Abstract
Rationale
F17464, a dopamine D3 receptor antagonist with relatively high D3 selectivity (70 fold vs D2 in vitro), exhibits an antipsychotic profile in preclinical studies, and therapeutic efficacy was demonstrated in a randomized placebo-controlled clinical trial in patients with schizophrenia (Bitter et al. Neuropsychopharmacology 44(11):1917–1924, 2019).
Objective
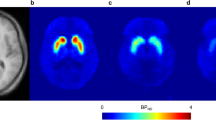
This open-label study in healthy male subjects aimed at characterizing F17464 binding to D3/D2 receptors and the time course of receptor occupancy using positron emission tomography (PET) imaging with a D3-preferring tracer, [11C]-(+)-PHNO.
Methods
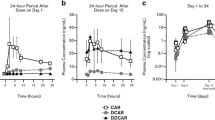
PET scans were performed at baseline and following a single 30 mg or 15 mg dose of F17464 (3 subjects/dose), and blood samples were collected for pharmacokinetic analysis. Receptor occupancy was calculated based upon reduction in binding potential of the tracer following F17464 administration. The relationship between plasma F17464 concentration and D3/D2 receptor occupancy was modeled and the plasma concentration corresponding to 50% receptor occupancy (EC50) calculated.
Results
Both doses of F17464 robustly blocked [11C]-(+)-PHNO D3 receptor binding, with substantial occupancy from 1 h post-administration, which increased at 6–9 h (89–98% and 79-87% for the 30 mg and 15 mg groups, respectively) and remained detectable at 22 h. In contrast, D2 binding was only modestly blocked at all time points (< 18%).
F17464 exhibited a combination of rapid peripheral kinetics and hysteresis (persistence of binding 22 h post-dose despite low plasma concentration). The best estimate of the EC50 was 19 ng ml−1 (~ 40 nM).
Conclusion
Overall, F17464 was strongly D3-selective in healthy volunteers, a unique profile for an antipsychotic candidate drug.




Similar content being viewed by others
References
Bitter I, Lieberman JA, Gaudoux F, Sokoloff P, Groc M, Chavda R et al (2019) Randomized, double-blind, placebo-controlled study of F17464, a preferential D3-antagonist, in the treatment of acute exacerbation of schizophrenia. Neuropsychopharmacology 44(11):1917–1924
Breden EL, Liu MT, Dean SR (2009) Metabolic and cardiacs side effects of second-generation antipsychotics: What every clinician should know. J Pharm Pract 22:478–488
Conley RR, Kelly DL (2005) Second generation antipsychotics for schizophrenia: a review of clinical pharmacology and medication-associated side effects. J Psychiatr Relat Sci 42:51–60
Fusar-Poli PF, Papanastasiou E, Stahl D, Rocchetti M, Carpenter W, Shergill S, McGuire P (2015) Treatments of negative symptoms in schizophrenia: meta-analysis of 168 randomized placebo-controlled trials. Schizophr Bull 41(4):892–899
Gallezot JD, Beaver JD, Gunn RN, Nabulsi N, Weinzimmer D, Singhal T, Slifstein M, Fowles K, Ding YS, Huang Y, Laruelle M, Carson RE, Rabiner EA (2012) Affinity and selectivity of [(1)(1)C]-(+)-PHNO for the D3 and D2 receptors in the rhesus monkey brain in vivo. Synapse 66(6):489–500
Gallezot JD, Zheng MQ, Lim K, Lin SF, Labaree D, Matuskey D, Huang Y, Ding YS, Carson RE, Malison RT (2014) Parametric imaging and test-retest variability of 11C-(+)-PHNO binding to D2/D3 dopamine receptors in humans on the high-resolution research tomograph PET scanner. J Nucl Med 55(6):960–966
Girgis RR, Slifstein M, D'Souza D, Lee Y, Periclou A, Ghahramani P, Laszlovszky I, Durgam S, Adham N, Nabulsi N, Huang Y, Carson RE, Kiss B, Kapás M, Abi-Dargham A, Rakhit A (2016) Preferential binding to dopamine D3 over D2 receptors by cariprazine in patients with schizophrenia using PET with the D3/D2 receptor ligand [(11)C]-(+)-PHNO. Psychopharmacol 233(19-20):3503–3512
Graff-Guerrero A, Mamo D, Shammi CM, Mizrahi R, Marcon H, Barsoum P, Rusjan P, Houle S, Wilson AA, Kapur S (2009) The effect of antipsychotics on the high-affinity state of D-2 and D-3 receptors A positron emission tomography study with [C-11]-(+)-PHNO. Arch Gen Psychiatry 66(6):606–615
Holford NH, Sheiner LB (1982) Kinetics of pharmacologic response. Pharmacol Ther 16(2):143–166
Innis RB, Cunningham VJ, Delforge J, Fujita M, Gjedde A, Gunn RN et al (2007) Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J Cereb Blood Flow Metab 27(9):1533–1539
Lammertsma AA, Hume SP (1996) Simplified reference tissue model for PET receptor studies. Neuro Image 4(3 Pt 1):153–158
Lecrubier Y (2003) A partial D3 receptor agonist in schizophrenia. Eur Neuropsychopharmacol 13(Suppl. 4):S167–S168
Levant B (1997) The D-3 dopamine receptor: neurobiology and potential clinical relevance. Pharmacol Rev 49(3):231–252
Lieberman JA, Stroup TS, McEvoy JP, Swartz MS, Rosenheck RA, Perkins DO, Keefe RS, Davis SM, Davis CE, Lebowitz BD, Severe J, Hsiao JK, Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators (2005) Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med 353:1209–1223
Martinez D, Slifstein M, Broft A, Mawlawi O, Hwang DR, Huang Y, Cooper T, Kegeles L, Zarahn E, Abi-Dargham A, Haber SN, Laruelle M (2003) Imaging human mesolimbic dopamine transmission with positron emission tomography. Part II: amphetamine-induced dopamine release in the functional subdivisions of the striatum. J Cereb Blood Flow Metab 23(3):285–300
Mizrahi R, Agid O, Borlido C, Suridjan I, Rusjan P, Houle S, Remington G, Wilson AA, Kapur S (2011) Effects of antipsychotics on D3 receptors: a clinical PET study in first episode antipsychotic naive patients with schizophrenia using [11C]-(+)-PHNO. Schizophr Res 131(1-3):63–68
Rabiner E, Slifstein M, Nobrega J, Plisson C, Huiban M, Raymond R, Diwan M, Wilson AA, McCormick P, Gentile G, Gunn RN, Laruelle MA (2009) In vivo quantification of regional dopamine-D3 receptor binding potential of (+)PHNO: studies in non-hman primates and transgenic mice. Synapse 63:782–793
Redden L, Rendenbach-Mueller B, Abi-Saab WM, Katz DA, Goenjian A, Robieson WZ, Wang Y, Goss SL, Greco N, Saltarelli MD (2011) A double-blind, randomized, placebo-controlled study of the dopamine D(3) receptor antagonist ABT-925 in patients with acute schizophrenia. J Clin Psychopharmacol 31:221–225
Rojo LE, Gaspar PA, Silvia H et al (2015) Metabolic syndrome and obesity among users of second-generation antipsychotics: a global challenge for modern psychopharmacology. Pharmacol Res 101:74–85
Salinas C, Weinzimmer D, Searle G, Labaree D, Ropchan J, Huang Y, Rabiner EA, Carson RE, Gunn RN (2013) Kinetic analysis of drug-target interactions with PET for characterization of pharmacological hysteresis. J Cereb Blood Flow Metab 33(5):700–707
Sernyak MJ, Leslie DL, Alarcon RD, Losonczy MF, Rosenheck R (2002) Association of diabetes mellitus with use of atypical neuroleptics in the treatment of schizophrenia. Am J Clin Psychiatry 159:561–566
Sokoloff P, Diaz J, LeFoll B, Guillin O, Leriche L, Bezard E, Gross C (2006) The dopamine D3 receptor: a therapeutic target for the treatment of neuropsychiatric disorders. CNS Neurol Disord Drug Targets 5:25–43
Sokoloff P, Leriche L, Diaz J, Louvel J, Pumain R (2013) Direct and indirect interactions of the dopamine D3 receptor with glutamate pathways: implications for the treatment of schizophrenia. Naunyn Schmiedeberg's Arch Pharmacol 386(2):107–124
Sokoloff P, Brunner V, Tonner F, Gaudoux F, Girgis R, Slifstein M, Abi-Dargham A, Leriche L (2014). F17464, a selective dopamine D3 antagonist/serotonin 5-HT1A partial agonist, as a clinical candidate with wide ranging antipsychotic-like activity in models of dopamine and glutamate dysfunctions. Neuropsychopharmacology 39 S571 ACNP 53rd Annual Meeting W153:S571
Tziortzi AC, Searle GE, Tzimopoulou S, Salinas C, Beaver JD, Jenkinson M, Laruelle M, Rabiner EA, Gunn RN (2011) Imaging dopamine receptors in humans with [11C]-(+)-PHNO: dissection of D3 signal and anatomy. NeuroImage 54(1):264–277
Wilson AA, McCormick P, Kapur S, Willeit M, Garcia A, Hussey D, Houle S, Seeman P, Ginovart N (2005) Radiosynthesis and evaluation of [11C]-(+)-4-propyl-3,4,4a,5,6,10b-hexahydro-2H-naphtho[1,2-b][1,4]oxazin -9-ol as a potential radiotracer for in vivo Imaging of the dopamine D2 high-affinity state with positron emission tomography. J Med Chem 48(12):4153–4160
Acknowledgments
The authors thank Agata Jurewicz for her aid with quality control of [11C]-(+)-PHNO; Xiaoyan Xu, Rawad Ayoub, Elizabeth Hackett, John Castrillon, and Jiayan Meng for the excellent technical support in the acquisition and analysis of the PET data; Marie-Thérèse Pétrissans, MD, for her participation in the study set-up and clinical monitoring; and Hannah Bartrum, PhD, for her editorial assistance in the preparation of this manuscript.
Funding
Institut de Recherche Pierre Fabre provided funding for the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The employers of AA-D, MS, RRG, RFS, and TBC received payment from Pierre Fabre for research support/services. AM and MG are employees of Institut de Recherche Pierre Fabre. VB, LL, FT, and PS were employees at the time of their involvement in the study. CD, PC, SJ, and SO declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 352 kb)
Rights and permissions
About this article
Cite this article
Slifstein, M., Abi-Dargham, A., Girgis, R.R. et al. Binding of the D3-preferring antipsychotic candidate F17464 to dopamine D3 and D2 receptors: a PET study in healthy subjects with [11C]-(+)-PHNO. Psychopharmacology 237, 519–527 (2020). https://doi.org/10.1007/s00213-019-05387-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-019-05387-w