mixed-mode columns were applied in bioanalysis, but their application was expanded to a variety of fields, including pharmaceutical analysis [88, 89], metabolism studies [90, 91], and recently also to environmental analysis [92,93,94,95,96,97]. González-Mariño et al. [97] described the first application of RP-ion exchange mixed-mode LC for the determination of drugs of abuse in wastewater. MMLC was shown to be a good alternative to traditional RPLC and HILIC for the separation of vPICs. Montes et al. [96] used a targeted MMLC-MS for the quantitative determination of 23 persistent and mobile organic contaminants (log D (pH 7) ranging from − 3.74 to 3.36) in surface and drinking water samples. In comparison with RPLC, MMLC proved superior in both retention and peak shape for ionic compounds, while also performing well for neutrals. In contrast to HILIC, the mobile phase of MMLC has a high aqueous content (as in RPLC); therefore, it is well suited for the direct injection of water samples. Further advantages of MMLC include the higher flexibility in adjusting separation selectivity and the possibility to use the mixed-mode sorbent in different elution modes [87]. MMLC can thus be an alternative to 2D applications. Besides many benefits, there are also some drawbacks related with these stationary phases. For example, when more than one type of interaction is active, asymmetric signals may evolve leading to reduced separation efficiency. Furthermore, method development has to consider the different separation modes and counteracting effects of optimization parameters may complicate this process. This necessitates compromises in separation performance, when analytes with a broad range of physicochemical characteristics are included. Additionally, Zahn et al. [3] indicated some limitations for RP-ion exchange columns, which are similar to the two retention mechanisms: poor retention of polar but neutral compounds and additionally the reliance on high electrolyte concentrations in the eluent to elute strongly retained ionic compounds which may cause detrimental effects in ESI [95]. However, with new mixed-mode stationary phases appearing on the market, MMLC-MS can be tailored to the polarity range of interest for specific questions and may be ideal for targeted or semi-non-target screening of classes of compounds.
Ion chromatography
IC as a special mode of LC has already proven to be a powerful tool for the analysis of ionic compounds, mostly inorganic anions and cations in water samples and biological fluids [98] as well as small organic compounds in a broad range of sample types such as milk [99] or biological fluids [100], but also environmental samples [101]. For reliable IC analysis, e.g., of wastewater treatment effluents, sample preparation is often mandatory given the high matrix (salt) load of many environmental samples (see section Sample preparation in environmental matrices). Zakaria et al. [102] listed three major strategies for sample pretreatment: (1) samples may be purified by suitable ion exchange resins, e.g., silver cartridges for the removal of chloride [103]; (2) use of sensitive methods like inductively coupled plasma (ICP)-MS after strongly diluting the samples [104]; or (3) use of high capacity columns to increase loadability [105]. We see an additional possibility in the use of electric fields (see section Electric field-driven sample preparation) as both sample preparation and separation methods require charged analytes. An EME-IC system for the analysis of inorganic anions was presented [106]. Another option is the use of 2D-IC techniques [107] (see also section 2D applications), as applied, e.g., for the analysis of bromate, chlorate, and five haloacetic acids in water [108]. Similar trends as in LC towards higher pressure and smaller particle sizes (see above) can be expected for IC to reach higher separation efficiencies and lower detection limits.
The successful analysis of acidic pharmaceuticals in biological fluids using IC with sensitive fluorescence detection after labeling was reported by Muhammad et al. [100]. A further increase in sensitivity is obtained when coupling IC with MS [109] upon further improvements of the interfaces. Recently, Stoll [110] summarized the state of the art of IC-MS. Limitations of current IC(-MS) instrumentation especially for (semi-)screening methods include the rather long column equilibration times and possible signal quenching by interactions of analytes with suppressors [111]. Publications in the field show a limited choice of suitable eluents as they must have a suitably high elution strength while being MS-compatible. Limitations for both ICP-MS and electrospray ionization (ESI)-MS were reported, describing decreased sensitivity in ICP-MS by using carbonate/bicarbonate eluents without a suppressor as a result of contamination of skimmer cones [112] and a general signal suppression in ESI due to high concentrations of eluents [113]. Overall, improvements with optimized elution buffers [113] as well as instrumentation and system design [114] are still subject to current research. Another approach is the reduction of the column inner diameters to, e.g., 0.4 mm [115], or to use an organic make-up solvent such as acetonitrile or isopropanol [116] to increase MS sensitivity.
The two major MS interfaces used for IC are ICP and ESI. Whereas IC-ICP-MS is suitable for elemental analysis in a broad range of different samples including, e.g., organic compounds with halogen substituents, IC-ESI-MS enables to analyze low molecular mass organic compounds. The general advantages of ICP-MS for different environmentally relevant pollutant species (e.g., pharmaceuticals, flame retardants, pesticides,…) were worked out nicely by Pfröfrock and Prange [117]. With ICP-MS, the detection and selective screening of different molecules containing covalently bound (hetero-)elements such as phosphorus, arsenic, or halogens is possible. Sacher et al. [118], for example, developed an IC-ICP-MS method for iodinated X-ray contrast agents in surface water. Whereas a full non-target screening cannot be reached with IC-ICP-MS, it may be possible to target sulfur species in a screening approach, interesting to study the transformation of pharmaceuticals and pesticides to sulfur- or phosphor-containing species, e.g., metolachlor with charged transformation products [119]. In addition, quantification is possible without internal standards, if the stoichiometry is known [120, 121]. Especially the complementary combination of ICP-MS for screening and ESI-MS for identification is an interesting approach [122, 123], which will likely be applied more often in the future.
Speciation analysis by IC-ICP-MS for environmental analysis was summarized in a book edited by Michalski [124]. For biota, the potential of IC-ICP-MS has already been exploited for the detection of arsenic and selenium species in fish tissue by Reyes et al. [125] but to our knowledge not yet for pharmaceuticals. IC-ICP-MS with its high selectivity will be of future interest especially for targeted or selective screening approaches in samples of high matrix load such as biota samples. Generally, this can enable a broader screening of vPICs and their transformation products in biota, especially for compounds with iodine or bromine, which are rare in biological samples.
Successful applications of IC-ESI-MS for the analysis of environmental samples show the increasing interest for the analysis of pesticides in food samples. Polar pesticides being charged over a broad pH range like glyphosate, ethephon [113], or paraquat [126] were successfully analyzed in food samples by IC-ESI-MS/MS. In environmental samples, this technique is mostly applied for the analysis of inorganic anions and cations as well as small organic acids [127]. However, in recent years, there has been progress in the analysis of polar pesticides in environmental waters by IC-MS. Glyphosate and two of its metabolites (all log P < − 2) were successfully analyzed in surface and drinking waters reaching LODs in the low ng/l range [128]. The analysis of haloacetic acids in drinking water was also successful [129], which is very fortunate given the new regulations in the European Union (Water Framework Directive, WFD, 2000/60/EC) requiring the sensitive analysis of these compounds. We expect further applications for the analysis of wastewater micropollutants and semi-targeted screenings, as they already proved valuable in forensic analysis of gunshot residues [130].
Electromigrative separation techniques
Electromigrative separation techniques like CE are an alternative to IC for the analysis of ionic or ionizable micropollutants. The general applicability and potential of CE in environmental analysis was shown in numerous applications of pesticide analysis summarized in recent reviews [131, 132]. The use and state of the art of CE analysis of various pharmaceuticals in environmental samples was described by Hamdan in 2017 [133].
Whereas CE is already well established for the analysis of small molecules in human body fluids [134], the use of electromigration separation techniques for the analysis of vPICs in environmental samples is still challenging. The loadability for the commonly used capillaries of only 50 μm inner diameter is limited and often gives rise to relatively high detection limits. This can be overcome improving sample preparation and enrichment methods dedicated to CE, e.g., sample preparation using online or offline SP(M)E (see Hamdan [133]). Online enrichment is possible via large volume sample stacking [135], field-amplified sample injection [136], and (transient) isotachophoresis [137, 138]. Offline electro-driven enrichment techniques like EME are gaining more and more popularity [139] and their hyphenation with CE for the analysis of environmental samples was reported [140] (see also section Electric field-driven sample preparation). There are optimized CE-diode array detection (DAD) methods which reach detection limits in the medium ng/l range [141]; however, these are rather exceptional. More often, MS [142] and sometimes fluorescence detection [143], mostly after derivatization [144], are used.
As for LC, MS is the detection method of choice for non-target screenings, but today, most CE-MS methods are still established in research laboratories only, although (1) there are less restrictions compared with IC-MS hyphenation (e.g., eluent composition) and (2) ESI-MS-interfaces compatible with both LC and CE separation systems are available. In addition, solvent consumption is low compared with chromatographic techniques, which makes CE and CE-MS interesting as “green” alternatives. Although many CE(-MS) methods barely reach detection limits below 1 μg/l [133], there are already promising approaches combining CE-MS with different sample preparation methods [145, 146]. Additionally, an enrichment-free approach was recently published by Höcker et al. [147] enabling the analysis of anionic micropollutants like haloacetic acids and halomethanesulfonic acids in drinking water, reaching LOQs between 30 and 500 ng/l.
CE was shown to be well suited for the analysis of biological fluids, but there are only few examples for its use in biota analysis: Deng et al. [148] analyzed the polar antibiotic tetracycline in crucian carp muscle using electrochemiluminescence detection. Sun et al. [149] analyzed sulfonamides in shrimp, sardine, and anchovy with CE-DAD. Both methods reached detection limits in the μg/l range. Coupling CE to sensitive MS detection even led to detection limits in the upper ng/l (low ng/g) range for the analysis of the antidiabetic drug metformin in fish without sample preconcentration [150], requiring only small sample volumes, which enabled analyzing individual fish organs.
2D applications
The aims of 2D approaches are: (1) maximum sample information, the comprehensive analysis of micropollutants over a broad range of physicochemical characteristics (today mostly polarity) for non-target analysis. For this, RPLC-HILIC has already been established by Bieber et al. [81]. Similar approaches are well known in peptide analysis [151]. (2) With 2D techniques, peak capacity can be increased using ideally orthogonal separation selectivity in the second dimension. The two separation dimensions can either be coupled consecutively or by heart-cutting selected zones to enhance resolution and minimize matrix effects, e.g., comprehensive couplings of IC×RPLC [152] or IC×CE [153]. Chromatographic and electromigration separations were successfully combined in bioanalysis [154]. (3) The first dimension may also be used as a clean-up step as shown for IC-IC by Zakaria et al. [102] using the first dimension to eliminate inorganic salt components prior to the quantification of bromate in sea water samples. Although LODs of only 60 μg/l of bromate were obtained, this approach might also be interesting for the analysis of pharmaceuticals in environmental samples containing high concentrations of salt using IC with sensitive MS detection. Possibly, additional preconcentration and desalting steps, e.g., using SPE, are necessary to meet the required detection limits.
It has to be noted that comprehensive applications are generally limited by the first dimension with regard to polarity or charge. Improved instrumental setups for hyphenation and testing some of the new materials available today (e.g., MMLC stationary phases, see section Mixed-mode liquid chromatography) can result in increased matrix tolerance and higher sensitivity for complex samples. Stevenson et al. [155] developed an offline 2D-LC separation of a β-lactoglobulin tryptic digest with the same mixed-mode stationary phase in both dimensions. In the first dimension, the mobile phase pH was 7, while it was adjusted to pH 2 in the second dimension evoking different separation mechanisms. Greater separation efficiency was observed for the mixed-mode column in comparison with classical C18, thereby providing larger peak capacity than the C18 column. Similar applications will be of interest in environmental analysis. With the advent of commercial equipment for 2D applications, its application in environmental analysis will surely rise, though merely for research than for routine analysis. A major limitation is the data evaluation, where further software developments are required.
Summary on future perspectives
The impressive development of new analytical methods enabled the analysis of many vPICs in environmental samples. These developments comprise new materials, instrumentation, miniaturization, and automation both for sample preparation and separation. In this article, we showed that some current trends are partly due to a revival of methods such as SFC, partly due to the use of developments from other fields such as QuEChERS extraction from food science, and MMLC from pharmaceutical analysis or some 2D applications from bioanalysis. In the following, we want to summarize future perspectives.
Due to the high demand for improved limits of detection and selectivity, many new SPE materials for vPICs were commercialized but none of the sorbents available can cover the whole range of compounds. Especially, the retention of very polar analytes is still an issue. Both mixed-bed SPE and EC were shown to also cover very polar compounds. However, only a few mixed-bed cartridges are commercially available and EC does not include a sample clean-up. New materials like carbon-based nanomaterials can be used to improve the extraction efficiency but they are mostly suitable only for target analysis or specialized applications.
For ionic and ionizable compounds, we expect electro-based enrichment techniques to find applications beyond inorganic ions [106], especially for the analysis of ionic or ionizable pharmaceuticals in environmental samples. For electro-based enrichment techniques, further effort is required to achieve an acceptable degree of sample throughput by automation and miniaturization for both monitoring strategies and (eco)toxicological research. For matrix removal, these developments are believed to be prioritized compared with online sample pretreatment and 2D approaches.
We are convinced that HILIC will complement RPLC routine analysis as the expertise for liquid chromatography is high and implementation is straightforward. Future research will show which stationary phases will become the gold standard in the future. SFC research is very active and due to the development of modern instrumentation, reproducible results are obtained today. In the pharmaceutical industry, SFC can already replace RPLC as a routine technique for the separation of chiral substances on a preparative scale [156]. In environmental analysis, the actual potential of SFC can only be estimated, since applications are still rare. However, it is anticipated that SFC may partly replace RPLC, as a wider polarity range is covered, even when compared with 2D techniques such as RPLC-HILIC. SFC may then become a suitable tool also for monitoring campaigns. Thus, it will be interesting to see if 2D techniques will find more application in environmental analysis as is currently seen in bioanalysis, especially peptide analysis. While instrumental developments led to the commercial availability of dedicated instrumentation for comprehensive 2D-LC, software tools still show limitations. Possibly, MMLC will open up new possibilities in 2D separation methods, when used as a first dimension with a broad analyte coverage. We see a great potential in MMLC since it improves the separation of vPICs (both basic and acidic) while also retaining neutral chemicals. MMLC thus combines the benefits of different chromatographic modes. Manufacturers of stationary phases also reacted with increased production of MMLC stationary phases.
Finally, attractive alternatives for ionizable compounds, also with regard to “green chemistry,” are IC-MS and CE-MS. However, environmental CE-MS applications afford more research for analyte enrichment and IC-MS applications for improved stationary phases for a greater choice of MS-compatible eluents. It is difficult to judge if CE-MS with a lower instrumental requirement than IC-MS will be implemented in laboratories dominated by chromatographic expertise.
In our opinion, the gap described in 2016 by Reemtsma et al. [2] for the analysis of persistent and mobile organic compounds still exists. With the latest developments in analytical chemistry, filling this gap seems feasible. However, most methods which are well suited have not yet left laboratory scale applications so that their applicability in monitoring campaigns remains to be shown. Establishing new analytical processes in general is a time-consuming process for laboratories. Acceptance by the community and regulatory bodies is another hindrance. Often, modified or optimized established methods are accepted more quickly and are easier to implement with regard to existing instrumentation and expertise. However, with the slow implementation of new methodology, chances are missed to increase efficiency, scope, matrix tolerance, environmental friendliness, etc.
All in all, however, we have to keep in mind that no method is capable to fulfill all requirements from environmental science and (eco)toxicology given the wealth of compounds with their broad range of physicochemical characteristics. New strategies have to find compromises between different needs, e.g., analyte coverage, matrix compatibility, or analysis time and costs. Additionally, other drivers like automation, miniaturization in biota analysis, and organic solvent consumption have to be considered for current and future developments for the analysis of vPICs.
References
Richardson ML, Bowron JM. The fate of pharmaceutical chemicals in the aquatic environment. J Pharm Pharmacol. 1985;37(1):1–12.
Reemtsma T, Berger U, Arp HPH, Gallard H, Knepper TP, Neumann M, et al. Mind the gap: persistent and mobile organic compounds - water contaminants that slip through. Environ Sci Technol. 2016;50(19):10308–15.
Zahn D, Neuwald IJ, Knepper TP. Analysis of mobile chemicals in the aquatic environment-current capabilities, limitations and future perspectives. Anal Bioanal Chem. 2020. https://doi.org/10.1007/s00216-020-02520-z.
van Nuijs AL, Tarcomnicu I, Covaci A. Application of hydrophilic interaction chromatography for the analysis of polar contaminants in food and environmental samples. J Chromatogr A. 2011;1218(35):5964–74.
Salas D, Borrull F, Fontanals N, Marcé RM. Hydrophilic interaction liquid chromatography coupled to mass spectrometry-based detection to determine emerging organic contaminants in environmental samples. Trends Anal Chem. 2017;94:141–9.
Schmidt TC. Recent trends in water analysis triggering future monitoring of organic micropollutants. Anal Bioanal Chem. 2018;410(17):3933–41.
Zhang Q, Yang F-Q, Ge L, Hu Y-J, Xia Z-N. Recent applications of hydrophilic interaction liquid chromatography in pharmaceutical analysis. J Sep Sci. 2017;40(1):49–80.
Zahn D, Reemtsma T, Berger U, Knepper TP. Quellen und Verbreitung persistenter und mobiler organischer Stoffe in Roh- und Trinkwasser. Vom Wasser. 2019;117(4):125–9.
Bicker W, Lämmerhofer M, Lindner W. Mixed-mode stationary phases as a complementary selectivity concept in liquid chromatography–tandem mass spectrometry-based bioanalytical assays. Anal Bioanal Chem. 2008;390(1):263–6.
Wasik A, Kot-Wasik A, Namiesnik J. New trends in sample preparation techniques for the analysis of the residues of pharmaceuticals in environmental samples. Curr Anal Chem. 2016;12(4):280–302.
Núñez M, Borrull F, Pocurull E, Fontanals N. Sample treatment for the determination of emerging organic contaminants in aquatic organisms. Trends Anal Chem. 2017;97:136–45.
Rossmann J, Schubert S, Gurke R, Oertel R, Kirch W. Simultaneous determination of most prescribed antibiotics in multiple urban wastewater by SPE-LC-MS/MS. J Chromatogr B. 2014;969:162–70.
David V, Galaon T, Bacalum E. Sample enrichment by solid-phase extraction for reaching parts per quadrillion levels in environmental analysis. Chromatographia. 2019;82(8):1139–50.
Ribeiro C, Ribeiro AR, Maia AS, Goncalves VM, Tiritan ME. New trends in sample preparation techniques for environmental analysis. Crit Rev Anal Chem. 2014;44(2):142–85.
Armenta S, Garrigues S, de la Guardia M. Green analytical chemistry. Trends Anal Chem. 2008;27(6):497–511.
Fontanals N, Marce RM, Borrull F. On-line solid-phase extraction coupled to hydrophilic interaction chromatography-mass spectrometry for the determination of polar drugs. J Chromatogr A. 2011;1218(35):5975–80.
Rossmann J, Schubert S, Gurke R, Oertel R, Kirch W. Simultaneous determination of most prescribed antibiotics in multiple urban wastewater by SPE-LC-MS/MS. J Chromatogr B. 2014;969:162–70.
Gomez MJ, Petrovic M, Fernandez-Alba AR, Barcelo D. Determination of pharmaceuticals of various therapeutic classes by solid-phase extraction and liquid chromatography-tandem mass spectrometry analysis in hospital effluent wastewaters. J Chromatogr A. 2006;1114(2):224–33.
Barron L, Tobin J, Paull B. Multi-residue determination of pharmaceuticals in sludge and sludge enriched soils using pressurized liquid extraction, solid phase extraction and liquid chromatography with tandem mass spectrometry. J Environ Monit. 2008;10(3):353–61.
Huerta B, Jakimska A, Gros M, Rodriguez-Mozaz S, Barcelo D. Analysis of multi-class pharmaceuticals in fish tissues by ultra-high-performance liquid chromatography tandem mass spectrometry. J Chromatogr A. 2013;1288:63–72.
Boulard L, Dierkes G, Ternes T. Utilization of large volume zwitterionic hydrophilic interaction liquid chromatography for the analysis of polar pharmaceuticals in aqueous environmental samples: benefits and limitations. J Chromatogr A. 2018;1535:27–43.
Chau HTC, Kadokami K, Ifuku T, Yoshida Y. Development of a comprehensive screening method for more than 300 organic chemicals in water samples using a combination of solid-phase extraction and liquid chromatography-time-of-flight-mass spectrometry. Environ Sci Pollut Res Int. 2017;24(34):26396–409.
Carneiro MC, Puignou L, Galceran MT. Comparison of silica and porous graphitic carbon as solid-phase extraction materials for the analysis of cationic herbicides in water by liquid chromatography and capillary electrophoresis. Anal Chim Acta. 2000;408(1–2):263–9.
Scheurer M, Sacher F, Brauch HJ. Occurrence of the antidiabetic drug metformin in sewage and surface waters in Germany. J Environ Monit. 2009;11(9):1608–13.
Köke N, Zahn D, Knepper TP, Frömel T. Multi-layer solid-phase extraction and evaporation-enrichment methods for polar organic chemicals from aqueous matrices. Anal Bioanal Chem. 2018;410(9):2403–11.
Kern S, Fenner K, Singer HP, Schwarzenbach RP, Hollender J. Identification of transformation products of organic contaminants in natural waters by computer-aided prediction and high-resolution mass spectrometry. Environ Sci Technol. 2009;43(18):7039–46.
Mechelke J, Longree P, Singer H, Hollender J. Vacuum-assisted evaporative concentration combined with LC-HRMS/MS for ultra-trace-level screening of organic micropollutants in environmental water samples. Anal Bioanal Chem. 2019;411(12):2555–67.
Rodriguez-Mozaz S, Lopez de Alda MJ, Barcelo D. Advantages and limitations of on-line solid phase extraction coupled to liquid chromatography-mass spectrometry technologies versus biosensors for monitoring of emerging contaminants in water. J Chromatogr A. 2007;1152(1–2):97–115.
Huntscha S, Singer HP, McArdell CS, Frank CE, Hollender J. Multiresidue analysis of 88 polar organic micropollutants in ground, surface and wastewater using online mixed-bed multilayer solid-phase extraction coupled to high performance liquid chromatography-tandem mass spectrometry. J Chromatogr A. 2012;1268:74–83.
Cai Q, Zhang L, Zhao P, Lun X, Li W, Guo Y, et al. A joint experimental-computational investigation: metal organic framework as a vortex assisted dispersive micro-solid-phase extraction sorbent coupled with UPLC-MS/MS for the simultaneous determination of amphenicols and their metabolite in aquaculture water. Microchem J. 2017;130:263–70.
He Z, Liu D, Zhou Z, Wang P. Ionic-liquid-functionalized magnetic particles as an adsorbent for the magnetic SPE of sulfonylurea herbicides in environmental water samples. J Sep Sci. 2013;36(19):3226–33.
Luo YB, Shi ZG, Gao Q, Feng YQ. Magnetic retrieval of graphene: extraction of sulfonamide antibiotics from environmental water samples. J Chromatogr A. 2011;1218(10):1353–8.
Arthur C, Pawliszyn J. The principle and application of solid—phase micro—extraction (SPME). Anal Chem. 1990;62:2145–8.
Li J, Wang Y-B, Li K-Y, Cao Y-Q, Wu S, Wu L. Advances in different configurations of solid-phase microextraction and their applications in food and environmental analysis. Trends Anal Chem. 2015;72:141–52.
Souza-Silva ÉA, Jiang R, Rodríguez-Lafuente A, Gionfriddo E, Pawliszyn J. A critical review of the state of the art of solid-phase microextraction of complex matrices I. Environmental analysis. Trends Anal Chem. 2015;71:224–35.
Piri-Moghadam H, Ahmadi F, Pawliszyn J. A critical review of solid phase microextraction for analysis of water samples. Trends Anal Chem. 2016;85:133–43.
Aresta A, Bianchi D, Calvano CD, Zambonin CG. Solid phase microextraction--liquid chromatography (SPME-LC) determination of chloramphenicol in urine and environmental water samples. J Pharm Biomed Anal. 2010;53(3):440–4.
López-Blanco MC, Cancho-Grande B, Simal-Gándara J. Comparison of solid-phase extraction and solid-phase microextraction for carbofuran in water analyzed by high-performance liquid chromatography–photodiode-array detection. J Chromatogr A. 2002;963(1–2):117–23.
Balakrishnan VK, Terry KA, Toito J. Determination of sulfonamide antibiotics in wastewater: a comparison of solid phase microextraction and solid phase extraction methods. J Chromatogr A. 2006;1131(1–2):1–10.
Suarez B, Santos B, Simonet BM, Cardenas S, Valcarcel M. Solid-phase extraction-capillary electrophoresis-mass spectrometry for the determination of tetracyclines residues in surface water by using carbon nanotubes as sorbent material. J Chromatogr A. 2007;1175(1):127–32.
Zhou Q, Xiao J, Wang W. Comparison of multiwalled carbon nanotubes and a conventional absorbent on the enrichment of sulfonylurea herbicides in water samples. Anal Sci. 2007;23(2):189–92.
Carasek E, Morés L, Merib J. Basic principles, recent trends and future directions of microextraction techniques for the analysis of aqueous environmental samples. Trends Environ Anal Chem. 2018;19(July):e00060.
Soares da Silva Burato J, Vargas Medina DA, de Toffoli AL, Vasconcelos Soares Maciel E, Mauro Lancas F. Recent advances and trends in miniaturized sample preparation techniques. J Sep Sci. 2020;43(1):202–25.
Yang L, Said R, Abdel-Rehim M. Sorbent, device, matrix and application in microextraction by packed sorbent (MEPS): a review. J Chromatogr B. 2017;1043:33–43.
Pereira JAM, Goncalves J, Porto-Figueira P, Figueira JA, Alves V, Perestrelo R, et al. Current trends on microextraction by packed sorbent - fundamentals, application fields, innovative improvements and future applications. Analyst. 2019;144(17):5048–74.
Saracino MA, Lazzara G, Prugnoli B, Raggi MA. Rapid assays of clozapine and its metabolites in dried blood spots by liquid chromatography and microextraction by packed sorbent procedure. J Chromatogr A. 2011;1218(16):2153–9.
Morales-Cid G, Cardenas S, Simonet BM, Valcarcel M. Fully automatic sample treatment by integration of microextraction by packed sorbents into commercial capillary electrophoresis-mass spectrometry equipment: application to the determination of fluoroquinolones in urine. Anal Chem. 2009;81(8):3188–93.
Prieto A, Schrader S, Moeder M. Determination of organic priority pollutants and emerging compounds in wastewater and snow samples using multiresidue protocols on the basis of microextraction by packed sorbents coupled to large volume injection gas chromatography–mass spectrometry analysis. J Chromatogr A. 2010;1217(38):6002–11.
Rezaee M, Assadi Y, Milani Hosseini MR, Aghaee E, Ahmadi F, Berijani S. Determination of organic compounds in water using dispersive liquid-liquid microextraction. J Chromatogr A. 2006;1116(1–2):1–9.
Sajid M, Alhooshani K. Dispersive liquid-liquid microextraction based binary extraction techniques prior to chromatographic analysis: a review. Trends Anal Chem. 2018;108:167–82.
Nojavan Y, Kamankesh M, Shahraz F, Hashemi M, Mohammadi A. Ion pair-based dispersive liquid-liquid microextraction followed by high performance liquid chromatography as a new method for determining five folate derivatives in foodstuffs. Talanta. 2015;137:31–7.
Vazquez MM, Vazquez PP, Galera MM, Garcia MD. Determination of eight fluoroquinolones in groundwater samples with ultrasound-assisted ionic liquid dispersive liquid-liquid microextraction prior to high-performance liquid chromatography and fluorescence detection. Anal Chim Acta. 2012;748:20–7.
Sereshti H, Khorram P, Nouri N. Recent trends in replacement of disperser solvent in dispersive liquid-liquid microextraction methods. Sep Purif Rev. 2018;48(2):159–78.
Bruzzoniti MC, Checchini L, De Carlo RM, Orlandini S, Rivoira L, Del Bubba M. QuEChERS sample preparation for the determination of pesticides and other organic residues in environmental matrices: a critical review. Anal Bioanal Chem. 2014;406(17):4089–116.
Anastassiades M, Scherbaum E, Bertsch D Validation of a simple and rapid multiresidue method (QuEChERS) and its implementation in routine pesticide analysis. In: MGPR Symposium: Aix en Provence, France, 2003.
Kachhawaha AS, Nagarnaik PM, Jadhav M, Pudale A, Labhasetwar PK, Banerjee K. Optimization of a modified QuEChERS method for multiresidue analysis of pharmaceuticals and personal care products in sewage and surface water by LC-MS/MS. J AOAC Int. 2017;100(3):592–7.
Cerqueira MBR, Guilherme JR, Caldas SS, Martins ML, Zanella R, Primel EG. Evaluation of the QuEChERS method for the extraction of pharmaceuticals and personal care products from drinking-water treatment sludge with determination by UPLC-ESI-MS/MS. Chemosphere. 2014;107:74–82.
Braganca I, Placido A, Paiga P, Domingues VF, Delerue-Matos C. QuEChERS: a new sample preparation approach for the determination of ibuprofen and its metabolites in soils. Sci Total Environ. 2012;433:281–9.
Wicht A-J (2018) Development and application of new analytical methods based on chromatographic and electrophoretic separations to assess the environmental behavior of anthropogenic pollutants and their uptake in fungi and chironomids. Universitätsbibliothek Tübingen,
Berlioz-Barbier A, Baudot R, Wiest L, Gust M, Garric J, Cren-Olive C, et al. MicroQuEChERS-nanoliquid chromatography-nanospray-tandem mass spectrometry for the detection and quantification of trace pharmaceuticals in benthic invertebrates. Talanta. 2015;132:796–802.
Núñez M, Borrull F, Fontanals N, Pocurull E. Determination of pharmaceuticals in bivalves using QuEChERS extraction and liquid chromatography-tandem mass spectrometry. Anal Bioanal Chem. 2015;407(13):3841–9.
Lopes RP, Reyes RC, Romero-Gonzalez R, Vidal JL, Frenich AG. Multiresidue determination of veterinary drugs in aquaculture fish samples by ultra high performance liquid chromatography coupled to tandem mass spectrometry. J Chromatogr B. 2012;895-896:39–47.
Pedersen-Bjergaard S, Rasmussen KE. Electrokinetic migration across artificial liquid membranes. New concept for rapid sample preparation of biological fluids. J Chromatogr A. 2006;1109(2):183–90.
Oedit A, Ramautar R, Hankemeier T, Lindenburg PW. Electroextraction and electromembrane extraction: advances in hyphenation to analytical techniques. Electrophoresis. 2016;37(9):1170–86.
Koruni MH, Tabani H, Gharari H, Fakhari AR. An all-in-one electro-membrane extraction: development of an electro-membrane extraction method for the simultaneous extraction of acidic and basic drugs with a wide range of polarities. J Chromatogr A. 2014;1361:95–9.
Drouin N, Rudaz S, Schappler J. Sample preparation for polar metabolites in bioanalysis. Analyst. 2017;143(1):16–20.
Oertel R, Neumeister V, Kirch W. Hydrophilic interaction chromatography combined with tandem-mass spectrometry to determine six aminoglycosides in serum. J Chromatogr A. 2004;1058(1):197–201.
Valette JC, Demesmay C, Rocca JL, Verdon E. Separation of tetracycline antibiotics by hydrophilic interaction chromatography using an amino-propyl stationary phase. Chromatographia. 2004;59(1):55–60.
Nguyen HP, Schug KA. The advantages of ESI-MS detection in conjunction with HILIC mode separations: fundamentals and applications. J Sep Sci. 2008;31(9):1465–80.
Seitz W, Schulz W, Weber WH. Novel applications of highly sensitive liquid chromatography/mass spectrometry/mass spectrometry for the direct detection of ultra-trace levels of contaminants in water. Rapid Commun Mass Spectrom. 2006;20(15):2281–5.
Fauvelle V, Mazzella N, Morin S, Moreira S, Delest B, Budzinski H. Hydrophilic interaction liquid chromatography coupled with tandem mass spectrometry for acidic herbicides and metabolites analysis in fresh water. Environ Sci Pollut Res Int. 2015;22(6):3988–96.
Hayama T, Yoshida H, Todoroki K, Nohta H, Yamaguchi M. Determination of polar organophosphorus pesticides in water samples by hydrophilic interaction liquid chromatography with tandem mass spectrometry. Rapid Commun Mass Spectrom. 2008;22(14):2203–10.
Kovalova L, McArdell CS, Hollender J. Challenge of high polarity and low concentrations in analysis of cytostatics and metabolites in wastewater by hydrophilic interaction chromatography/tandem mass spectrometry. J Chromatogr A. 2009;1216(7):1100–8.
van Nuijs AL, Tarcomnicu I, Simons W, Bervoets L, Blust R, Jorens PG, et al. Optimization and validation of a hydrophilic interaction liquid chromatography-tandem mass spectrometry method for the determination of 13 top-prescribed pharmaceuticals in influent wastewater. Anal Bioanal Chem. 2010;398(5):2211–22.
Sordet M, Bulete A, Vulliet E. A rapid and easy method based on hydrophilic interaction chromatography coupled with tandem mass spectrometry (HILIC-MS/MS/MS) to quantify iodinated X-ray contrast in wastewaters. Talanta. 2018;190:480–6.
Lindner U, Lingott J, Richter S, Jiang W, Jakubowski N, Panne U. Analysis of gadolinium-based contrast agents in tap water with a new hydrophilic interaction chromatography (ZIC-cHILIC) hyphenated with inductively coupled plasma mass spectrometry. Anal Bioanal Chem. 2015;407(9):2415–22.
Gago-Ferrero P, Schymanski EL, Bletsou AA, Aalizadeh R, Hollender J, Thomaidis NS. Extended suspect and non-target strategies to characterize emerging polar organic contaminants in raw wastewater with LC-HRMS/MS. Environ Sci Technol. 2015;49(20):12333–41.
dos Santos PA, David F, Vanhoenacker G, Sandra P. The acetonitrile shortage: is reversed HILIC with water an alternative for the analysis of highly polar ionizable solutes? J Sep Sci. 2009;32(12):2001–7.
Hendrickx S, Adams E, Cabooter D. Recent advances in the application of hydrophilic interaction chromatography for the analysis of biological matrices. Bioanalysis. 2015;7(22):2927–45.
Pilařová V, Plachká K, Khalikova MA, Svec F, Nováková L. Recent developments in supercritical fluid chromatography – mass spectrometry: is it a viable option for analysis of complex samples? Trends Anal Chem. 2019;112:212–25.
Bieber S, Greco G, Grosse S, Letzel T. RPLC-HILIC and SFC with mass spectrometry: polarity-extended organic molecule screening in environmental (water) samples. Anal Chem. 2017;89(15):7907–14.
West C. Current trends in supercritical fluid chromatography. Anal Bioanal Chem. 2018;410(25):6441–57.
Ordonez EY, Benito Quintana J, Rodil R, Cela R. Computer assisted optimization of liquid chromatographic separations of small molecules using mixed-mode stationary phases. J Chromatogr A. 2012;1238:91–104.
Walshe M, Kelly MT, Smyth MR, Ritchie H. Retention studies on mixed-mode columns in high-performance liquid chromatography. J Chromatogr A. 1995;708(1):31–40.
Kazarian AA, Nesterenko PN, Soisungnoen P, Burakham R, Srijaranai S, Paull B. Comprehensive analysis of pharmaceutical products using simultaneous mixed-mode (ion-exchange/reversed-phase) and hydrophilic interaction liquid chromatography. J Sep Sci. 2014;37(16):2138–44.
Zhang L, Dai Q, Qiao X, Yu C, Qin X, Yan H. Mixed-mode chromatographic stationary phases: recent advancements and its applications for high-performance liquid chromatography. Trends Anal Chem. 2016;82:143–63.
Sykora D, Rezanka P, Zaruba K, Kral V. Recent advances in mixed-mode chromatographic stationary phases. J Sep Sci. 2019;42(1):89–129.
Strege MA, Stevenson S, Lawrence SM. Mixed-mode anion-cation exchange/hydrophilic interaction liquid chromatography-electrospray mass spectrometry as an alternative to reversed phase for small molecule drug discovery. Anal Chem. 2000;72(19):4629–33.
Martin F, Aerts A, Ahrén D, Brun A, Danchin EGJ, Duchaussoy F, et al. The genome of Laccaria bicolor provides insights into mycorrhizal symbiosis. Nature. 2008;452(7183):88–92.
Mashige F, Ohkubo A, Matsushima Y, Takano M, Tsuchiya E, Kanazawa H, et al. High-performance liquid chromatographic determination of catecholamine metabolites and 5-hydroxyindoleacetic acid in human urine using a mixed-mode column and an eight-channel electrode electrochemical detector. J Chromatogr B. 1994;658(1):63–8.
Bicker W, Lammerhofer M, Lindner W. Determination of chlorpyrifos metabolites in human urine by reversed-phase/weak anion exchange liquid chromatography-electrospray ionisation-tandem mass spectrometry. J Chromatogr B. 2005;822(1–2):160–9.
Ferguson PL, Iden CR, Brownawell BJ. Analysis of nonylphenol and nonylphenol ethoxylates in environmental samples by mixed-mode high-performance liquid chromatography–electrospray mass spectrometry. J Chromatogr A. 2001;938(1–2):79–91.
Lara-Martin PA, Gomez-Parra A, Gonzalez-Mazo E. Development of a method for the simultaneous analysis of anionic and non-ionic surfactants and their carboxylated metabolites in environmental samples by mixed-mode liquid chromatography-mass spectrometry. J Chromatogr A. 2006;1137(2):188–97.
Gao L, Shi Y, Li W, Ren W, Liu J, Cai Y. Determination of organophosphate esters in water samples by mixed-mode liquid chromatography and tandem mass spectrometry. J Sep Sci. 2015;38(13):2193–200.
Montes R, Aguirre J, Vidal X, Rodil R, Cela R, Quintana JB. Screening for polar chemicals in water by trifunctional mixed-mode liquid chromatography-high resolution mass spectrometry. Environ Sci Technol. 2017;51(11):6250–9.
Montes R, Rodil R, Cela R, Quintana JB. Determination of persistent and mobile organic contaminants (PMOCs) in water by mixed-mode liquid chromatography-tandem mass spectrometry. Anal Chem. 2019;91(8):5176–83.
González-Mariño I, Estevez-Danta A, Rodil R, Da Silva KM, Sodre FF, Cela R, et al. Profiling cocaine residues and pyrolytic products in wastewater by mixed-mode liquid chromatography-tandem mass spectrometry. Drug Test Anal. 2019;11(7):1018–27.
Small H, Stevens TS, Bauman WC. Novel ion exchange chromatographic method using conductimetric detection. Anal Chem. 1975;47(11):1801–9.
Wei D, Wang X, Wang N, Zhu Y. A rapid ion chromatography column-switching method for online sample pretreatment and determination of l-carnitine, choline and mineral ions in milk and powdered infant formula. RSC Adv. 2017;7(10):5920–7.
Muhammad N, Guo D, Zhang Y, Intisar A, Subhani Q, Qadir MA, et al. Online clean-up setup for the determination of non-fluorescent acidic pharmaceutical drugs in complex biological samples. J Chromatogr A. 2019;1126-1127:121708.
Luo X, Chen L, Zhao Y. Simultaneous determination of three chloroacetic acids, three herbicides, and 12 anions in water by ion chromatography. J Sep Sci. 2015;38(17):3096–102.
Zakaria P, Bloomfield C, Shellie RA, Haddad PR, Dicinoski GW. Determination of bromate in sea water using multi-dimensional matrix-elimination ion chromatography. J Chromatogr A. 2011;1218(50):9080–5.
Gyparakis S, Diamadopoulos E. Formation and reverse osmosis removal of bromate ions during ozonation of groundwater in coastal areas. Sep Sci Technol. 2007;42(7):1465–76.
Chen Z, Megharaj M, Naidu R. Determination of bromate and bromide in seawater by ion chromatography, with an ammonium salt solution as mobile phase, and inductively coupled plasma mass spectrometry. Chromatographia. 2007;65(1–2):115–8.
Evenhuis CJ, Buchberger W, Hilder EF, Flook KJ, Pohl CA, Nesterenko PN, et al. Separation of inorganic anions on a high capacity porous polymeric monolithic column and application to direct determination of anions in seawater. J Sep Sci. 2008;31(14):2598–604.
Nojavan S, Bidarmanesh T, Memarzadeh F, Chalavi S. Electro-driven extraction of inorganic anions from water samples and water miscible organic solvents and analysis by ion chromatography. Electrophoresis. 2014;35(17):2446–53.
Liu J-M, Liu C-C, Fang G-Z, Wang S. Advanced analytical methods and sample preparation for ion chromatography techniques. RSC Adv. 2015;5(72):58713–26.
Teh HB, Li SF. Simultaneous determination of bromate, chlorite and haloacetic acids by two-dimensional matrix elimination ion chromatography with coupled conventional and capillary columns. J Chromatogr A. 2015;1383:112–20.
Pacholec F, Eaton DR, Rossi DT. Characterization of mixtures of organic acids by ion-exclusion partition chromatography-mass spectrometry. Anal Chem. 1986;58(12):2581–3.
Stoll DR. Pass the salt: evolution of coupling ion-exchange separations and mass spectrometry. LCGC N Am. 2019;37(8):504–8.
Karu N, Dicinoski GW, Hanna-Brown M, Haddad PR. Determination of pharmaceutically related compounds by suppressed ion chromatography: II. Interactions of analytes with the suppressor. J Chromatogr A. 2012;1224:35–42.
Pantsar-Kallio M, Manninen PKG. Speciation of halogenides and oxyhalogens by ion chromatography-inductively coupled plasma mass spectrometry. Anal Chim Acta. 1998;360(1–3):161–6.
Jansons M, Pugajeva I, Bartkevics V. Evaluation of selected buffers for simultaneous determination of ionic and acidic pesticides including glyphosate using anion exchange chromatography with mass spectrometric detection. J Sep Sci. 2019;42(19):3077–85.
Wouters S, Haddad PR, Eeltink S. System design and emerging hardware technology for ion chromatography. Chromatographia. 2016;80(5):689–704.
Eghbali H, Bruggink C, Agroskin Y, Pohl CA, Eeltink S. Performance evaluation of ion-exchange chromatography in capillary format. J Sep Sci. 2012;35(24):3461–8.
Wang J, Schnute WC. Optimizing mass spectrometric detection for ion chromatographic analysis. I. Common anions and selected organic acids. Rapid Commun Mass Spectrom. 2009;23(21):3439–47.
Pröfrock D, Prange A. Inductively coupled plasma-mass spectrometry (ICP-MS) for quantitative analysis in environmental and life sciences: a review of challenges, solutions, and trends. Appl Spectrosc. 2012;66(8):843–68.
Sacher F, Raue B, Brauch HJ. Analysis of iodinated X-ray contrast agents in water samples by ion chromatography and inductively-coupled plasma mass spectrometry. J Chromatogr A. 2005;1085(1):117–23.
Aga DS, Thurman EM, Yockel ME, Zimmerman LR, Williams TD. Identification of a new sulfonic acid metabolite of metolachlor in soil. Environ Sci Technol. 1996;30(2):592–7.
Profrock D, Prange A. Compensation of gradient related effects when using capillary liquid chromatography and inductively coupled plasma mass spectrometry for the absolute quantification of phosphorylated peptides. J Chromatogr A. 2009;1216(39):6706–15.
Axelsson BO, Jornten-Karlsson M, Michelsen P, Abou-Shakra F. The potential of inductively coupled plasma mass spectrometry detection for high-performance liquid chromatography combined with accurate mass measurement of organic pharmaceutical compounds. Rapid Commun Mass Spectrom. 2001;15(6):375–85.
Bettmer J, Montes Bayon M, Encinar JR, Fernandez Sanchez ML, Fernandez de la Campa Mdel R, Sanz Medel A. The emerging role of ICP-MS in proteomic analysis. J Proteome. 2009;72(6):989–1005.
Prange A, Pröfrock D. Chemical labels and natural element tags for the quantitative analysis of bio-molecules. J Anal At Spectrom. 2008;23(4):432.
Michalski R (2016) Application of IC-MS and IC-ICP-MS in environmental research. John Wiley & Sons, Hoboken, New Jersey.
Reyes LH, Mar JL, Rahman GM, Seybert B, Fahrenholz T, Kingston HM. Simultaneous determination of arsenic and selenium species in fish tissues using microwave-assisted enzymatic extraction and ion chromatography-inductively coupled plasma mass spectrometry. Talanta. 2009;78(3):983–90.
Christison T, Madden JE, Rohrer J (2019) Determination of cationic polar pesticides in homogenized fruit and vegetable samples using IC-HRAM MS. Thermo Fisher, Sunnyvale, CA, USA.
Michalski R. Ion chromatography applications in wastewater analysis. Separations. 2018;5:16.
Kurz A, Bousova K, Beck J, Schoutsen F, Bruggink C, Kozeluh M, Kule L, Godula M (2017) Routine analysis of polar pesticides in water at low ng/L levels by ion chromatography coupled to triple quadrupole mass spectrometer. Thermo Scientific.
Gallidabino MD, Hamdan L, Murphy B, Barron LP. Suspect screening of halogenated carboxylic acids in drinking water using ion exchange chromatography - high resolution (Orbitrap) mass spectrometry (IC-HRMS). Talanta. 2018;178:57–68.
Gallidabino MD, Irlam RC, Salt MC, O'Donnell M, Beardah MS, Barron LP. Targeted and non-targeted forensic profiling of black powder substitutes and gunshot residue using gradient ion chromatography - high resolution mass spectrometry (IC-HRMS). Anal Chim Acta. 2019;1072:1–14.
Ali I, Alharbi OML, Marsin Sanagi M. Nano-capillary electrophoresis for environmental analysis. Environ Chem Lett. 2015;14(1):79–98.
Bol’shakova DS, Amelin VG. Determination of pesticides in environmental materials and food products by capillary electrophoresis. J Anal Chem. 2016;71(10):965–1013.
Hamdan II. Capillary electrophoresis in the analysis of pharmaceuticals in environmental water: a critical review. J Liq Chromatogr Relat Technol. 2017;40(3):111–25.
Altria KD, Elder D. Overview of the status and applications of capillary electrophoresis to the analysis of small molecules. J Chromatogr A. 2004;1023(1):1–14.
Quesada-Molina C, Md O-I, García-Campaña AM. Analysis of cephalosporin residues in environmental waters by capillary zone electrophoresis with off-line and on-line preconcentration. Anal Methods. 2012;4(8):2341.
Herrera-Herrera AV, Hernandez-Borges J, Borges-Miquel TM, Rodriguez-Delgado MA. Dispersive liquid-liquid microextraction combined with nonaqueous capillary electrophoresis for the determination of fluoroquinolone antibiotics in waters. Electrophoresis. 2010;31(20):3457–65.
Timerbaev AR, Hirokawa T. Recent advances of transient isotachophoresis-capillary electrophoresis in the analysis of small ions from high-conductivity matrices. Electrophoresis. 2006;27(1):323–40.
Horciciak M, Masar M, Bodor R, Danc L, Bel P. Trace analysis of glyphosate in water by capillary electrophoresis on a chip with high sample volume loadability. J Sep Sci. 2012;35(5–6):674–80.
Krishna Marothu V, Gorrepati M, Vusa R. Electromembrane extraction - a novel extraction technique for pharmaceutical, chemical, clinical and environmental analysis. J Chromatogr Sci. 2013;51(7):619–31.
Tabani H, Fakhari AR, Zand E. Low-voltage electromembrane extraction combined with cyclodextrin modified capillary electrophoresis for the determination of phenoxy acid herbicides in environmental samples. Anal Methods. 2013;5(6):1548.
Herrera-Herrera AV, Ravelo-Perez LM, Hernandez-Borges J, Afonso MM, Palenzuela JA, Rodriguez-Delgado MA. Oxidized multi-walled carbon nanotubes for the dispersive solid-phase extraction of quinolone antibiotics from water samples using capillary electrophoresis and large volume sample stacking with polarity switching. J Chromatogr A. 2011;1218(31):5352–61.
Maijo I, Fontanals N, Borrull F, Neususs C, Calull M, Aguilar C. Determination of UV filters in river water samples by in-line SPE-CE-MS. Electrophoresis. 2013;34(3):374–82.
Lombardo-Agui M, Gamiz-Gracia L, Garcia-Campana AM, Cruces-Blanco C. Sensitive determination of fluoroquinolone residues in waters by capillary electrophoresis with laser-induced fluorescence detection. Anal Bioanal Chem. 2010;396(4):1551–7.
Rageh AH, Klein K-F, Pyell U. Off-line and on-line enrichment of α-aminocephalosporins for their analysis in surface water samples using CZE coupled to LIF. Chromatographia. 2016;79(3–4):225–41.
Zhang Z, Zhang D, Zhang X. Simultaneous determination of pharmaceutical and personal care products in wastewater by capillary electrophoresis with head-column field-amplified sample stacking. Anal Methods. 2014;6(19):7978–83.
Ahrer W, Buchberger W. Combination of aqueous and non-aqueous capillary electrophoresis with electrospray mass spectrometry for the determination of drug residues in water. Monatsh Chem. 2001;132(3):329–37.
Höcker O, Bader T, Schmidt TC, Schulz W, Neusüss C. Enrichment-free analysis of anionic micropollutants in the sub-ppb range in drinking water by capillary electrophoresis-high resolution mass spectrometry. Anal Bioanal Chem. 2020.
Deng B, Xu Q, Lu H, Ye L, Wang Y. Pharmacokinetics and residues of tetracycline in crucian carp muscle using capillary electrophoresis on-line coupled with electrochemiluminescence detection. Food Chem. 2012;134(4):2350–4.
Sun H, Qi H, Li H. Development of capillary electrophoretic method combined with accelerated solvent extraction for simultaneous determination of residual sulfonamides and their acetylated metabolites in aquatic products. Food Anal Methods. 2012;6(4):1049–55.
Knoll S, Jacob S, Mieck S, Triebskorn R, Braunbeck T, Huhn C. Relevance of charged micropollutants – quantification of metformin and its transformation product guanylurea in biota by capillary electrophoresis-mass spectrometry. Anal Bioanal Chem. 2020.
Chapel S, Rouvière F, Heinisch S (2019) Pushing the limits of resolving power and analysis time in on-line comprehensive hydrophilic interaction x reversed phase liquid chromatography for the analysis of complex peptide samples. J Chromatogr A:460753.
Brudin SS, Shellie RA, Haddad PR, Schoenmakers PJ. Comprehensive two-dimensional liquid chromatography: ion chromatography x reversed-phase liquid chromatography for separation of low-molar-mass organic acids. J Chromatogr A. 2010;1217(43):6742–6.
Beutner A, Kochmann S, Mark JJ, Matysik FM. Two-dimensional separation of ionic species by hyphenation of capillary ion chromatography x capillary electrophoresis-mass spectrometry. Anal Chem. 2015;87(6):3134–8.
Ranjbar L, Foley JP, Breadmore MC. Multidimensional liquid-phase separations combining both chromatography and electrophoresis - a review. Anal Chim Acta. 2017;950:7–31.
Stevenson PG, Fairchild JN, Guiochon G. Retention mechanism divergence of a mixed mode stationary phase for high performance liquid chromatography. J Chromatogr A. 2011;1218(14):1822–7.
Miller L. Preparative enantioseparations using supercritical fluid chromatography. J Chromatogr A. 2012;1250:250–5.
Funding
Open Access funding provided by Projekt DEAL. We thank for the support from Effect-Net (Effect Network in Water Research) which is part of the Wassernetzwerk Baden-Württemberg and funded by the Ministry for Science, Research and Arts of Baden-Württemberg as well as the Excellence Initiative, a jointly funded program of the German Federal and State governments, organized by the German Research Foundation (DFG).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Published in the topical collection featuring Female Role Models in Analytical Chemistry.
Electronic supplementary material
ESM 1
(PDF 380 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Knoll, S., Rösch, T. & Huhn, C. Trends in sample preparation and separation methods for the analysis of very polar and ionic compounds in environmental water and biota samples. Anal Bioanal Chem 412, 6149–6165 (2020). https://doi.org/10.1007/s00216-020-02811-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-020-02811-5