Abstract
Rationale
Identifying the long-term neurocognitive implications of opioid addiction may further our understanding of the compulsive nature of this brain disorder. The aim of this study was to examine the effects of paternal adolescent opiate exposure on cognitive performance (visual attention, impulsivity, and compulsivity) in the next generation.
Methods
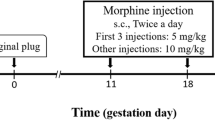
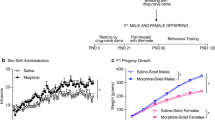
Male Wistar rats received escalating doses of morphine (2.5–25 mg/kg, s.c.) or saline for 10 days during adolescence (P30–39). In adulthood (P70–80), these rats were allowed to mate with drug-naive females. Male offspring from morphine- and saline-exposed sires, once in adulthood, were trained and tested in the 5-choice serial reaction time test (5-CSRTT) to evaluate their cognitive abilities under baseline, drug-free conditions as well as following acute (1, 3, 5 mg/kg morphine) and subchronic morphine (5 mg/kg morphine for 5 days) treatment. Behavioral effects of the opioid receptor antagonist naloxone were also assessed.
Results
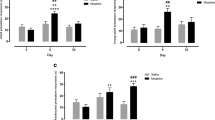
Morphine-sired offspring exhibited delayed learning when the shortest stimulus duration (1 s) was introduced, i.e., when cognitive load was highest. These subjects also exhibited a reduced ability to exert inhibitory control, as reflected by increased premature and perseverative responding under drug-free baseline conditions in comparison to saline-sired rats. These impairments could not be reversed by administration of naloxone. Moreover, impulsive behavior was further enhanced in morphine-sired rats following acute and subchronic morphine treatment.
Conclusion
Paternal opiate exposure during adolescence was found to primarily impair inhibitory control in male progeny. These results further our understanding of the long-term costs and risk of opioid abuse, extending across generations.





Similar content being viewed by others
References
Alaee E, Moazen P, Pattij T, Semnanian S, Azizi H (2021) Prenatal exposure to morphine impairs attention and impulsivity in adult rats. Psychopharmacology. https://doi.org/10.1007/s00213-021-05888-7
Ashabi G, Sadat-Shirazi M-S, Akbarabadi A, Vousooghi N, Kheiri Z, Toolee H, Khalifeh S, Zarrindast M-R (2018) Is the nociception mechanism altered in offspring of morphine-abstinent rats? J Pain 19:529–541
Azadi M, Azizi H, Haghparast A (2019) Paternal exposure to morphine during adolescence induces reward-resistant phenotype to morphine in male offspring. Brain Res Bull 147:124–132
Azadi M, Gompf HS, Azizi H (2020) Paternal exposure to morphine during adolescence potentiates morphine withdrawal in male offspring: involvement of the lateral paragigantocellularis nucleus. J Psychopharmacol 34:1289–1299
Azadi M, Zare M, Pachenari N, Shojaei A, Semnanian S, Azizi H (2021) Sex-specific transgenerational effects of adolescent morphine exposure on short-term memory and anxiety-like behavior: male linage. Neuroscience Letters: 136111.
Baldacchino A, Balfour DJ, Passetti F, Humphris G, Matthews K (2012) Neuropsychological consequences of chronic opioid use: a quantitative review and meta-analysis. Neurosci Biobehav Rev 36:2056–2068
Bari A, Dalley JW, Robbins TW (2008) The application of the 5-choice serial reaction time task for the assessment of visual attentional processes and impulse control in rats. Nat Protoc 3:759–767
Befort K, Mahoney MK, Chow C, Hayton SJ, Kieffer BL, Olmstead MC (2011) Effects of delta opioid receptors activation on a response inhibition task in rats. Psychopharmacology 214:967–976
Bird A (2007) Perceptions of epigenetics. Nature 447:396
Brent DA, Hur K, Gibbons RD (2019) Association between parental medical claims for opioid prescriptions and risk of suicide attempt by their children. JAMA Psychiat 76:941–947
Byrnes JJ, Babb JA, Scanlan VF, Byrnes EM (2011) Adolescent opioid exposure in female rats: transgenerational effects on morphine analgesia and anxiety-like behavior in adult offspring. Behav Brain Res 218:200–205
Byrnes JJ, Johnson NL, Carini LM, Byrnes EM (2013) Multigenerational effects of adolescent morphine exposure on dopamine D2 receptor function. Psychopharmacology 227:263–272
Casey B, Jones RM (2010) Neurobiology of the adolescent brain and behavior: implications for substance use disorders. J Am Acad Child Adolesc Psychiatry 49:1189–1201
Clark L, Robbins TW, Ersche KD, Sahakian BJ (2006) Reflection impulsivity in current and former substance users. Biol Psychiatry 60:515–522
Counotte DS, Spijker S, Van de Burgwal LH, Hogenboom F, Schoffelmeer AN, De Vries TJ, Smit AB, Pattij T (2009) Long-lasting cognitive deficits resulting from adolescent nicotine exposure in rats. Neuropsychopharmacology 34:299–306
Dalley JW, Ersche KD (2019) Neural circuitry and mechanisms of waiting impulsivity: relevance to addiction. Philos Trans R Soc B 374:20180145
Dalley JW, Robbins TW (2017) Fractionating impulsivity: neuropsychiatric implications. Nat Rev Neurosci 18:158–171
Edition F (2013) Diagnostic and statistical manual of mental disorders. Am Psychiatric Assoc 21.
Edward (2000) Psychiatric disorders and impairment in the children of opiate addicts: prevalances and distribution by ethnicity. American Journal on Addictions 9:232–241
Ellis AS, Toussaint AB, Knouse MC, Thomas AS, Bongiovanni AR, Mayberry HL, Bhakta S, Peer K, Bangasser DA, Wimmer ME (2020) Paternal morphine self-administration produces object recognition memory deficits in female, but not male offspring. Psychopharmacology: 1–13.
Evenden JL (1999) Varieties of impulsivity. Psychopharmacology 146:348–361
Gladwin TE, Figner B, Crone EA, Wiers RW (2011) Addiction, adolescence, and the integration of control and motivation. Dev Cogn Neurosci 1:364–376
Goldberg LR, Gould TJ (2018) Multigenerational and transgenerational effects of paternal exposure to drugs of abuse on behavioral and neural function. European Journal of Neuroscience.
Goldberg LR, Gould TJ (2019) Multigenerational and transgenerational effects of paternal exposure to drugs of abuse on behavioral and neural function. Eur J Neurosci 50:2453–2466
Hamidullah S, Thorpe HH, Frie JA, Mccurdy RD, Khokhar JY (2020) Adolescent Substance Use and the Brain: Behavioral, Cognitive and Neuroimaging Correlates. Front Hum Neurosci 14:298
He F, Lidow IA, Lidow MS (2006) Consequences of paternal cocaine exposure in mice. Neurotoxicol Teratol 28:198–209
Holloway ZR, Hawkey AB, Pippin E, White H, Wells C, Kenou B, Rezvani AH, Murphy SK, Levin ED (2020) Paternal factors in neurodevelopmental toxicology: THC exposure of male rats causes long-lasting neurobehavioral effects in their offspring. Neurotoxicology 78:57–63
Infante MA, Nguyen-Louie TT, Worley M, Courtney KE, Coronado C, Jacobus J (2020) Neuropsychological trajectories associated with adolescent alcohol and cannabis use: a prospective 14-year study. Journal of the International Neuropsychological Society: JINS 26:480
Jenab S, Morris PL (2000) Interleukin-6 regulation of kappa opioid receptor gene expression in primary sertoli cells. Endocrine 13:11–15
Johnson JL, Leff M (1999) Children of substance abusers: Overview of research findings. Pediatrics 103:1085–1099
Johnson KR, Boomhower SR, Newland MC (2019) Behavioral effects of chronic WIN 55,212–2 administration during adolescence and adulthood in mice. Exp Clin Psychopharmacol 27:348
Kirby KN, Petry NM, Bickel WK (1999) Heroin addicts have higher discount rates for delayed rewards than non-drug-using controls. J Exp Psychol Gen 128:78–87
Le Q, Yan B, Yu X, Li Y, Song H, Zhu H, Hou W, Ma D, Wu F, Zhou Y (2017) Drug-seeking motivation level in male rats determines offspring susceptibility or resistance to cocaine-seeking behaviour. Nat Commun 8:1–13
Maguire D, Henson C, France C (2016) Daily morphine administration increases impulsivity in rats responding under a 5-choice serial reaction time task. Br J Pharmacol 173:1350–1362
Mahoney MK, Silveira MM, Olmstead MC (2013) Increased impulsive action in rats: effects of morphine in a short and long fixed-delay response inhibition task. Psychopharmacology 230:569–577
Masrouri H, Azadi M, Semnanian S, Azizi H (2020) Maternal deprivation induces persistent adaptations in putative dopamine neurons in rat ventral tegmental area: in vivo electrophysiological study. Exp Brain Res.
Masrouri H, Azadi M, Semnanian S, Azizi H (2021) Early life maternal deprivation attenuates morphine induced inhibition in lateral paragigantocellularis neurons in adult rats. Brain Res Bull 169:128–135
McCarthy DM, Morgan Jr TJ, Lowe SE, Williamson MJ, Spencer TJ, Biederman J, Bhide PG (2018) Nicotine exposure of male mice produces behavioral impairment in multiple generations of descendants. PLoS biology 16: e2006497.
Mills KL, Goddings A-L, Clasen LS, Giedd JN, Blakemore S-J (2014) The developmental mismatch in structural brain maturation during adolescence. Dev Neurosci 36:147–160
Mintzer MZ, Copersino ML, Stitzer ML (2005) Opioid abuse and cognitive performance. Drug Alcohol Depend 78:225–230
Moazen P, Azizi H, Salmanzadeh H, Semnanian S (2018) Adolescent morphine exposure induces immediate and long-term increases in impulsive behavior. Psychopharmacology 235:3423–3434
Moazen P, Torabi M, Azizi H, Fathollahi Y, Mirnajafi-Zadeh J, Semnanian S (2020) The locus coeruleus noradrenergic system gates deficits in visual attention induced by chronic pain. Behavioural Brain Research: 112600.
Mychasiuk R, Metz GA (2016) Epigenetic and gene expression changes in the adolescent brain: What have we learned from animal models? Neurosci Biobehav Rev 70:189–197
Nunes EV, Weissman MM, Goldstein RB, McAVAY G, Seracini AM, Verdeli H, Wickramaratne PJ (1998) Psychopathology in children of parents with opiate dependence and/or major depression. J Am Acad Child Adolesc Psychiatry 37:1142–1151
Pachenari N, Azizi H, Ghasemi E, Azadi M, Semnanian S (2018) Exposure to opiates in male adolescent rats alters pain perception in the male offspring. Behav Pharmacol 29:255–260
Pachenari N, Azizi H, Semnaniann S (2019) Adolescent morphine exposure in male rats alters the electrophysiological properties of locus coeruleus neurons of the male offspring. Neuroscience 410:108–117
Parvaresh N, Mazhari S, Nazari-Noghabi M (2015) Frequency of psychiatric disorders in children of opioid or methamphetamine-dependent patients. Addict Health 7:140
Pattij T, De Vries TJ (2013) The role of impulsivity in relapse vulnerability. Curr Opin Neurobiol 23:700–705
Pattij T, Schetters D, Janssen MC, Wiskerke J, Schoffelmeer AN (2009) Acute effects of morphine on distinct forms of impulsive behavior in rats. Psychopharmacology 205:489–502
Pattij T, Vanderschuren LJ (2020) The neuropharmacology of impulsive behaviour, an update. Recent Advances in Research on Impulsivity and Impulsive Behaviors: 3–22.
Renaud SM, Fountain SB (2016) Transgenerational effects of adolescent nicotine exposure in rats: Evidence for cognitive deficits in adult female offspring. Neurotoxicol Teratol 56:47–54
Reynolds LM, Yetnikoff L, Pokinko M, Wodzinski M, Epelbaum JG, Lambert LC, Cossette M-P, Arvanitogiannis A, Flores C (2019) Early adolescence is a critical period for the maturation of inhibitory behavior. Cereb Cortex 29:3676–3686
Robbins T (2002) The 5-choice serial reaction time task: behavioural pharmacology and functional neurochemistry. Psychopharmacology 163:362–380
Sadat-Shirazi M-S, Asgari P, Mahboubi S, Zadeh-Tehrani SN, Ashabi G, Rohbani K, Sabzevari S, Soltani H, Khalifeh S, Zarrindast M-R (2020) Effect of morphine exposure on novel object memory of the offspring: The role of histone H3 and ΔFosB. Brain Res Bull 156:141–149
Salmanzadeh H, Ahmadi-Soleimani M, Azadi M, Halliwell RF, Azizi H (2021) Adolescent Substance abuse, transgenerational consequences and epigenetics. Current neuropharmacology.
Salmanzadeh H, Ahmadi-Soleimani SM, Pachenari N, Azadi M, Halliwell RF, Rubino T, Azizi H (2020) Adolescent drug exposure: a review of evidence for the development of persistent changes in brain function. Brain Res Bull 156:105–117
Sanchez‐Roige S, Peña‐Oliver Y, Ripley TL, Stephens DN (2014) Repeated ethanol exposure during early and late adolescence: double dissociation of effects on waiting and choice impulsivity. Alcoholism: Clinical and Experimental Research 38: 2579–2589.
Schiele MA, Bandelow B, Baldwin DS, Pini S, Domschke K (2020) A neurobiological framework of separation anxiety and related phenotypes. Eur Neuropsychopharmacol 33:45–57
Schippers MC, Binnekade R, Schoffelmeer AN, Pattij T, De Vries TJ (2012) Unidirectional relationship between heroin self-administration and impulsive decision-making in rats. Psychopharmacology 219:443–452
Selleck RA, Lake C, Estrada V, Riederer J, Andrzejewski M, Sadeghian K, Baldo BA (2015) Endogenous opioid signaling in the medial prefrontal cortex is required for the expression of hunger-induced impulsive action. Neuropsychopharmacology 40:2464–2474
Skóra MN, Pattij T, Beroun A, Kogias G, Mielenz D, de Vries T, Radwanska K, Müller CP (2020) Personality driven alcohol and drug abuse: New mechanisms revealed. Neuroscience & Biobehavioral Reviews.
Sliedrecht W, Roozen HG, Witkiewitz K, de Waart R, Dom G (2020) The association between impulsivity and relapse in patients with alcohol use disorder: a literature review. Alcohol and Alcoholism.
Spear LP (2018) Effects of adolescent alcohol consumption on the brain and behaviour. Nat Rev Neurosci 19:197
Vassoler F, Byrnes E, Pierce R (2014) The impact of exposure to addictive drugs on future generations: physiological and behavioral effects. Neuropharmacology 76:269–275
Vassoler FM, Byrnes EM (2021) Transgenerational effects on anxiety-like behavior following adolescent morphine exposure in female rats. Behavioural Brain Research 406: 113239.
Vassoler FM, Oliver DJ, Wyse C, Blau A, Shtutman M, Turner JR, Byrnes EM (2017) Transgenerational attenuation of opioid self-administration as a consequence of adolescent morphine exposure. Neuropharmacology 113:271–280
Vassoler FM, Sadri-Vakili G (2014) Mechanisms of transgenerational inheritance of addictive-like behaviors. Neuroscience 264:198–206
Vassoler FM, Toorie AM, Teceno DN, Walia P, Moore DJ, Patton TD, Byrnes EM (2020) Paternal morphine exposure induces bidirectional effects on cocaine versus opioid self-administration. Neuropharmacology 162: 107852.
Vassoler FM, White SL, Schmidt HD, Sadri-Vakili G, Pierce RC (2013) Epigenetic inheritance of a cocaine-resistance phenotype. Nat Neurosci 16:42–47
Vassoler FM, Wimmer ME (2020) Consequences of parental opioid exposure on neurophysiology, behavior, and health in the next generations. Cold Spring Harbor Perspectives in Medicine: a040436.
Vassoler FM, Wright SJ, Byrnes EM (2016) Exposure to opiates in female adolescents alters mu opiate receptor expression and increases the rewarding effects of morphine in future offspring. Neuropharmacology 103:112–121
Vathy I, Šlamberová R, Rimanóczy Á, Riley MA, Bar N (2003) Autoradiographic evidence that prenatal morphine exposure sex-dependently alters μ-opioid receptor densities in brain regions that are involved in the control of drug abuse and other motivated behaviors. Prog Neuropsychopharmacol Biol Psychiatry 27:381–393
Verdejo-Garcia A, Albein-Urios N (2020) Special issue on vulnerabilities to substance abuse impulsivity traits and neurocognitive mechanisms conferring vulnerability to substance use disorders. Neuropharmacology: 108402.
Vidal SI, Vandeleur C, Rothen S, Gholam-Rezaee M, Castelao E, Halfon O, Aubry J-M, Ferrero F, Preisig M (2012) Risk of mental disorders in children of parents with alcohol or heroin dependence: a controlled high-risk study. Eur Addict Res 18:253–264
Wimmer M, Briand L, Fant B, Guercio L, Arreola A, Schmidt H, Sidoli S, Han Y, Garcia B, Pierce R (2017) Paternal cocaine taking elicits epigenetic remodeling and memory deficits in male progeny. Mol Psychiatry 22:1641–1650
Wiskerke J, Schetters D, van Es IE, van Mourik Y, den Hollander BR, Schoffelmeer AN, Pattij T (2011) mu-Opioid receptors in the nucleus accumbens shell region mediate the effects of amphetamine on inhibitory control but not impulsive choice. J Neurosci 31:262–272
Wiskerke J, Van Mourik Y, Schetters D, Schoffelmeer AN, Pattij T (2012) On the role of cannabinoid CB1-and μ-opioid receptors in motor impulsivity. Front Pharmacol 3:108
Witte K (1995) The differential-allocation hypothesis: does the evidence support it? Evolution 49:1289–1290
Yohn NL, Bartolomei MS, Blendy JA (2015) Multigenerational and transgenerational inheritance of drug exposure: the effects of alcohol, opiates, cocaine, marijuana, and nicotine. Prog Biophys Mol Biol 118:21–33
Acknowledgements
We are immensely grateful to Professor Trevor Robbins (Department of Psychiatry, University of Cambridge) for helpful comments on the manuscript. In addition, we would like to thank the financial support of the Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors report no potential conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Azadi, M., Moazen, P., Wiskerke, J. et al. Preconception paternal morphine exposure leads to an impulsive phenotype in male rat progeny. Psychopharmacology 238, 3435–3446 (2021). https://doi.org/10.1007/s00213-021-05962-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-021-05962-0