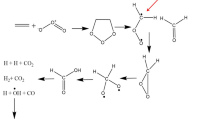
Abstract
A comprehensive theoretical study on the gas-phase decomposition of the CH2OO intermediate is performed at the CCSD(T)//B3LYP/6-311+G(2df,2p) level in the presence of water molecules [n(H2O), n = 1–3]. The calculated results show that the most favorable route is the formation of dioxirane pathway (R3) in the unimolecular decomposition of CH2OO. For the reactions of CH2OO with uni-, bi-, and trimolecular water, the predominant pathway is the CH2OO with bimolecular water reaction as the major product is cis-HMHP. Compared with the barrier of CH2OO reaction with unimolecular water, an addition of water molecule contributes to a reduction of 4.91 kcal mol−1 for the barrier. The elimination processes of cis-HMHP have two parallel competitive pathways: trans-HCOOH + H2O (R9) and HCOH + H2O2 (R10). The apparent activation barriers of these two reactions are significantly reduced with the increase in the number of water molecules involved. The barrier of R9 is higher than that of R10 about 8–15 kcal mol−1 in the presence of water molecules, meaning that the favorable route is the formation of HCOH + H2O2 in the decomposition of cis-HMHP. The rate coefficients of CH2OO reaction with unimolecular water satisfy a positive temperature coefficient behavior at 298–500 K, whereas the rate coefficients of CH2OO reaction with bimolecular water exhibit a negative temperature dependence.









Similar content being viewed by others
References
Edmond PFL, Daniel KWM, Dudley ES, Carl JP, David LO, Craig AT, John MD (2012) Chem Eur J 18:12411
Vereecken L, Harder H, Novelli A (2012) Phys Chem Chem Phys 14:14682
Smith MC, Chang CH, Chao W, Lin LC, Takahashi K, Boering KA, Lin JJM (2015) J Phys Chem Lett 6:2708
Criegee R, Wenner G (1949) Chem Ber 82:9
Su YT, Huang YH, Witek HA, Lee YP (2013) Science 340:174
Anglada JM, González J, Torrent-Sucarrat M (2011) Phys Chem Chem Phys 13:13034
Li J, Carter S, Bowman JM, Dawes R, Xie DQ, Guo H (2014) J Phys Chem Lett 5:2364
Ouyang B, McLeod MW, Jones RL, Bloss WJ (2013) Phys Chem Chem Phys 15:17070
Kuwata KT, Guinn EJ, Hermes MR, Fernandez JA, Mathison JM, Huang K (2015) J Phys Chem A 119:10316
Berndt T, Kaethner R, Voigtländer J, Stratmann F, Pfeifle M, Reichle P, Sipilä M, Kulmala M, Olzmann M (2015) Phys Chem Chem Phys 17:19862
Zhang WC, Du BN, Qin ZL (2014) J Phys Chem A 118:4797
Zhang TL, Wang R, Chen H, Min ST, Wang ZY, Zhao CB, Xu Q, Jin LX, Wang WL, Wang ZQ (2015) Phys Chem Chem Phys 17:15046
Inaba S (2014) J Phys Chem A 118:3026
Stone D, Blitz M, Daubney L, Howes NUM, Seakins P (2014) Phys Chem Chem Phys 16:1139
Chao W, Hsieh JT, Chang CH, Lin JJM (2015) Science 347:751
Lewis TR, Blitz MA, Heard DE, Seakins PW (2015) Phys Chem Chem Phys 17:4859
Nguyen TN, Putikam R, Lin MC (2015) J Chem Phys 142:124312
Kumar M, Busch DH, Subramaniam B, Thompson WH (2014) J Phys Chem A 118:9701
Frisch MJ et al (2009) Gaussian 09, revision C.01. Gaussian Inc, Wallingford
Werner HJ et al MOLPRO, version 2010.1. http://www.molpro.net
Zheng JJ, Truhlar DG (2009) J Phys Chem A 113:11919
Su YT, Lin HY, Putikam R, Matsui H, Lin MC, Lee YP (2014) Nat Chem 6:477
Buras ZJ, Elsamra RMI, Jalan A, Middaugh JE, Green WH (2014) J Phys Chem A 118:1997
Vereecken L, Glowacki DR, Pilling MJ (2015) Chem Rev 115:4063
Miliordos E, Xantheas SS (2016) Angew Chem 128:1027
Gonzalez C, Schlegel HB (1989) J Chem Phys 90:2154
Gonzalez C, Schlegel HB (1990) J Phys Chem 94:5523
Fukui K (1981) Acc Chem Res 14:363
Page M, Mclver JW (1988) J Chem Phys 88:922
Mendes J, Zhou CW, Curran HJ (2013) J Phys Chem A 117:4515
Mendes J, Zhou CW, Curran HJ (2013) J Phys Chem A 117:14006
Eckart C (1930) Phys Rev 35:1303
Garrett BC, Truhlar DG (1979) J Phys Chem 83:2921
Johnston HS, Heicklen J (1962) J Phys Chem 66:532
Zhang TL, Wang WL, Zhang P, Lü J, Zhang Y (2011) Phys Chem Chem Phys 13:20794
Zhang P, Wang WL, Zhang TL, Chen L, Du YM, Li CY, Lü J (2012) J Phys Chem A 116:4610
Duncan WT, Bell RL, Truong TN (1998) J Comput Chem 19:1039
Curtiss LA, Redfern PC, Raghavachari K (2007) J Chem Phys 126:084108
McCarthy MC, Cheng L, Crabtree KN, Martinez O, Nguyen TL, Womack CC, Stanton JF (2013) J Phys Chem Lett 4:4133
Nakajima M, Endo Y (2013) J Chem Phys 139:101103
Nguyen MT, Nguyen TL, Ngan VT, Nguyen HMT (2007) Chem Phys Lett 448:183
Shallcross DE, Leather KE, Bacak A, Xiao P, Lee EPF, Ng M, Mok DKW, Dyke JM, Hossaini R, Chipperfield MP, Khan MAH, Percival CJ (2015) J Phys Chem A 119:4618
Aranda A, LeBras G, LaVerdet G, Poulet G (1997) Geophys Res Lett 24:2745
Karton A, Kettner M, Wild DA (2013) Chem Phys Lett 585:15
Aplincourt P, Ruiz-López MF (2000) J Am Chem Soc 122:8990
Harding LB, Klippenstein SJ (2015) J Chem Phys 143:167101
Ryzhkov AB, Ariya PA (2006) Chem Phys Lett 419:479
Cao J, Wang ZX, Gao LJ, Fu F (2015) J Mol Model 21:66
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Nos: 21473108, 21473107) and Shaanxi Innovative Team of Key Science and Technology (2013KCT-17). The authors thank Researcher Hongyan Xiao of Technical Institute of Physics and Chemistry for valuable discussions.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, L., Wang, W., Wang, W. et al. Water-catalyzed decomposition of the simplest Criegee intermediate CH2OO. Theor Chem Acc 135, 131 (2016). https://doi.org/10.1007/s00214-016-1894-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-016-1894-9