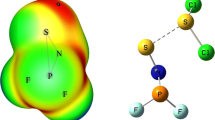
Abstract
Quantum chemical calculations are performed to study the bifurcated chalcogen bonding (BCB) interactions in Ph2N2O2…XCY (X=S, Se, Te and Y=O, S, Se, Te) complexes. The interaction of the only one σ-hole on X atom with two negative regions, which leads to BCB interactions, is exceptionally remarkable. The nature of the BCB interactions is probed by a variety of means, including electrostatic potentials, AIM, NBO, energy decomposition and electron density differences maps. The EDA analysis reveals that the dominant attractive force in X…O chalcogen bonded complexes is the electrostatic energy. The interaction energy values, obtained using the QTAIM-based Espinosa’s approach, provide a relatively distinguishable result than DFT method and EDA analysis reported in this study.





Similar content being viewed by others
References
Clark T (2013) σ-hole. WIREs Comput Mol Sci 3:13–20
Clark T, Hennemann M, Murray JS, Politzer P (2007) Halogen bonding: the σ-hole. J Mol Model 13:291–296
Politzer P, Murray JS, Clark T (2013) Halogen bonding and other σ-hole interactions: a perspective. Phys Chem Chem Phys 15:11178–11190
Murray JS, Lane P, Politzer P (2009) Expansion of the σ-hole concept. J Mol Model 15:723–729
Murray JS, Lane P, Clark T, Politzer P (2007) σ-Hole bonding: molecules containing group VI atoms. J Mol Model 13:1033–1038
Murray JS, Lane P, Politzer P (2007) A predicted new type of directional noncovalent interaction. Int J Quant Chem 107:2286–2292
Wang W, Ji B, Zhang Y (2009) Chalcogen bond: a sister noncovalent bond to halogen bond. J Phys Chem A 113:8132–8135
Nziko VPN, Scheiner S (2014) chalcogen bonding between tetravalet SF4 and amines. J Phys Chem A 118:10849–10856
Oliveira VP, Cremer D, Kraka E (2017) The many facets of chalcogen bonding described by vibrational spectroscopy. J Phys Chem A 121:6845–6862
Sanz P, Yáñez M, Mó O (2002) Competition between X···H···Y intramolecular hydrogen bonds and X····Y (X = O, S, and Y = Se, Te) chalcogen−chalcogen interactions. J Phys Chem A 106:4661–4668
Sanz P, Yáñez M, Mó O (2003) Resonance-assisted intramolecular chalcogen–chalcogen interactions? Chem Eur J 9:4548–4555
Alkorta I, Elguero J, Del Bene JE (2018) Complexes of O=C=S with nitrogen bases: chalcogen bonds, tetrel bonds, and other secondary interactions. Chem Phys Chem 19:1886–1894
Wang H, Liu J, Wang W (2018) Intermolecular and very strong intramolecular C-Se…O/N chalcogen bonds in nitroPhenyl selenocyanate crystals. Phys Chem Chem Phys 20:5227–5234
Pascoe DJ, Ling KB, Cockroft SL (2017) The origin of chalcogen-bonding interactions. J Am Chem Soc 139:15160–15167
Li QZ, Li R, Guo P, Li H, Li WZ, Cheng JB (2012) Competition of chalcogen bond, halogen bond, and hydrogen bond in SCS-HOX and SeCSe-HOX (X = Cl and Br) complexes. Comput Theor Chem 980:56–61
Zhang Y, Wang WZ (2018) Pseudo-bifurcated chalcogen bond in crystal engineering. Crystals 8:163–171
Nayak SK, Kumar V, Murray JS, Politzer P, Terraneo G, Pilati T, Metrangolo P, Resnati G (2017) Fluorination promotes chalcogen bonding in crystalline solids. Cryst Eng Comm 19:4955–4959
Mikherdov AS, Novikov AS, Kinzhalov MA, Zolotarev AA, Boyarskiy VP (2018) Intra-/intermolecular bifurcated chalcogen bonding in crystal structure of thiazole/thiadiazole derived binuclear (diaminocarbene)PdII complexes. Crystals 8:112–127
Bora PL, Novák M, Novotný J, Foroutan-Nejad C, Marek R (2017) Supramolecular covalence in bifurcated chalcogen bonding. Chem Eur J 23:7315–7323
Bhandary S, Sirohiwal A, Kadu R, Kumar S, Chopra D (2018) Dispersion stabilized Se/Te…π double chalcogen bonding synthonsin insitu cryocrystallized divalent organochalcogen liquids. Cryst Growth Des 18:3734–3739
Vančik H (2013) Aromatic C-nitroso compounds. Springer, New York
Yamamoto H, Momiyama N (2005) Rich chemistry of nitroso compounds. Chem Commun 28:3514–3525
Carosso S, Miller MJ (2014) Nitroso Diels–Alder (NDA) reaction as an efficient tool for the functionalization of diene-containing natural products. Org Biomol Chem 12:7445–7468
Yamamoto H, Kawasaki M (2007) Nitroso and azo compounds in modern organic synthesis: late blooming but very rich. Bull Chem Soc Jpn 80:595–607
Peng L, Turesky RJ (2014) Optimizing proteolytic digestion conditions for the analysis of serum albumin adducts of 2-amino-1-methyl-6-Phenylimidazo[4,5-b]pyridine, a potential human carcinogen formed in cooked meat. J Proteomics 103:267–278
Trefzer C, Rengifo-Gonzalez M, J. Hinner M, Schneider P, et al (2010) Benzothiazinones: prodrugs that covalently modify the decaprenylPhosPhoryl-β-d-ribose 2′-epimerase DprE1 of mycobacterium tuberculosis. J Am Chem Soc 132:13663–13665
Liu L, Wagner CR, Hanna PE (2009) Isoform-selective inactivation of human arylamine N-acetyltransferases by reactive metabolites of carcinogenic arylamines. Chem Res Toxicol 22:1962–1974
Hawkins CL, Davies MJ (2014) Detection and characterisation of radicals in biological materials using EPR methodology. Biochim BioPhys Acta Gen Subj 1840:708–721
Piloty O (1898) Ueber aliphatische nitrosoverbindunge. Ber Dtsch Chem Ges 31:452–457
Beaudoin D, Wuest JD (2016) Dimerization of aromatic C-Nitroso compounds. Chem Rev 116:258–286
Esrafili MD, Mohammadian-Sabet F (2015) Bifurcated chalcogen bonds: a theoretical study on the structure, strength and bonding properties. Chem Phys Lett 634:210–215
Boys SF, Bernardi F (1970) The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol Phys 19:553–566
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark MJ, Heyd J, Brothers EN, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell AP, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09 Revision A.02. Gaussian Inc, Wallingford
Bulat FA, Toro-Labbe A, Brinck T, Murray JS, Politzer P (2010) Quantitative analysis of molecular surfaces: areas, volumes, electrostatic potentials and average local ionization energies. J Mol Model 16:1679–1691
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, New York
Biegler-Konig F, Schonbohm J (2002) AIM 2000 Program Package, Ver.2.0, University of Applied Sciences, Bielefield, Germany
Reed AE, Curtiss LA, Weinhold F (1988) Intermolecular interactions from a natural bond orbital, donor-acceptor viewpoint. Chem Rev 88:899–926
ADF2013, SCM, Theoretical chemistry; Vrije Universiteit: Amsterdam, The Netherlands. Available at: https://www.scm.com
E-van L, Baerends EJ, Snijders JG (1993) Relativistic regular two-component hamiltonians. J Chem Phys 99:4597–4610
E-van L, R-van L, Baerends EJ, Snijders JG (1996) Relativistic regular two-component hamiltonians. Int J Quantum Chem 57:281–293
Dieterich DA, Paul IC, Curtin DY (1974) Structural studies on nitrosobenzene and 2-nitrosobenzoic acid. Crystal and molecular structures of cis-azobenzene dioxide and trans-2,2-dicarboxyazobenzene dioxide. J Am Chem Soc 96:6372–6380
Vleeschouwer FD, Denayer M, Pinter B, Geerlings P, Proft FD (2017) Characterization of chalcogen bonding interactions via an in-depth conceptual quantum chemical analysis. J Comput Chem 39:557–572
Esrafili MD, Mohammadian-Sabet F (2015) Does single-electron chalcogen bond exist? Some theoretical insights. J Mol Model 21:65–73
Koch U, Popelier PLA (1995) Characterization of CHO hydrogen bonds on the basis of the charge density. J Phys Chem 99:9747–9754
Rozas I, Alkorta I, Elguero J (1998) Non-conventional hydrogen bonds. Chem Soc Rev 27:163–170
Del Bene JE, Alkorta I, Sanchez-Sanz G, Elguero J (2011) 31P–31P spin–spin coupling constants for pnicogen homodimers. Chem Phys Lett 512:184–187
Gue X, Liu Y-W, Li Q-Z, Li W-Z, Cheng J-B (2015) Competition and cooperativity between tetrel bond and chalcogen bond in complexes involving F2CX (X = Se and Te). Chem Phys Lett 620:7–12
Brezgunova M (2013) Charge density analysis and topological properties of weak intermolecular interactions–halogen and chalcogen bonding–and their comparison with hydrogen bonding. University of Lorraine, Nancy
Wang Y, Zhang Y, Xu Z, Tong J, Teng W, Lu Y (2017) Intramolecular C–S…O=S(C) chalcogen bonds: a theoretical study of the effects of substituents and intermolecular hydrogen bonds. Comput Theor Chem 1115:190–196
Espinosa E, Molins E (2000) Retrieving interaction potentials from the topology of the electron density distribution: the case of hydrogen bonds. J Chem Phys 113:5686–5694
Espinosa E, Molins E, Lecomte C (1998) Hydrogen bond strengths revealed by topological analyses of experimentally observed electron densitie. Chem Phys Lett 285:170–173
Espinosa E, Alkorta I, Rozas I, Elguero J, Molins E (2001) About the evaluation of the local kinetic, potential and total energy densities in closed-shell interactions. Chem Phys Lett 336:457–461
Blanco F, Kelly B, Sánchez-Sanz G, Trujillo C, Alkorta I, Elguero J, Rozas I (2013) Non-covalent interactions: complexes of guanidinium with DNA and RNA nucleobases. J Phys Chem B 117:11608–11616
Esrafili MD, Mohammadian-Sabet F (2015) An ab initio study on chalcogen–chalcogen bond interactions in cyclic (SHX)3 complexes (X = F, Cl, CN, NC, CCH, OH, OCH3, NH2). Chem Phys Lett 628:71–75
Harrison JF (2003) On the role of the electron density difference in the interpretation of molecular properties. J Chem Phys 119:8763–8764
Foster JP, Weinhold F (1980) Natural hybrid orbitals. J Am Chem Soc 102:7211–7218
Glendening ED, Landis CR, Weinhold F (2012) Natural bond orbital methods. WIREs, Comput Mol Sci 2:1–42
Ziegler T, Rauk A (1979) A theoretical study of the ethylene-metal bond in complexes between copper(1+), silver(1+), gold(1+), platinum(0) or platinum(2+) and ethylene, based on the Hartree-Fock-Slater transition-state method. Inorg Chem 18:1558–1565
Acknowledgements
S.M. wishes to acknowledge Dr. M. Bayat, Bu-Ali Sina University, for supporting this research by ADF.2013.01 program.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Massahi, S., Ghobadi, M. & Nikoorazm, M. Exceptional bifurcated chalcogen bonding interaction between Ph2N2O2 and only one σ–hole on XCY (X=S, Se, Te and Y=O, S, Se, Te): a DFT study. Theor Chem Acc 139, 162 (2020). https://doi.org/10.1007/s00214-020-02669-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-020-02669-x