Abstract
Rationale
Nicotine is a principal psychoactive agent in tobacco, contributing to tobacco’s addictive potential. Preclinical studies on the effects of voluntary nicotine intake typically use self-administration procedures that provide continuous nicotine access during each self-administration session. However, many smokers consume cigarettes intermittently rather than continuously throughout each day. For drugs including cocaine and opioids, research in laboratory rats shows that intermittent intake can be more effective than continuous intake in producing patterns of drug use relevant to addiction.
Objective
We determined how intermittent versus continuous nicotine self-administration influences nicotine seeking and taking behaviours.
Methods
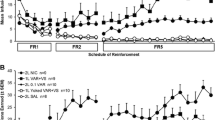
Female and male rats had continuous (i.e., Long Access; LgA, 6 h/day) or intermittent (IntA; 12 min ON, 60 min OFF, for 6 h/day) access to intravenous nicotine (15 µg/kg/infusion), for 12 daily sessions. We then assessed intake, responding for nicotine under a progressive ratio schedule of drug reinforcement and cue- and nicotine-induced reinstatement of drug seeking. We also estimated nicotine pharmacokinetic parameters during LgA and IntA self-administration.
Results
Overall, LgA rats took twice more nicotine than did IntA rats, yielding more sustained increases in estimated brain concentrations of the drug. However, the two groups showed similar motivation to seek and take nicotine, as measured using reinstatement and progressive ratio procedures, respectively.
Conclusions
Intermittent nicotine use is just as effective as continuous use in producing addiction-relevant behaviours, despite significantly less nicotine exposure. This has implications for modeling nicotine self-administration patterns in human smokers and resulting effects on brain and behaviour.






Similar content being viewed by others
References
Abarkan M, Fois GR, Vouillac-Mendoza C, Ahmed SH, Guillem K (2023) Altered neuronal activity in the ventromedial prefrontal cortex drives nicotine intake escalation. Neuropsychopharmacology 48(6):887–896. https://doi.org/10.1038/s41386-022-01428-9
Ahmed SH, Koob GF (1999) Long-lasting increase in the set point for cocaine self-administration after escalation in rats. Psychopharmacology 146(3):3. https://doi.org/10.1007/s002130051121
Algallal H, Allain F, Ndiaye NA, Samaha A-N (2020) Sex differences in cocaine self-administration behaviour under long access versus intermittent access conditions. Addict Biol 25(5):5. https://doi.org/10.1111/adb.12809
Allain F, Samaha A-N (2019) Revisiting long-access versus short-access cocaine self-administration in rats: Intermittent intake promotes addiction symptoms independent of session length. Addict Biol 24(4):4. https://doi.org/10.1111/adb.12629
Allain F, Minogianis E-A, Roberts DCS, Samaha A-N (2015) How fast and how often: The pharmacokinetics of drug use are decisive in addiction. Neurosci Biobehav Rev 56:166–179. https://doi.org/10.1016/j.neubiorev.2015.06.012
Allain F, Roberts DCS, Lévesque D, Samaha A-N (2017) Intermittent intake of rapid cocaine injections promotes robust psychomotor sensitization, increased incentive motivation for the drug and mGlu2/3 receptor dysregulation. Neuropharmacology 117:227–237. https://doi.org/10.1016/j.neuropharm.2017.01.026
Allain F, Bouayad-Gervais K, Samaha A-N (2018) High and escalating levels of cocaine intake are dissociable from subsequent incentive motivation for the drug in rats. Psychopharmacology 235(1):1. https://doi.org/10.1007/s00213-017-4773-8
Allain F, Delignat-Lavaud B, Beaudoin M-P, Jacquemet V, Robinson TE, Trudeau L-E, Samaha A-N (2021) Amphetamine maintenance therapy during intermittent cocaine self-administration in rats attenuates psychomotor and dopamine sensitization and reduces addiction-like behavior. Neuropsychopharmacology 46(2):2. https://doi.org/10.1038/s41386-020-0773-1
Beasley MM, Gunawan T, Tunstall BJ, Kearns DN (2022) Intermittent access training produces greater motivation for a non-drug reinforcer than long access training. Learn Behav 50(4):4. https://doi.org/10.3758/s13420-022-00512-w
Bover MT, Foulds J, Steinberg MB, Richardson D, Marcella SW (2008) Waking at night to smoke as a marker for tobacco dependence: Patient characteristics and relationship to treatment outcome: Waking at night to smoke as a marker for tobacco dependence. Int J Clin Pract 62(2):182–190. https://doi.org/10.1111/j.1742-1241.2007.01653.x
Braznell S, Van Den Akker A, Metcalfe C, Taylor GMJ, Hartmann-Boyce J (2022) Critical appraisal of interventional clinical trials assessing heated tobacco products: A systematic review. Tobacco Control tobaccocontrol-2022–057522. https://doi.org/10.1136/tc-2022-057522
Caggiula AR, Donny EC, Palmatier MI, Liu X, Chaudhri N, Sved AF (2009) Chapter 6: The role of nicotine in smoking: A dual-reinforcement model. Nebraska Symposium on Motivation. Nebr Symp Motiv 55:91–109
Caldeira KM, O’Grady KE, Garnier-Dykstra LM, Vincent KB, Pickworth WB, Arria AM (2012) Cigarette smoking among college students: Longitudinal trajectories and health outcomes. Nicotine Tob Res 14(7):7. https://doi.org/10.1093/ntr/nts131
Calipari ES, Jones SR (2014) Sensitized nucleus accumbens dopamine terminal responses to methylphenidate and dopamine transporter releasers after intermittent-access self-administration. Neuropharmacology 82:1–10. https://doi.org/10.1016/j.neuropharm.2014.02.021
Calipari ES, Ferris MJ, Zimmer BA, Roberts DC, Jones SR (2013) Temporal pattern of cocaine intake determines tolerance vs sensitization of cocaine effects at the dopamine transporter. Neuropsychopharmacology 38(12):12. https://doi.org/10.1038/npp.2013.136
Calipari ES, Ferris MJ, Siciliano CA, Zimmer BA, Jones SR (2014) Intermittent cocaine self-administration produces sensitization of stimulant effects at the dopamine transporter. J Pharmacol Exp Ther 349(2):2. https://doi.org/10.1124/jpet.114.212993
Carr CC, Ferrario CR, Robinson TE (2020) Intermittent access cocaine self-administration produces psychomotor sensitization: Effects of withdrawal, sex and cross-sensitization. Psychopharmacology 237(6):6. https://doi.org/10.1007/s00213-020-05500-4
Chaiton M, Diemert L, Cohen JE, Bondy SJ, Selby P, Philipneri A, Schwartz R (2016) Estimating the number of quit attempts it takes to quit smoking successfully in a longitudinal cohort of smokers. BMJ Open 6(6):6. https://doi.org/10.1136/bmjopen-2016-011045
Chaudhri N, Caggiula AR, Donny EC, Booth S, Gharib MA, Craven LA, Allen SS, Sved AF, Perkins KA (2005) Sex differences in the contribution of nicotine and nonpharmacological stimuli to nicotine self-administration in rats. Psychopharmacology 180(2):2. https://doi.org/10.1007/s00213-005-2152-3
Clinical Practice Guideline Treating Tobacco Use and Dependence 2008 Update Panel, Liaisons, and Staff (2008) A clinical practice guideline for treating tobacco use and dependence: 2008 update. A U.S. Public Health Service report. Am J Prev Med 35(2):2. https://doi.org/10.1016/j.amepre.2008.04.009
Cohen A, Koob GF, George O (2012) Robust escalation of nicotine intake with extended access to nicotine self-administration and intermittent periods of abstinence. Neuropsychopharmacology 37(9):2153–2160. https://doi.org/10.1038/npp.2012.67
Corrigall WA, Coen KM (1989) Nicotine maintains robust self-administration in rats on a limited-access schedule. Psychopharmacology 99(4):4. https://doi.org/10.1007/BF00589894
Craig EL, Zhao B, Cui JZ, Novalen M, Miksys S, Tyndale RF (2014) Nicotine pharmacokinetics in rats is altered as a function of age, impacting the interpretation of animal model data. Drug Metab Dispos 42(9):9. https://doi.org/10.1124/dmd.114.058719
Danaei G, Ding EL, Mozaffarian D, Taylor B, Rehm J, Murray CJL, Ezzati M (2009) The preventable causes of death in the United States: Comparative risk assessment of dietary, lifestyle, and metabolic risk factors. PLoS Med 6(4):4. https://doi.org/10.1371/journal.pmed.1000058
Donny EC, Caggiula AR, Knopf S, Brown C (1995) Nicotine self-administration in rats. Psychopharmacology 122(4):4. https://doi.org/10.1007/BF02246272
Donny EC, Caggiula AR, Rowell PP, Gharib MA, Maldovan V, Booth S, Mielke MM, Hoffman A, McCallum S (2000) Nicotine self-administration in rats: Estrous cycle effects, sex differences and nicotinic receptor binding. Psychopharmacology 151(4):4. https://doi.org/10.1007/s002130000497
Donny EC, Chaudhri N, Caggiula AR, Evans-Martin FF, Booth S, Gharib MA, Clements LA, Sved AF (2003) Operant responding for a visual reinforcer in rats is enhanced by noncontingent nicotine: Implications for nicotine self-administration and reinforcement. Psychopharmacology 169(1):1. https://doi.org/10.1007/s00213-003-1473-3
Dutta S, Matsumoto Y, Ebling WF (1997) Propofol pharmacokinetics and pharmacodynamics assessed from a cremophor EL formulation. J Pharm Sci 86(8):8. https://doi.org/10.1021/js970118m
Feltenstein MW, Ghee SM, See RE (2012) Nicotine self-administration and reinstatement of nicotine-seeking in male and female rats. Drug Alcohol Depend 121(3):3. https://doi.org/10.1016/j.drugalcdep.2011.09.001
Fragale JE, Pantazis CB, James MH, Aston-Jones G (2019) The role of orexin-1 receptor signaling in demand for the opioid fentanyl. Neuropsychopharmacology 44(10):10. https://doi.org/10.1038/s41386-019-0420-x
George O, Lloyd A, Carroll FI, Damaj MI, Koob GF (2011) Varenicline blocks nicotine intake in rats with extended access to nicotine self-administration. Psychopharmacology 213(4):715–722. https://doi.org/10.1007/s00213-010-2024-3
Goldberg SR, Spealman RD, Goldberg DM (1981) Persistent behavior at high rates maintained by intravenous self-administration of nicotine. Science (New York, N.Y.) 214(4520):4520. https://doi.org/10.1126/science.7291998
Grebenstein P, Burroughs D, Zhang Y, LeSage MG (2013) Sex differences in nicotine self-administration in rats during progressive unit dose reduction: Implications for nicotine regulation policy. Pharmacol Biochem Behav 114–115:70–81. https://doi.org/10.1016/j.pbb.2013.10.020
Gueye AB, Allain F, Samaha A-N (2019) Intermittent intake of rapid cocaine injections promotes the risk of relapse and increases mesocorticolimbic BDNF levels during abstinence. Neuropsychopharmacology 44(6):6. https://doi.org/10.1038/s41386-018-0249-8
Hughes JR (2016) National institutes of health funding for tobacco versus harm from tobacco. Nicotine Tob Res 18(5):5. https://doi.org/10.1093/ntr/ntv137
Hughes JR, Keely J, Naud S (2004) Shape of the relapse curve and long-term abstinence among untreated smokers. Addiction (Abingdon, England) 99(1):1. https://doi.org/10.1111/j.1360-0443.2004.00540.x
Hwa Jung B, Chul Chung B, Chung SJ, Shim CK (2001) Different pharmacokinetics of nicotine following intravenous administration of nicotine base and nicotine hydrogen tartarate in rats. J Control Release 77(3):3. https://doi.org/10.1016/s0168-3659(01)00452-7
James MH, Stopper CM, Zimmer BA, Koll NE, Bowrey HE, Aston-Jones G (2019) Increased number and activity of a lateral subpopulation of hypothalamic orexin/hypocretin neurons underlies the expression of an addicted state in rats. Biol Psychiatry 85(11):11. https://doi.org/10.1016/j.biopsych.2018.07.022
Jarvis MJ, Cohen JE, Delnevo CD, Giovino GA (2013) Dispelling myths about gender differences in smoking cessation: Population data from the USA, Canada and Britain. Tob Control 22(5):5. https://doi.org/10.1136/tobaccocontrol-2011-050279
Kawa AB, Robinson TE (2019) Sex differences in incentive-sensitization produced by intermittent access cocaine self-administration. Psychopharmacology 236(2):2. https://doi.org/10.1007/s00213-018-5091-5
Kawa AB, Bentzley BS, Robinson TE (2016) Less is more: Prolonged intermittent access cocaine self-administration produces incentive-sensitization and addiction-like behavior. Psychopharmacology 233(19–20):19–20. https://doi.org/10.1007/s00213-016-4393-8
Kawa AB, Valenta AC, Kennedy RT, Robinson TE (2019) Incentive and dopamine sensitization produced by intermittent but not long access cocaine self-administration. Eur J Neurosci 50(4):4. https://doi.org/10.1111/ejn.14418
Kenny PJ, Markou A (2006) Nicotine self-administration acutely activates brain reward systems and induces a long-lasting increase in reward sensitivity. Neuropsychopharmacology 31(6):6. https://doi.org/10.1038/sj.npp.1300905
Kyerematen GA, Vesell ES (1991) Metabolism of nicotine. Drug Metab Rev 23(1–2):1–2. https://doi.org/10.3109/03602539109029754
Kyerematen GA, Taylor LH, de Bethizy JD, Vesell ES (1988) Pharmacokinetics of nicotine and 12 metabolites in the rat. Application of a new radiometric high performance liquid chromatography assay. Drug Metab Dispos 16(1):1
Lawson GM, Hurt RD, Dale LC, Offord KP, Croghan IT, Schroeder DR, Jiang NS (1998) Application of serum nicotine and plasma cotinine concentrations to assessment of nicotine replacement in light, moderate, and heavy smokers undergoing transdermal therapy. J Clin Pharmacol 38(6):6. https://doi.org/10.1002/j.1552-4604.1998.tb05787.x
Le AD, Li Z, Funk D, Shram M, Li TK, Shaham Y (2006) Increased vulnerability to nicotine self-administration and relapse in alcohol-naive offspring of rats selectively bred for high alcohol intake. J Neurosci 26(6):1872–1879. https://doi.org/10.1523/JNEUROSCI.4895-05.2006
Lenoir M, Tang JS, Woods AS, Kiyatkin EA (2013) Rapid Sensitization of Physiological, Neuronal, and Locomotor Effects of Nicotine: Critical Role of Peripheral Drug Actions. J Neurosci 33(24):24. https://doi.org/10.1523/JNEUROSCI.4940-12.2013
LeSage MG, Keyler DE, Shoeman D, Raphael D, Collins G, Pentel PR (2002) Continuous nicotine infusion reduces nicotine self-administration in rats with 23-h/day access to nicotine. Pharmacol Biochem Behav 72(1–2):1–2. https://doi.org/10.1016/s0091-3057(01)00775-4
Levy DE, Biener L, Rigotti NA (2009) The natural history of light smokers: A population-based cohort study. Nicotine Tob Res 11(2):2. https://doi.org/10.1093/ntr/ntp011
Li X, Anderson SJ, Shiffman S, Zhang B (2022) Time-varying coefficient cumulative gap time models for intensive longitudinal ecological momentary assessment data with missingness. J Appl Stat 49(2):2. https://doi.org/10.1080/02664763.2020.1815676
Lim SS, Vos T, Flaxman AD, Danaei G, Shibuya K, Adair-Rohani H, Amann M, Anderson HR, Andrews KG, Aryee M, Atkinson C, Bacchus LJ, Bahalim AN, Balakrishnan K, Balmes J, Barker-Collo S, Baxter A, Bell ML, Blore JD, Memish ZA (2012) A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet (London, England) 380(9859):9859. https://doi.org/10.1016/S0140-6736(12)61766-8
Livingstone-Banks J, Lindson N, Hartmann-Boyce J, Aveyard P (2022) Effects of interventions to combat tobacco addiction: Cochrane update of 2019 and 2020 reviews. Addiction (Abingdon, England) 117(6):6. https://doi.org/10.1111/add.15769
Martin B, Ji S, Maudsley S, Mattson MP (2010) “Control” laboratory rodents are metabolically morbid: Why it matters. Proc Natl Acad Sci USA 107(14):14. https://doi.org/10.1073/pnas.0912955107
Matta SG, Balfour DJ, Benowitz NL, Boyd RT, Buccafusco JJ, Caggiula AR, Craig CR, Collins AC, Damaj MI, Donny EC, Gardiner PS, Grady SR, Heberlein U, Leonard SS, Levin ED, Lukas RJ, Markou A, Marks MJ, McCallum SE, Zirger JM (2007) Guidelines on nicotine dose selection for in vivo research. Psychopharmacology 190(3):3. https://doi.org/10.1007/s00213-006-0441-0
Nicolas C, Russell TI, Pierce AF, Maldera S, Holley A, You Z-B, McCarthy MM, Shaham Y, Ikemoto S (2019) Incubation of cocaine craving after intermittent-access self-administration: Sex differences and estrous cycle. Biol Psychiatry 85(11):11. https://doi.org/10.1016/j.biopsych.2019.01.015
Oleson EB, Roberts DCS (2009) Behavioral economic assessment of price and cocaine consumption following self-administration histories that produce escalation of either final ratios or intake. Neuropsychopharmacology 34(3):3. https://doi.org/10.1038/npp.2008.195
O’Neal TJ, Nooney MN, Thien K, Ferguson SM (2020) Chemogenetic modulation of accumbens direct or indirect pathways bidirectionally alters reinstatement of heroin-seeking in high- but not low-risk rats. Neuropsychopharmacology 45(8):8. https://doi.org/10.1038/s41386-019-0571-9
Paterson NE, Markou A (2004) Prolonged nicotine dependence associated with extended access to nicotine self-administration in rats. Psychopharmacology 173(1–2):1–2. https://doi.org/10.1007/s00213-003-1692-7
Pogun S, Yararbas G, Nesil T, Kanit L (2017) Sex differences in nicotine preference. J Neurosci Res 95(1–2):1–2. https://doi.org/10.1002/jnr.23858
Reyes-Guzman CM, Pfeiffer RM, Lubin J, Freedman ND, Cleary SD, Levine PH, Caporaso NE (2017) Determinants of Light and Intermittent Smoking in the United States: Results from Three Pooled National Health Surveys. Cancer Epidemiol Biomark Prev 26(2):2. https://doi.org/10.1158/1055-9965.EPI-16-0028
Richardson NR, Roberts DC (1996) Progressive ratio schedules in drug self-administration studies in rats: a method to evaluate reinforcing efficacy. J Neurosci Meth 66(1):1–11. https://doi.org/10.1016/0165-0270(95)00153-0
Rieder A, Kunze U, Groman E, Kiefer I, Schoberberger R (2001) Nocturnal sleep-disturbing nicotine craving: A newly described symptom of extreme nicotine dependence. Acta Med Austriaca 28(1):21–22. https://doi.org/10.1046/j.1563-2571.2001.01005.x
Risner ME, Goldberg SR (1983) A comparison of nicotine and cocaine self-administration in the dog: Fixed-ratio and progressive-ratio schedules of intravenous drug infusion. J Pharmacol Exp Ther 224(2):2
Rosecrans JA (1971) Effects of nicotine on brain area 5-hydroxytryptamine function in male and female rats separated for differences of activity. Eur J Pharmacol 16(1):123–127. https://doi.org/10.1016/0014-2999(71)90067-7
Rowland NE (2007) Food or fluid restriction in common laboratory animals: Balancing welfare considerations with scientific inquiry. Comp Med 57(2):2
Samaha A-N, Yau W-YW, Yang P, Robinson TE (2005) Rapid delivery of nicotine promotes behavioral sensitization and alters its neurobiological impact. Biol Psychiatry 57(4):4. https://doi.org/10.1016/j.biopsych.2004.11.040
Samaha A-N, Minogianis E-A, Nachar W (2011) Cues paired with either rapid or slower self-administered cocaine injections acquire similar conditioned rewarding properties. PloS One 6(10):10. https://doi.org/10.1371/journal.pone.0026481
Samaha A-N, Khoo SY-S, Ferrario CR, Robinson TE (2021) Dopamine “ups and downs” in addiction revisited. Trends Neurosci 44(7):7. https://doi.org/10.1016/j.tins.2021.03.003
Sanchez V, Moore CF, Brunzell DH, Lynch WJ (2014) Sex differences in the effect of wheel running on subsequent nicotine-seeking in a rat adolescent-onset self-administration model. Psychopharmacology 231(8):8. https://doi.org/10.1007/s00213-013-3359-3
Sannerud CA, Prada J, Goldberg DM, Goldberg SR (1994) The effects of sertraline on nicotine self-administration and food-maintained responding in squirrel monkeys. Eur J Pharmacol 271(2–3):2–3. https://doi.org/10.1016/0014-2999(94)90807-9
Schechter MD, Jellinek P (1975) Evidence for a cortical locus for the stimulus effect of nicotine. Eur J Pharmacol 34(1):1. https://doi.org/10.1016/0014-2999(75)90226-5
Shaham Y, Adamson LK, Grocki S, Corrigall WA (1997) Reinstatement and spontaneous recovery of nicotine seeking in rats. Psychopharmacology 130(4):396–403. https://doi.org/10.1007/s002130050256
Shiffman S, Dunbar MS, Li X, Scholl SM, Tindle HA, Anderson SJ, Ferguson SG (2014) Smoking patterns and stimulus control in intermittent and daily smokers. PLoS One 9(3):3. https://doi.org/10.1371/journal.pone.0089911
Shiffman S, Scholl SM, Mao J, Ferguson SG, Hedeker D, Primack B, Tindle HA (2020) Using nicotine gum to assist nondaily smokers in quitting: A randomized clinical trial - PubMed. Nicotine Tob Res 22(3):390–397. https://doi.org/10.1093/ntr/ntz090
Shivange AV, Borden PM, Muthusamy AK, Nichols AL, Bera K, Bao H, Bishara I, Jeon J, Mulcahy MJ, Cohen B, O’Riordan SL, Kim C, Dougherty DA, Chapman ER, Marvin JS, Looger LL, Lester HA (2019) Determining the pharmacokinetics of nicotinic drugs in the endoplasmic reticulum using biosensors. J Gen Physiol 151(6):738–757. https://doi.org/10.1085/jgp.201812201
Singer BF, Fadanelli M, Kawa AB, Robinson TE (2018) Are Cocaine-Seeking “Habits” Necessary for the Development of Addiction-Like Behavior in Rats? J Neurosci 38(1):1. https://doi.org/10.1523/JNEUROSCI.2458-17.2017
Sorge RE, Clarke PBS (2009) Rats self-administer intravenous nicotine delivered in a novel smoking-relevant procedure: Effects of dopamine antagonists. J Pharmacol Exp Ther 330(2):2. https://doi.org/10.1124/jpet.109.154641
Stennett A, Krebs NM, Liao J, Richie JP, Muscat JE (2018) Ecological momentary assessment of smoking behaviors in native and converted intermittent smokers. Am J Addict 27(2):2. https://doi.org/10.1111/ajad.12690
Swalve N, Smethells JR, Carroll ME (2016) Sex differences in the acquisition and maintenance of cocaine and nicotine self-administration in rats. Psychopharmacology 233(6):6. https://doi.org/10.1007/s00213-015-4183-8
Tapia MA, Jin X-T, Tucker BR, Thomas LN, Walker NB, Kim VJ, Albertson SE, Damuka N, Krizan I, Edassery S, Savas JN, Solingapuram Sai KK, Jones SR, Drenan RM (2022) Relapse-like behavior and nAChR sensitization following intermittent access nicotine self-administration. Neuropharmacology 212:109066. https://doi.org/10.1016/j.neuropharm.2022.109066
World Health Organisation (2020) World Health Organisation. URL: https://www.who.int/en/news-room/fact-sheets/detail/tobacco [accessed 2021–01–05]
Watkins SS, Epping-Jordan MP, Koob GF, Markou A (1999) Blockade of nicotine self-administration with nicotinic antagonists in rats. Pharmacol Biochem Behav 62(4):4. https://doi.org/10.1016/s0091-3057(98)00226-3
Weeks JR (1962) Experimental morphine addiction: Method for automatic intravenous injections in unrestrained rats. Science (New York, N.Y.) 138(3537):3537. https://doi.org/10.1126/science.138.3537.143
Zhai D, van Stiphout R, Schiavone G, De Raedt W, Van Hoof C (2022) Characterizing and modeling smoking behavior using automatic smoking event detection and mobile surveys in naturalistic environments: Observational study. JMIR mHealth and uHealth 10(2):2. https://doi.org/10.2196/28159
Zimmer BA, Dobrin CV, Roberts DCS (2011) Brain-cocaine concentrations determine the dose self-administered by rats on a novel behaviorally dependent dosing schedule. Neuropsychopharmacology 36(13):13. https://doi.org/10.1038/npp.2011.165
Zimmer BA, Oleson EB, Roberts DC (2012) The motivation to self-administer is increased after a history of spiking brain levels of cocaine. Neuropsychopharmacology 37(8):8. https://doi.org/10.1038/npp.2012.37
Acknowledgements
We thank Dr. Paul Clarke for reading and commenting an earlier version of this manuscript. A version of this manuscript is posted on the preprint server, bioRxiv (BIORXIV/2023/563876).
Funding
This research was supported by a grant from the Canadian Institutes of Health Research (grant number 157572) to ANS. HEA was supported by a PhD scholarship from the Libyan-North American Scholarship Program of the Ministry of Higher Education in Libya.
Author information
Authors and Affiliations
Contributions
HEA and ANS designed the research. HEA performed all experiments and analyzed all data with guidance from ANS. VJ modelled nicotine concentrations. HEA and ANS wrote the article with revisions from VJ.
Corresponding author
Ethics declarations
Competing interests
The authors have nothing to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Algallal, H.E., Jacquemet, V. & Samaha, AN. Intermittent nicotine access is as effective as continuous access in promoting nicotine seeking and taking in rats. Psychopharmacology (2024). https://doi.org/10.1007/s00213-024-06546-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00213-024-06546-4