Abstract
Rationale
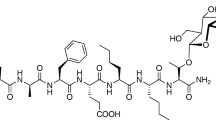
Tianeptine is a mu-opioid receptor (MOR) agonist with increasing reports of abuse in human populations. Preclinical data regarding the abuse potential and other opioid-like adverse effects of tianeptine at supratherapeutic doses are sparse.
Objectives
The present study evaluated tianeptine in a rat model of abuse potential assessment and in mouse models of motor, gastrointestinal, and respiratory adverse effects.
Methods
Abuse potential was assessed in adult male Sprague–Dawley rats using an intracranial self-stimulation (ICSS) procedure to determine effects of acute and repeated tianeptine on responding for electrical brain stimulation. Male ICR mice were used to determine the effects of tianeptine in assays of locomotor behavior and gastrointestinal motility. Male Swiss-Webster mice were monitored for respiratory changes using whole-body plethysmography.
Results
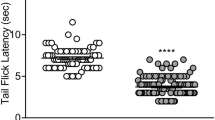
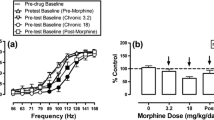
In rats, acute tianeptine produced weak and delayed evidence for abuse-related ICSS facilitation at an intermediate dose (10 mg/kg, IP) and pronounced, naltrexone-preventable ICSS depression at a higher dose (32 mg/kg, IP). Repeated 7-day tianeptine (10 and 32 mg/kg/day, IP) produced no increase in abuse-related ICSS facilitation, only modest tolerance to ICSS depression, and no evidence of physical dependence. In mice, tianeptine produced dose-dependent, naltrexone-preventable locomotor activation. Tianeptine (100 mg/kg, SC) also significantly inhibited gastrointestinal motility and produced naloxone-reversible respiratory depression.
Conclusions
Tianeptine presents as a MOR agonist with resistance to tolerance and dependence in our ICSS assay in rats, and it has lower abuse potential by this metric than many commonly abused opioids. Nonetheless, tianeptine produces MOR agonist-like acute adverse effects that include motor impairment, constipation, and respiratory depression.





Similar content being viewed by others
References
Alici T, Kayir H, Aygoren MO, Saglam E, Uzbay IT (2006) Discriminative stimulus properties of tianeptine. Psychopharmacology 183(4):446–451. https://doi.org/10.1007/s00213-005-0210-5
Altarifi AA, David B, Muchhala KH, Blough BE, Akbarali H, Negus SS (2017) Effects of acute and repeated treatment with the biased mu opioid receptor agonist TRV130 (oliceridine) on measures of antinociception, gastrointestinal function, and abuse liability in rodents. J Psychopharmacol 31(6):730–739. https://doi.org/10.1177/0269881116689257
Altarifi AA, Miller LL, Stevens Negus S (2012) Role of µ-opioid receptor reserve and µ-agonist efficacy as determinants of the effects of µ-agonists on intracranial self-stimulation in rats. Behav Pharmacol 23(7):678–692. https://doi.org/10.1097/FBP.0b013e328358593c
Altarifi AA, Moerke MJ, Alsalem MI, Negus SS (2020) Preclinical assessment of tramadol abuse potential: effects of acute and repeated tramadol on intracranial self-stimulation in rats. J Psychopharmacol 10. https://doi.org/10.1177/0269881120944153
Altarifi AA, Negus SS (2011) Some determinants of morphine effects on intracranial self-stimulation in rats: dose, pretreatment time, repeated treatment, and rate dependence. Behav Pharmacol 22(7):663–673. https://doi.org/10.1097/FBP.0b013e32834aff54
Altarifi AA, Rice KC, Negus SS (2013) Abuse-related effects of µ-opioid analgesics in an assay of intracranial self-stimulation in rats: modulation by chronic morphine exposure. Behavioural Pharmacology 24(5 and 6):459–470. https://doi.org/10.1097/FBP.0b013e328364c0bd
Baird TR, Davies RA, Glennon RA, Peace MR, Negus SS (2021) A strategy to prioritize emerging drugs of abuse for analysis: abuse liability testing using intracranial self-stimulation (ICSS) in rats and validation with α-pyrrolidinohexanophenone (α-PHP). Emerging Trends in Drugs, Addictions, and Health 1:100004. https://doi.org/10.1016/j.etdah.2021.100004
Bakota EL, Samms WC, Gray TR, Oleske DA, Hines MO (2018) Case reports of fatalities involving tianeptine in the United States. J Anal Toxicol 42(7):503–509. https://doi.org/10.1093/jat/bky023
Baumann MH, Kopajtic TA, Madras BK (2018) Pharmacological research as a key component in mitigating the opioid overdose crisis. Trends Pharmacol Sci 39(12):995–998. https://doi.org/10.1016/j.tips.2018.09.006
Bernard K, Penelaud P-F, Mocaër E, Donazzolo Y (2011) Absence of psychostimulant effects of a supratherapeutic dose of tianeptine: a placebo-controlled study versus methylphenidate in young healthy volunteers. J Clin Psychopharmacol 31(4):441–448. https://doi.org/10.1097/JCP.0b013e3182217a50
Bolton VN, Bassil AK, Lee K, Sanger GJ (2008) Inhibition by tianeptine of neuronally mediated contractions in the rat isolated gastrointestinal tract. Pharmacol Res 57(5):339–343. https://doi.org/10.1016/j.phrs.2008.03.006
Botz-Zapp CA, Foster SL, Pulley DM, Hempel B, Bi G-H, Xi Z-X, Newman AH, Weinshenker D, Manvich DF (2021) Effects of the selective dopamine D3 receptor antagonist PG01037 on morphine-induced hyperactivity and antinociception in mice. Behav Brain Res 415:113506. https://doi.org/10.1016/j.bbr.2021.113506
Cavalla D, Chianelli F, Korsak A, Hosford PS, Gourine AV, Marina N (2015) Tianeptine prevents respiratory depression without affecting analgesic effect of opiates in conscious rats. Eur J Pharmacol 761:268–272. https://doi.org/10.1016/j.ejphar.2015.05.067
Crowley ML, Restrepo LF, Gamez-Jimenez LR, Patel A, Braun T, Pallares VLC, Ho NP, Reeves ME, McCurdy CR, McMahon LR, Hiranita T (2021) The use of hypercapnic conditions to assess opioid-induced respiratory depression in rats. J Pharmacol Toxicol Methods 111:107101. https://doi.org/10.1016/j.vascn.2021.107101
Dempsey SK, Poklis JL, Sweat K, Cumpston K, Wolf CE (2017) Acute toxicity from intravenous use of the tricyclic antidepressant tianeptine. J Anal Toxicol 41(6):547–550. https://doi.org/10.1093/jat/bkx034
Diester CM, Santos EJ, Moerke MJ, Negus SS (2021) Behavioral battery for testing candidate analgesics in mice I Validation with positive and negative controls. J Pharmacol Exp Ther 377(2):232–241. https://doi.org/10.1124/jpet.120.000464
Duberstein JL, Kaufman DM (1971) A clinical study of an epidemic of heroin intoxication and heroin-induced pulmonary edema. Am J Med 51(6):704–714. https://doi.org/10.1016/0002-9343(71)90298-1
El Zahran T, Schier J, Glidden E, Kieszak S, Law R, Bottei E, Aaron C, King A, Chang A (2018) Characteristics of Tianeptine exposures reported to the national poison data system—United States 2000–2017. MMWR Morbidity and Mortality Weekly Report 67(30):815–818. https://doi.org/10.15585/mmwr.mm6730a2
Frischknecht H-R, Siegfried B, Riggio G, Waser PG (1983) Inhibition of morphine-induced analgesia and locomotor activity in strains of mice: a comparison of long-acting opiate antagonists. Pharmacol Biochem Behav 19(6):939–944. https://doi.org/10.1016/0091-3057(83)90395-7
Funada M, Suzuki T, Narita M, Misawa M, Nagase H (1993) Modification of morphine-induced locomotor activity by pertussis toxin: biochemical and behavioral studies in mice. Brain Res 619(1–2):163–172. https://doi.org/10.1016/0006-8993(93)91608-U
Gassaway MM, Rives M-L, Kruegel AC, Javitch JA, Sames D (2014) The atypical antidepressant and neurorestorative agent tianeptine is a μ-opioid receptor agonist. Transl Psychiatry 4(7):e411–e411. https://doi.org/10.1038/tp.2014.30
Gummin DD, Mowry JB, Spyker DA, Brooks DE, Osterthaler KM, Banner W (2018) 2017 annual report of the american association of poison control centers’ National Poison Data System (NPDS): 35th annual report. Clin Toxicol 56(12):1213–1415. https://doi.org/10.1080/15563650.2018.1533727
Hill R, Disney A, Conibear A, Sutcliffe K, Dewey W, Husbands S, Bailey C, Kelly E, Henderson G (2018) The novel μ-opioid receptor agonist PZM21 depresses respiration and induces tolerance to antinociception: PZM21 depresses respiration. Br J Pharmacol 175(13):2653–2661. https://doi.org/10.1111/bph.14224
Hill R, Lyndon A, Withey S, Roberts J, Kershaw Y, MacLachlan J, Lingford-Hughes A, Kelly E, Bailey C, Hickman M, Henderson G (2016) Ethanol reversal of tolerance to the respiratory depressant effects of morphine. Neuropsychopharmacology 41(3):762–773. https://doi.org/10.1038/npp.2015.201
Hill R, Santhakumar R, Dewey W, Kelly E, Henderson G (2020) Fentanyl depression of respiration: comparison with heroin and morphine. Br J Pharmacol 177(2):254–265. https://doi.org/10.1111/bph.14860
Kasper S, Olié JP (2002) A meta-analysis of randomized controlled trials of tianeptine versus SSRI in the short-term treatment of depression. Eur Psychiatry 17(S3):331s–340s. https://doi.org/10.1016/S0924-9338(02)00651-X
Lauhan R, Hsu A, Alam A, Beizai K (2018) Tianeptine abuse and dependence: case report and literature review. Psychosomatics 59(6):547–553. https://doi.org/10.1016/j.psym.2018.07.006
Malen C, Danree B, Poignant J-C (1973) Tricyclic compounds (United States Patent Office Patent No. 3,758,528)
Miller LL, Altarifi AA, Negus SS (2015) Effects of repeated morphine on intracranial self-stimulation in male rats in the absence or presence of a noxious pain stimulus. Exp Clin Psychopharmacol 23(5):405–414. https://doi.org/10.1037/pha0000042
Moerke MJ, Negus SS (2019) Role of agonist efficacy in exposure-induced enhancement of Mu opioid reward in rats. Neuropharmacology 151:180–188. https://doi.org/10.1016/j.neuropharm.2019.03.020
Moerke MJ, Negus SS (2021) Temporal parameters of enhanced opioid reward after initial opioid exposure in rats. Psychopharmacology 238(3):725–734. https://doi.org/10.1007/s00213-020-05725-3
Negus SS, Miller LL (2014) Intracranial self-stimulation to evaluate abuse potential of drugs. Pharmacol Rev 66(3):869–917. https://doi.org/10.1124/pr.112.007419
Negus SS, Moerke MJ (2019) Determinants of opioid abuse potential: Insights using intracranial self-stimulation. Peptides 112:23–31. https://doi.org/10.1016/j.peptides.2018.10.007
Osborn MD, Lowery JJ, Skorput AGJ, Giuvelis D, Bilsky EJ (2010) In vivo characterization of the opioid antagonist nalmefene in mice. Life Sci 86(15–16):624–630. https://doi.org/10.1016/j.lfs.2010.02.013
Proença P, Teixeira H, Pinheiro J, Monsanto PV, Vieira DN (2007) Fatal intoxication with tianeptine (Stablon®). Forensic Sci Int 170(2–3):200–203. https://doi.org/10.1016/j.forsciint.2007.03.035
Rethy CR, Smith CB, Villarreal JE (1971) Effects of narcotic analgesics upon the locomotor activity and brain catecholamine content of the mouse. J Pharmacol Exp Ther 176(2):472–479
Ross GR, Gabra BH, Dewey WL, Akbarali HI (2008) Morphine tolerance in the mouse ileum and colon. J Pharmacol Exp Ther 327(2):561–572. https://doi.org/10.1124/jpet.108.143438
Rushton W, Whitworth B, Brown J, Kurz M, Rivera J (2021) Characteristics of tianeptine effects reported to a poison control center: a growing threat to public health. Clin Toxicol 59(2):152–157. https://doi.org/10.1080/15563650.2020.1781151
Samuels BA, Nautiyal KM, Kruegel AC, Levinstein MR, Magalong VM, Gassaway MM, Grinnell SG, Han J, Ansonoff MA, Pintar JE, Javitch JA, Sames D, Hen R (2017) The behavioral effects of the antidepressant tianeptine require the Mu-opioid receptor. Neuropsychopharmacology 42(10):2052–2063. https://doi.org/10.1038/npp.2017.60
Severino AL, Mittal N, Hakimian JK, Velarde N, Minasyan A, Albert R, Torres C, Romaneschi N, Johnston C, Tiwari S, Lee AS, Taylor AM, Gavériaux-Ruff C, Kieffer BL, Evans CJ, Cahill CM, Walwyn WM (2020) μ-opioid receptors on distinct neuronal populations mediate different aspects of opioid reward-related behaviors. Eneuro 7(5). https://doi.org/10.1523/ENEURO.0146-20.2020
Smith KE, Rogers JM, Strickland JC, Epstein DH (2021) When an obscurity becomes trend: Social-media descriptions of tianeptine use and associated atypical drug use. Am J Drug Alcohol Abuse :1–12. https://doi.org/10.1080/00952990.2021.1904408
Urs NM, Caron MG (2014) The physiological relevance of functional selectivity in dopamine signalling. Int J Obesity Supplements 4(S1):S5–S8. https://doi.org/10.1038/ijosup.2014.3
U.S. Food and Drug Administration (2018) FDA warns marketers of products labeled as dietary supplements that contain tianeptine for making unproven claims to treat serious conditions, including opioid use disorder. FDA; FDA. https://www.fda.gov/news-events/press-announcements/fda-warns-marketers-products-labeled-dietary-supplements-contain-tianeptine-making-unproven-claims
Uzbay TI, Kayir H, Ceyhan M (2007) Effects of Tianeptine on onset time of pentylenetetrazole-induced seizures in mice: possible role of adenosine A1 receptors. Neuropsychopharmacology 32(2):412–416. https://doi.org/10.1038/sj.npp.1301143
Varshneya NB, Hassanien SH, Holt MC, Stevens DL, Layle NK, Bassman JR, Iula DM, Beardsley PM (2022) Respiratory depressant effects of fentanyl analogs are opioid receptor-mediated. Biochem Pharmacol 195:114805. https://doi.org/10.1016/j.bcp.2021.114805
Varshneya NB, Walentiny DM, Moisa LT, Walker TD, Akinfiresoye LR, Beardsley PM (2019) Opioid-like antinociceptive and locomotor effects of emerging fentanyl-related substances. Neuropharmacology 151:171–179. https://doi.org/10.1016/j.neuropharm.2019.03.023
Varshneya NB, Walentiny DM, Moisa LT, Walker TD, Akinfiresoye LR, Beardsley PM (2021) Fentanyl-related substances elicit antinociception and hyperlocomotion in mice via opioid receptors. Pharmacol Biochem Behav 208:173242. https://doi.org/10.1016/j.pbb.2021.173242
Wagstaff AJ, Ormrod D, Spencer CM (2001) Tianeptine: a review of its use in depressive disorders. CNS Drugs 15(3):231–259. https://doi.org/10.2165/00023210-200115030-00006
Webster LR (2015) Opioid-induced constipation. Pain Med 16(suppl 1):S16–S21. https://doi.org/10.1111/pme.12911
Wilde MI, Benfield P (1995) Tianeptine: a review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy in depression and coexisting anxiety and depression. Drugs 49(3):411–439. https://doi.org/10.2165/00003495-199549030-00007
Funding
This project was supported by Award No. P30DA033934, awarded by the National Institute on Drug Abuse, and Award No. 2019-R2-CX-0046, awarded by the National Institute of Justice, Office of Justice Programs, US Department of Justice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclaimer
The opinions, findings, and conclusions or recommendations expressed in this publication are those of the authors and do not necessarily reflect those of the National Institutes of Health or the Department of Justice.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Baird, T.R., Akbarali, H.I., Dewey, W.L. et al. Opioid-like adverse effects of tianeptine in male rats and mice. Psychopharmacology 239, 2187–2199 (2022). https://doi.org/10.1007/s00213-022-06093-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-022-06093-w