Abstract
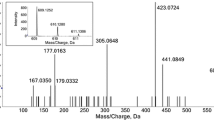
In a comprehensive study, more than 60 phenolic compounds were detected in methanolic extracts from different tissues of pineapple infructescence by high-performance liquid chromatography with diode array detection and electrospray ionisation multiple-stage mass spectrometry (HPLC-DAD-ESI-MSn) as well as by gas chromatography-mass spectrometry (GC-MS). The analytical workflow combining both methods revealed numerous compounds assigned for the first time as pineapple constituents by their mass fragmentations. Pineapple crown tissue was characterised by depsides of p-coumaric and ferulic acid. In contrast, major phenolic compounds in pineapple pulp extracts were assigned to diverse S-p-coumaryl, S-coniferyl and S-sinapyl derivatives of glutathione, N-l-γ-glutamyl-l-cysteine and l-cysteine, which were also identified in the peel. The latter was additionally characterised by elevated concentrations of p-coumaric, ferulic and caffeic acid depsides and glycerides, respectively. Two peel-specific cyanidin hexosides were found. Elevated concentrations of isomeric N,N′-diferuloylspermidines may be a useful tool for the detection of fraudulent peel usage for pineapple juice production. Mass fragmentation pathways of characteristic pineapple constituents are proposed, and their putative biological functions are discussed.



Similar content being viewed by others
Abbreviations
- bp intensity:
-
Base peak intensity
- CID:
-
Collision-induced dissociation
- CR:
-
Crown
- E(Z):
-
Pyroglutamic acid
- ESI-MS:
-
Electrospray ionisation mass spectrometry
- GC-MS:
-
Gas chromatography-mass spectrometry
- GSH:
-
Glutathione
- hex:
-
Hexose
- HPLC-DAD:
-
High-performance liquid chromatography-diode array detection
- LLE:
-
Liquid-liquid extraction
- LRI:
-
Linear retention index
- MS:
-
Mass spectrometry
- NMR:
-
Nuclear magnetic resonance
- PTFE:
-
Polytetrafluoroethylene
- RP:
-
Reversed phase
- SPE:
-
Solid-phase extraction
- TIC:
-
Total ion current
- t R :
-
Retention time
- γ-EC:
-
N-l-γ-Glutamyl-l-cysteine
References
Bartholomew DP, Paull RE, Rohrbach KG (eds) (2003) The pineapple—botany, production and uses. CABI, Wallingford
Steingass CB, Carle R, Schmarr H-G (2015) Ripening-dependent metabolic changes in the volatiles of pineapple (Ananas comosus (L.) Merr.) fruit: I. Characterization of pineapple aroma compounds by comprehensive two-dimensional gas chromatography-mass spectrometry. Anal Bioanal Chem 407(9):2591–2608
Steingass CB, Jutzi M, Müller J, Carle R, Schmarr H-G (2015) Ripening-dependent metabolic changes in the volatiles of pineapple (Ananas comosus (L.) Merr.) fruit: II. Multivariate statistical profiling of pineapple aroma compounds based on comprehensive two-dimensional gas chromatography-mass spectrometry. Anal Bioanal Chem 407(9):2609–2624
Steingass CB, Langen J, Carle R, Schmarr H-G (2015) Authentication of pineapple (Ananas comosus [L.] Merr.) fruit maturity stages by quantitative analysis of γ- and δ-lactones using headspace solid-phase microextraction and chirospecific gas chromatography-selected ion monitoring mass spectrometry (HS-SPME-GC-MS-SIM). Food Chem 168:496–503
Montero-Calderón M, Rojas-Graü MA, Martín-Belloso O (2010) Pineapple (Ananas comosus [L.] Merril) flavor. In: Hui YH (ed) Handbook of fruit and vegetable flavors. Wiley, Hoboken, pp 391–414
Steingass CB, Grauwet T, Carle R (2014) Influence of harvest maturity and fruit logistics on pineapple (Ananas comosus [L.] Merr.) volatiles assessed by headspace solid phase microextraction and gas chromatography–mass spectrometry (HS-SPME-GC/MS). Food Chem 150:382–391
Wen L, Wrolstad RE, Hsu VL (1999) Characterization of sinapyl derivatives in pineapple (Ananas comosus [L.] Merill) juice. J Agric Food Chem 47(3):850–853
Wrolstad RE, Wen L (2001) Natural antibrowning and antioxidant compositions and methods for making the same US 62774926 B1
Wen L, Wrolstad RE (2002) Phenolic composition of authentic pineapple juice. J Food Sci 67(1):155–163
Zheng Z, Ma J, Cheng K, Chao J, Zhu Q, Chang RC, Zhao M, Lin Z, Wang M (2010) Sulfur-containing constituents and one 1H-pyrrole-2-carboxylic acid derivative from pineapple [Ananas comosus (L.) Merr.] fruit. Phytochemistry 71(17–18):2046–2051
Peñalvo JL, Haajanen KM, Botting N, Adlercreutz H (2005) Quantification of lignans in food using isotope dilution gas chromatography/mass spectrometry. J Agric Food Chem 53(24):9342–9347
Ma C, Xiao S, Li Z, Wang W, Du L (2007) Characterization of active phenolic components in the ethanolic extract of Ananas comosus L. leaves using high-performance liquid chromatography with diode array detection and tandem mass spectrometry. J Chromatogr A 1165(1–2):39–44
Schieber A, Stintzing FC, Carle R (2001) By-products of plant food processing as a source of functional compounds—recent developments. Trends Food Sci Technol 12(11):401–413
Sweeley CC, Bentley R, Makita M, Wells WW (1963) Gas-liquid chromatography of trimethylsilyl derivatives of sugars and related substances. J Am Chem Soc 85(16):2497–2507
Kim D, Lee CY (2002) Extraction and isolation of polyphenolics. In: Wrolstad RE, Acree TE, Decker EA, Penner MH, Reid DS, Schwartz SJ, Shoemaker CF, Smith DM, Sporns P (eds) Current protocols in food analytical chemistry. Wiley, New York, pp I1.2.1–I1.2.12
Kammerer D, Claus A, Carle R, Schieber A (2004) Polyphenol screening of pomace from red and white grape varieties (Vitis vinifera L.) by HPLC-DAD-MS/MS. J Agric Food Chem 52(14):4360–4367
Krygier K, Sosulski F, Hogge L (1982) Free, esterified, and insoluble-bound phenolic acids. 1. Extraction and purification procedure. J Agric Food Chem 30(2):330–334
Lorenz P, Conrad J, Bertrams J, Berger M, Duckstein S, Meyer U, Stintzing FC (2012) Investigations into the phenolic constituents of dog’s mercury (Mercurialis perennis L.) by LC-MS/MS and GC-MS analyses. Phytochem Anal 23(1):60–71
Suzuki S, Sakakibara N, Li L, Umezawa T, Chiang VL (2010) Profiling of phenylpropanoid monomers in developing xylem tissue of transgenic aspen (Populus tremuloides). J Wood Sci 56(1):71–76
Lorenz P, Knödler M, Bertrams J, Berger M, Meyer U, Stintzing FC (2010) n-Alkylresorcinol occurrence in Mercurialis perennis L. (Euphorbiaceae). Z Naturforsch 65(3–4):174–179
Lorenz P, Berger M, Bertrams J, Wende K, Wenzel K, Lindequist U, Meyer U, Stintzing FC (2008) Natural wax constituents of a supercritical fluid CO2 extract from quince (Cydonia oblonga Mill.) pomace. Anal Bioanal Chem 391(2):633–646
Duckstein SM, Lorenz P, Stintzing FC (2012) Conversion of phenolic constituents in aqueous Hamamelis virginiana leaf extracts during fermentation. Phytochem Anal 23(6):588–597
Van den Dool H, Kratz PD (1963) A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J Chromatogr A 11:463–471
National Institute of Standards and Technology (2013) NIST Chemistry WebBook. http://webbook.nist.gov/. Accessed 3 Feb 2015
Molnár-Perl I, Vasanits A, Horváth K (1998) Simultaneous GC-MS quantitation of phosphoric, aliphatic and aromatic carboxylic acids, proline and hydroxymethylfurfurol as their trimethylsilyl derivatives: in model solutions II. Chromatographia 48(1–2):111–119
Horman I, Viani R (1971) Plant-polyphenols and related compounds. A mass-spectrometric study of the trimethylsilyl derivatives of hydroxy- and/or methoxy-substituted cinnamic acids and of their methyl esters. Org Mass Spectrom 5(2):203–219
Molnár-Perl I, Horváth K, Bartha R (1998) GC-MS quantitation of benzoic and aralkyl carboxylic acids as their trimethylsilyl derivatives: in model solutions I. Chromatographia 48(1–2):101–110
Lin L, Harnly JM (2007) A screening method for the identification of glycosylated flavonoids and other phenolic compounds using a standard analytical approach for all plant materials. J Agric Food Chem 55(4):1084–1096
Clifford MN, Johnston KL, Knight S, Kuhnert N (2003) Hierarchical scheme for LC-MSn identification of chlorogenic acids. J Agric Food Chem 51(10):2900–2911
Stintzing FC, Kammerer D, Schieber A, Adama H, Nacoulma OG, Carle R (2004) Betacyanins and phenolic compounds from Amaranthus spinosus L. and Boerhavia erecta L. Z Naturforsch 59(1–2):1–8
Vanholme R, Storme V, Vanholme B, Sundin L, Christensen JH, Goeminne G, Halpin C, Rohde A, Morreel K, Boerjana W (2012) A systems biology view of responses to lignin biosynthesis perturbations in Arabidopsis. Plant Cell 24(9):3506–3529
Maier C, Conrad J, Carle R, Weiss J, Schweiggert RM (2015) Phenolic constituents in commercial aqueous Quillaja (Quillaja saponaria Molina) wood extracts. J Agric Food Chem 63(6):1756–1762
Zywicki B, Reemtsma T, Jekel M (2002) Analysis of commercial vegetable tanning agents by reversed-phase liquid chromatography-electrospray ionization-tandem mass spectrometry and its application to wastewater. J Chromatogr A 970(1–2):191–200
Eklund PC, Backman MJ, Kronberg LÅ, Smeds AI, Sjöholm RE (2008) Identification of lignans by liquid chromatography-electrospray ionization ion-trap mass spectrometry. J Mass Spectrom 43(1):97–107
Dieckhaus CM, Fernández-Metzler CL, King R, Krolikowski PH, Baillie TA (2005) Negative ion tandem mass spectrometry for the detection of glutathione conjugates. Chem Res Toxicol 18(4):630–638
Thakur SS, Balaram P (2008) Fragmentation of peptide disulfides under conditions of negative Ion mass spectrometry: studies of oxidized glutathione and contryphan. J Am Soc Mass Spectrom 19(3):358–366
Youhnovski N, Bigler L, Werner C, Hesse M (1998) On-line coupling of high-performance liquid chromatography to atmospheric pressure chemical ionization mass spectrometry (HPLC/APCI-MS and MS/MS). The pollen analysis of hippeastrum x hortorum (Amaryllidaceae). Helv Chim Acta 81(9):1654–1671
Kite GC, Larsson S, Veitch NC, Porter EA, Ding N, Simmonds MSJ (2013) Acyl spermidines in inflorescence extracts of elder (Sambucus nigra L., Adoxaceae) and elderflower drinks. J Agric Food Chem 61(14):3501–3508
Sobolev VS, Sy AA, Gloer JB (2008) Spermidine and flavonoid conjugates from peanut (Arachis hypogaea) flowers. J Agric Food Chem 56(9):2960–2969
Kammerer D, Carle R, Schieber A (2004) Quantification of anthocyanins in black carrot extracts (Daucus carota ssp. sativus var. atrorubens Alef.) and evaluation of their color properties. Eur Food Res Technol 219(5):479–486
Saito N, Harborne JB (1983) A cyanidin glycoside giving scarlet coloration in plants of the Bromeliaceae. Phytochemistry 22(8):1735–1740
Umano K, Hagi Y, Nakahara K, Shoji A, Shibamoto T (1992) Volatile constituents of green and ripened pineapple (Ananas comosus [L.] Merr.). J Agric Food Chem 40(4):599–603
Wu P, Kuo MC, Hartman TG, Rosen RT, Ho CT (1991) Free and glycosidically bound aroma compounds in pineapple (Ananas comosus L. Merr.). J Agric Food Chem 39(1):170–172
Elliger CA, Lundin RE, Haddon WF (1981) Caffeyl esters of glucaric acid in Lycopersicon esculentum leaves. Phytochemistry 20(5):1133–1134
Strack D, Gross W, Wray V, Grotjahn L (1987) Enzymic synthesis of caffeoylglucaric acid from chlorogenic acid and glucaric acid by a protein preparation from tomato cotyledons. Plant Physiol 83(3):475–478
Strack D, Keller H, Weissenbock G (1987) Enzymatic synthesis of hydroxycinnamic acid esters of sugar acids and hydroaromatic acids by protein preparations from rye (Secale cereale) primary leaves. J Plant Physiol 131(1–2):61–73
Risch B, Herrmann K, Wray V, Grotjahn L (1987) 2′-(E)-O-p-Coumaroylgalactaric acid and 2′-(E)-O-feruloylgalactaric acid in citrus. Phytochemistry 26(2):509–510
Risch B, Herrmann K, Wray V (1988) (E)-O-p-coumaroyl-, (E)-O-feruloyl-derivatives of glucaric acid in citrus. Phytochemistry 27(10):3327–3329
Maas M, Petereit F, Hensel A (2009) Caffeic acid derivatives from Eupatorium perfoliatum L. Molecules 14(1):36–45
Smith BG, Harris PJ (2001) Ferulic acid is esterified to glucuronoarabinoxylans in pineapple cell walls. Phytochemistry 56(5):513–519
Strack D, Paula L, Maria B, Victor W, Lutz G (1987) Hydroxycinnamic acid esters of isocitric acid: accumulation and enzymatic synthesis in Amaranthus cruentus. Phytochemistry 26(11):2919–2922
Hauck B, Gallagher JA, Morris SM, Leemans D, Winters AL (2014) Soluble phenolic compounds in fresh and ensiled orchard grass (Dactylis glomerata L.), a common species in permanent pastures with potential as a biomass feedstock. J Agric Food Chem 62(2):468–475
Martin-Tanguy J, Cabanne F, Perdrizet E, Martin C (1978) The distribution of hydroxycinnamic acid amides in flowering plants. Phytochemistry 17(11):1927–1928
Silva DB, Turatti ICC, Gouveia DR, Ernst M, Teixeira SP, Lopes NP (2014) Mass spectrometry of flavonoid vicenin-2, based sunlight barriers in Lychnophora species. Sci Rep 4:1–8
Delaporte RH, Guzen KP, Laverde A Jr, dos Santos AR, Sarragiotto MH (2006) Phenylpropanoid glycerols from Tillandsia streptocarpa Baker (Bromeliaceae). Biochem Syst Ecol 34(7):599–602
Jandrić Z, Roberts D, Rathor MN, Abrahim A, Islam M, Cannavan A (2014) Assessment of fruit juice authenticity using UPLC–QToF MS: a metabolomics approach. Food Chem 148:7–17
Mullen W, Marks SC, Crozier A (2007) Evaluation of phenolic compounds in commercial fruit juices and fruit drinks. J Agric Food Chem 55(8):3148–3157
Thompson D, Norbeck K, Olsson L, Constantin-Teodosiu D, Van der Zee J, Moldeus P (1989) Peroxidase-catalyzed oxidation of eugenol: formation of a cytotoxic metabolite(s). J Biol Chem 264(2):1016–1021
Thompson D, Constantin-Teodosiu D, Egestad B, Mickos H, Moldeus P (1990) Formation of glutathione conjugates during oxidation of eugenol by microsomal fractions of rat liver and lung. Biochem Pharmacol 39(10):1587–1595
Kritzinger EC, Bauer FF, Du Toit WJ (2013) Role of glutathione in winemaking: a review. J Agric Food Chem 61(2):269–277
Silverstein RM, Rodin JO, Himel CM, Leeper RW (1965) Volatile flavor and aroma components of pineapple II. Isolation and identification of chavicol and γ-caprolactone. J Food Sci 30(4):668–672
Marrs KA (1996) The functions and regulation of glutathione S-transferases in plants. Annu Rev Plant Physiol Plant Mol Biol 47(1):127–158
Heinicke RM, Gortner WA (1957) Stem bromelain—a new protease preparation from pineapple plants. Econ Bot 11(3):225–234
Cejudo-Bastante MJ, Pérez-Coello MS, Hermosín-Gutiérrez I (2010) Identification of new derivatives of 2-S-glutathionylcaftaric acid in aged white wines by HPLC-DAD-ESI-MSn. J Agric Food Chem 58(21):11483–11492
Cheynier VF, Trousdale EK, Singleton VL, Salgues MJ, Wylde R (1986) Characterization of 2-S-glutathionylcaftaric acid and its hydrolysis in relation to grape wines. J Agric Food Chem 34(2):217–221
Richard FC, Goupy PM, Nicolas JJ, Lacombe J, Pavia AA (1991) Cysteine as an inhibitor of enzymatic browning. 1. Isolation and characterization of addition compounds formed during oxidation of phenolics by apple polyphenol oxidase. J Agric Food Chem 39(5):841–847
Dean JV, Devarenne TP, Lee IS, Orlofsky LE (1995) Properties of a maize glutathione S-transferase that conjugates coumaric acid and other phenylpropanoids. Plant Physiol 108(3):985–994
Acknowledgments
C.B.S. gratefully acknowledges a Ph.D. scholarship granted by the Landesgraduiertenförderung Baden-Württemberg. We thank Frank Oberschilp (Peelco, Accra, Ghana) and Fritz Schumacher (Schumacher, Filderstadt-Bernhausen, Germany) for kindly supplying the pineapple fruits.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 700 kb)
Rights and permissions
About this article
Cite this article
Steingass, C.B., Glock, M.P., Schweiggert, R.M. et al. Studies into the phenolic patterns of different tissues of pineapple (Ananas comosus [L.] Merr.) infructescence by HPLC-DAD-ESI-MSn and GC-MS analysis. Anal Bioanal Chem 407, 6463–6479 (2015). https://doi.org/10.1007/s00216-015-8811-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-015-8811-2